Cadherin
Cadherins (named for "calcium-dependent adhesion") are cell adhesion molecules important in forming adherens junctions that let cells adhere to each other.[1] Cadherins are a class of type-1 transmembrane proteins, and they depend on calcium (Ca2+) ions to function, hence their name. Cell-cell adhesion is mediated by extracellular cadherin domains, whereas the intracellular cytoplasmic tail associates with numerous adaptors and signaling proteins, collectively referred to as the cadherin adhesome.
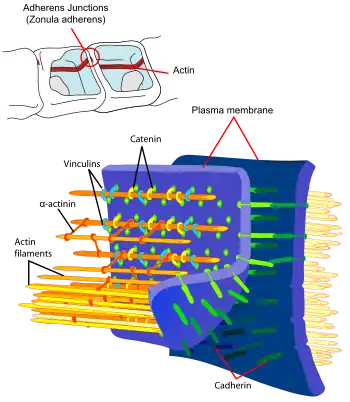
Background
The cadherin family is essential in maintaining cell-cell contact and regulating cytoskeletal complexes. The cadherin superfamily includes cadherins, protocadherins, desmogleins, desmocollins, and more.[2][3] In structure, they share cadherin repeats, which are the extracellular Ca2+-binding domains. There are multiple classes of cadherin molecules, each designated with a prefix for tissues with which it associates. Classical cadherins maintain the tone of tissues by forming a homodimer in cis while desmosomal cadherins are heterodimeric.[4] The intracellular portion of classical cadherins interacts with a complex of proteins that allows connection to the actin cytoskeleton. Although classical cadherins take a role in cell layer formation and structure formation, desmosomal cadherins focus on resisting cell damage. Desmosomal cadherins maintain the function of desmosomes that is to overturn the mechanical stress of the tissues. Similar to classical cadherins, desmosomal cadherins have a single transmembrane domain, five EC repeats, and an intracellular domain. There are two types of desmosomal cadherins: desmogleins and desmocollins. These contain an intracellular anchor and cadherin like sequence (ICS). The adaptor proteins that associate with desmosomal cadherins are plakoglobin (related to -catenin), plakophilins (p120 catenin subfamily), and desmoplakins. The major function of desmoplakins is to bind to intermediate filament by interacting with plakoglobin, which attach to the ICS of desmogleins, desmocollins and plakophilins.[4] Typical cadherins are different from other types of cadherins and consist of one or more extracellular repeat domains. The components that build an atypical cadherin are flamingo (seven pass transmembrane) and Dcad102F-like cadherins. They take part in signaling pathways instead of performing cell-cell adhesion.
Cells containing a specific cadherin subtype tend to cluster together to the exclusion of other types, both in cell culture and during development.[5] For example, cells containing N-cadherin tend to cluster with other N-cadherin-expressing cells. However, mixing speed in cell culture experiments can effect the extent of homotypic specificity.[6] In addition, several groups have observed heterotypic binding affinity (i.e., binding of different types of cadherin together) in various assays.[7][8] One current model proposes that cells distinguish cadherin subtypes based on kinetic specificity rather than thermodynamic specificity, as different types of cadherin homotypic bonds have different lifetimes.[9]
Structure
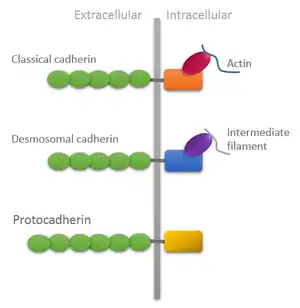
Cadherins are synthesized as polypeptides and undergo many post-translational modifications to become the proteins which mediate cell-cell adhesion and recognition.[10] These polypeptides are approximately 720–750 amino acids long. Each cadherin has a small C-terminal cytoplasmic component, a transmembrane component, and the remaining bulk of the protein is extra-cellular (outside the cell). The transmembrane component consists of single chain glycoprotein repeats.[11] Because cadherins are Ca2+ dependent, they have five tandem extracellular domain repeats that act as the binding site for Ca2+ ions.[12] Their extracellular domain interacts with two separate trans dimer conformations: strand-swap dimers (S-dimers) and X-dimers.[12] To date, over 100 types of cadherins in humans have been identified and sequenced.[13]
The functionality of cadherins relies upon the formation of two identical subunits, known as homodimers.[11] The homodimeric cadherins create cell-cell adhesion with cadherins present in the membranes of other cells through changing conformation from cis-dimers to trans-dimers.[11] Once the cell-cell adhesion between cadherins present in the cell membranes of two different cells has formed, adherens junctions can then be made when protein complexes, usually composed of α-, β-, and γ-catenins, bind to the cytoplasmic portion of the cadherin.[11] Regulatory proteins include p-120 catenin, -catenin, -catenin, and vinculin. Binding of p-120 catenin and -catenin to the homodimer increases the stability of the classical cadherin. -catenin is engaged by p120-catenin complex, where vinculin is recruited to take a role in indirect association with actin cytoskeleton.[4] However, cadherin-catenin complex can also bind directly to the actin without the help of vinculin. Moreover, the strength of cadherin adhesion can increase by dephosphorylation of p120 catenin and the binding of -catenin and vinculin.
