Aplaviroc
Aplaviroc (INN, codenamed AK602 and GSK-873140) is a CCR5 entry inhibitor that belongs to a class of 2,5-diketopiperazines[1] developed for the treatment of HIV infection.[2][3] It was developed by GlaxoSmithKline.
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
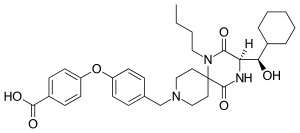
| Formula | C33H43N3O6 |
| Molar mass | 577.722 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
In October 2005, all studies of aplaviroc were discontinued due to liver toxicity concerns.[4][5] Some authors have claimed that evidence of poor efficacy may have contributed to termination of the drug's development;[6] the ASCENT study, one of the discontinued trials, showed aplaviroc to be under-effective in many patients even at high concentrations.[7]
See also
References
- Borthwick AD (July 2012). "2,5-Diketopiperazines: synthesis, reactions, medicinal chemistry, and bioactive natural products". Chemical Reviews. 112 (7): 3641–3716. doi:10.1021/cr200398y. PMID 22575049.
- Maeda K, Ogata H, Harada S, Tojo Y, Miyakawa T, Nakata H, et al. (2004). Determination of binding sites of a unique CCR5 inhibitor AK602 on human CCR5 (PDF). 11th conference on retroviruses and opportunistic infections. San Francisco, CA. Archived from the original (PDF) on November 3, 2005.
- Nakata H, Maeda K, Miyakawa T, Shibayama S, Matsuo M, Takaoka Y, et al. (February 2005). "Potent anti-R5 human immunodeficiency virus type 1 effects of a CCR5 antagonist, AK602/ONO4128/GW873140, in a novel human peripheral blood mononuclear cell nonobese diabetic-SCID, interleukin-2 receptor gamma-chain-knocked-out AIDS mouse model". Journal of Virology. 79 (4): 2087–2096. doi:10.1128/jvi.79.4.2087-2096.2005. PMC 546550. PMID 15681411.
- "Aplaviroc (GSK-873,140)". AIDSmeds.com. October 25, 2005. Archived from the original on January 13, 2007. Retrieved September 5, 2008.
- Nichols WG, Steel HM, Bonny T, Adkison K, Curtis L, Millard J, et al. (March 2008). "Hepatotoxicity observed in clinical trials of aplaviroc (GW873140)". Antimicrobial Agents and Chemotherapy. 52 (3): 858–865. doi:10.1128/aac.00821-07. PMC 2258506. PMID 18070967.
- Moyle G (December 19, 2006). "The Last Word on Aplaviroc: A CCR5 Antagonist With Poor Efficacy". The Body. Archived from the original on 6 October 2008. Retrieved September 5, 2008.
- Currier J, Lazzarin A, Sloan L, Clumeck N, Slims J, McCarty D, et al. (2008). "Antiviral activity and safety of aplaviroc with lamivudine/zidovudine in HIV-infected, therapy-naive patients: the ASCENT (CCR102881) study". Antiviral Therapy. 13 (2): 297–306. doi:10.1177/135965350801300204. PMID 18505181. S2CID 21839689.
Further reading
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.