Function
Development
Cadherins behave as both receptors and ligands for other molecules. During development, their behavior assists at properly positioning cells: they are responsible for the separation of the different tissue layers and for cellular migration.[14] In the very early stages of development, E-cadherins (epithelial cadherin) are most greatly expressed. Many cadherins are specified for specific functions in the cell, and they are differentially expressed in a developing embryo. For example, during neurulation, when a neural plate forms in an embryo, the tissues residing near the cranial neural folds have decreased N-cadherin expression.[15] Conversely, the expression of the N-cadherins remains unchanged in other regions of the neural tube that is located on the anterior-posterior axis of the vertebrate.[15] N-cadherins have different functions that maintain the cell structure, cell-cell adhesion, internal adhesions. They participate greatly in keeping the ability of the structured heart due to pumping and release blood. Because of the contribution of N-cadherins adhering strongly between the cardiomyocytes, the heart can overcome the fracture, deformation, and fatigue that can result from the blood pressure.[16] N-cadherin takes part in the development of the heart during embryogenesis, especially in sorting out of the precardiac mesoderm. N-cadherins are robustly expressed in precardiac mesoderm, but they do not take a role in cardiac linage. An embryo with N-cadherin mutation still forms the primitive heart tube; however, N-cadherin deficient mice will have difficulties in maintaining the cardiomyocytes development.[16] The myocytes of these mice will end up with dissociated myocytes surrounding the endocardial cell layer when they cannot preserve the cell adhesion due to the heart starting to pump. As a result, the cardiac outflow tract will be blocked causing cardiac swelling. The expression of different types of cadherins in the cells varies dependent upon the specific differentiation and specification of an organism during development. Cadherins play a vital role in the migration of cells through the epithelial–mesenchymal transition, which requires cadherins to form adherents junctions with neighboring cells. In neural crest cells, which are transient cells that arise in the developing organism during gastrulation and function in the patterning of the vertebrate body plan, the cadherins are necessary to allow migration of cells to form tissues or organs.[15] In addition, cadherins that are responsible in the epithelial–mesenchymal transition event in early development have also been shown to be critical in the reprogramming of specified adult cells into a pluripotent state, forming induced pluripotent stem cells (iPSCs).[1]
After development, cadherins play a role in maintaining cell and tissue structure, and in cellular movement.[13] Regulation of cadherin expression can occur through promoter methylation among other epigenetic mechanisms.[17]
Tumour metastasis
The E-cadherin–catenin complex plays a key role in cellular adhesion; loss of this function has been associated with increased invasiveness and metastasis of tumors.[18] The suppression of E-cadherin expression is regarded as one of the main molecular events responsible for dysfunction in cell-cell adhesion, which can lead to local invasion and ultimately tumor development. Because E-cadherins play an important role in tumor suppression, they are also referred to as the "suppressors of invasion".[19]
Correlation to cancer
It has been discovered that cadherins and other additional factors are correlated to the formation and growth of some cancers and how a tumor continues to grow. The E-cadherins, known as the epithelial cadherins, are on the surface of one cell and can bind with those of the same kind on another to form bridges.[20] The loss of the cell adhesion molecules, E cadherins, is causally involved in the formation of epithelial types of cancers such as carcinomas. The changes in any types of cadherin expression may not only control tumor cell adhesion but also may affect signal transduction leading to the cancer cells growing uncontrollably.[21]
In epithelial cell cancers, disrupted cell to cell adhesion might lead to the development of secondary malignant growths; they are distant from the primary site of cancer and can result from the abnormalities in the expression of E-cadherins or its associated catenins. CAMs such as the cadherin glycoproteins that normally function as the glue and holds cells together act as important mediators of cell to cell interactions. E-cadherins, on the surface of all epithelial cells, are linked to the actin cytoskeleton through interactions with catenins in the cytoplasm. Thus, anchored to the cytoskeleton, E-cadherins on the surface of one cell can bind with those on another to form bridges. In epithelial cell cancers, disrupted cell-cell adhesion that might lead to metastases can result from abnormalities in the expression of E-cadherin or its associated catenins.[20]
Correlation to endometrium and embryogenesis
This family of glycoproteins is responsible for calcium-dependent mechanism of intracellular adhesion. E-cadherins are crucial in embryogenesis during several processes, including gastrulation, neurulation, and organogenesis. Furthermore, suppression of E-cadherins impairs intracellular adhesion. The levels of these molecules increase during the luteal phase while their expression is regulated by progesterone with endometrial calcitonin.[22]
Types
| Cadherin domain (repeat) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 | |||||||||
| Identifiers | |||||||||
| Symbol | Cadherin | ||||||||
| Pfam | PF00028 | ||||||||
| InterPro | IPR002126 | ||||||||
| SMART | CA | ||||||||
| PROSITE | PDOC00205 | ||||||||
| SCOP2 | 1nci / SCOPe / SUPFAM | ||||||||
| Membranome | 114 | ||||||||
| |||||||||
| See Pfam CL0159 for other Cadherin families. | |||||||||
There are said to be over 100 different types of cadherins found in vertebrates, which can be classified into four groups: classical, desmosomal, protocadherins, and unconventional.[24][25] These large amount of diversities are accomplished by having multiple cadherin encoding genes combined with alternative RNA splicing mechanisms. Invertebrates contain fewer than 20 types of cadherins.[25]
Classical
Different members of the cadherin family are found in different locations.
- CDH1 – E-cadherin (epithelial): E-cadherins are found in epithelial tissue; not to be confused with the APC/C activator protein CDH1.
- CDH2 – N-cadherin (neural): N-cadherins are found in neurons
- CDH12 – cadherin 12, type 2 (N-cadherin 2)
- CDH3 – P-cadherin (placental): P-cadherins are found in the placenta.
Desmosomal
- Desmoglein (DSG1, DSG2, DSG3, DSG4)
- Desmocollin (DSC1, DSC2, DSC3)
Protocadherins
Protocadherins are the largest mammalian subgroup of the cadherin superfamily of homophilic cell-adhesion proteins.
- PCDH1
- PCDH7
- PCDH8
- PCDH9
- PCDH10
- PCDH11X/11Y
- PCDH12
- PCDH15
- PCDH17
- PCDH18
- PCDH19
- PCDH20
- PCDHA1
- PCDHA2
- PCDHA3
- PCDHA4
- PCDHA5
- PCDHA6
- PCDHA7
- PCDHA8
- PCDHA9
- PCDHA10
- PCDHA11
- PCDHA12
- PCDHA13
- PCDHAC1
- PCDHAC2
- PCDHB1
- PCDHB2
- PCDHB3
- PCDHB4
- PCDHB5
- PCDHB6
- PCDHB7
- PCDHB8
- PCDHB9
- PCDHB10
- PCDHB11
- PCDHB12
- PCDHB13
- PCDHB14
- PCDHB15
- PCDHB16
- PCDHB17
- PCDHB18
- PCDHGA1
- PCDHGA2
- PCDHGA3
- PCDHGA4
- PCDHGA5
- PCDHGA6
- PCDHGA7
- PCDHGA8
- PCDHGA9
- PCDHGA10
- PCDHGA11
- PCDHGA12
- PCDHGB1
- PCDHGB2
- PCDHGB3
- PCDHGB4
- PCDHGB5
- PCDHGB6
- PCDHGB7
- PCDHGC3
- PCDHGC4
- PCDHGC5
- FAT
- FAT2
- FAT4
Unconventional/ungrouped
- CDH4 – R-cadherin (retinal)
- CDH5 – VE-cadherin (vascular endothelial)
- CDH6 – K-cadherin (kidney)
- CDH7 – cadherin 7, type 2
- CDH8 – cadherin 8, type 2
- CDH9 – cadherin 9, type 2 (T1-cadherin)
- CDH10 – cadherin 10, type 2 (T2-cadherin)
- CDH11 – OB-cadherin (osteoblast)
- CDH13 – T-cadherin – H-cadherin (heart)
- CDH15 – M-cadherin (myotubule)
- CDH16 – KSP-cadherin
- CDH17 – LI cadherin (liver-intestine)
- CDH18 – cadherin 18, type 2
- CDH19 – cadherin 19, type 2
- CDH20 – cadherin 20, type 2
- CDH23 – cadherin 23 (neurosensory epithelium)
- CDH22, CDH24, CDH26, CDH28
- CELSR1, CELSR2, CELSR3
- CLSTN1, CLSTN2, CLSTN3
- DCHS1, DCHS2,
- LOC389118
- PCLKC
- RESDA1
- RET
References
- Alimperti S, Andreadis ST (May 2015). "CDH2 and CDH11 act as regulators of stem cell fate decisions". Stem Cell Research. 14 (3): 270–82. doi:10.1016/j.scr.2015.02.002. PMC 4439315. PMID 25771201.
- Hulpiau P, van Roy F (February 2009). "Molecular evolution of the cadherin superfamily". The International Journal of Biochemistry & Cell Biology. 41 (2): 349–69. doi:10.1016/j.biocel.2008.09.027. PMID 18848899.
- Angst BD, Marcozzi C, Magee AI (February 2001). "The cadherin superfamily: diversity in form and function". Journal of Cell Science. 114 (Pt 4): 629–41. doi:10.1242/jcs.114.4.629. PMID 11171368.
- Priest AV, Koirala R, Sivasankar S (December 2019). "Single-molecule studies of classical and desmosomal cadherin adhesion". Current Opinion in Biomedical Engineering. 12: 43–50. doi:10.1016/j.cobme.2019.08.006. PMC 6859941. PMID 31742239.
- Bello SM, Millo H, Rajebhosale M, Price SR (January 2012). "Catenin-dependent cadherin function drives divisional segregation of spinal motor neurons". The Journal of Neuroscience. 32 (2): 490–505. doi:10.1523/jneurosci.4382-11.2012. PMC 3292792. PMID 22238085.
- Duguay D, Foty RA, Steinberg MS (January 2003). "Cadherin-mediated cell adhesion and tissue segregation: qualitative and quantitative determinants". Developmental Biology. 253 (2): 309–23. doi:10.1016/S0012-1606(02)00016-7. PMID 12645933.
- Niessen CM, Gumbiner BM (January 2002). "Cadherin-mediated cell sorting not determined by binding or adhesion specificity". The Journal of Cell Biology. 156 (2): 389–399. doi:10.1083/jcb.200108040. PMC 2199232. PMID 11790800.
- Volk T, Cohen O, Geiger B (September 1987). "Formation of heterotypic adherens-type junctions between L-CAM-containing liver cells and A-CAM-containing lens cells". Cell. 50 (6): 987–94. doi:10.1016/0092-8674(87)90525-3. PMID 3621349. S2CID 21428349.
- Bayas MV, Leung A, Evans E, Leckband D (February 2006). "Lifetime measurements reveal kinetic differences between homophilic cadherin bonds". Biophysical Journal. 90 (4): 1385–95. Bibcode:2006BpJ....90.1385B. doi:10.1529/biophysj.105.069583. PMC 1367289. PMID 16326909.
- Harris TJ, Tepass U (July 2010). "Adherens junctions: from molecules to morphogenesis". Nature Reviews. Molecular Cell Biology. 11 (7): 502–14. doi:10.1038/nrm2927. PMID 20571587. S2CID 13638902.
- Marie PJ, Haÿ E, Modrowski D, Revollo L, Mbalaviele G, Civitelli R (January 2014). "Cadherin-mediated cell-cell adhesion and signaling in the skeleton". Calcified Tissue International. 94 (1): 46–54. doi:10.1007/s00223-013-9733-7. PMC 4272239. PMID 23657489.
- Priest AV, Shafraz O, Sivasankar S (September 2017). "Biophysical basis of cadherin mediated cell-cell adhesion". Experimental Cell Research. 358 (1): 10–13. doi:10.1016/j.yexcr.2017.03.015. PMID 28300566.
- Tepass U, Truong K, Godt D, Ikura M, Peifer M (November 2000). "Cadherins in embryonic and neural morphogenesis". Nature Reviews. Molecular Cell Biology. 1 (2): 91–100. doi:10.1038/35040042. PMID 11253370. S2CID 1632053.
- Gumbiner BM (August 2005). "Regulation of cadherin-mediated adhesion in morphogenesis". Nature Reviews. Molecular Cell Biology. 6 (8): 622–34. doi:10.1038/nrm1699. PMID 16025097. S2CID 25094246.
- Taneyhill LA, Schiffmacher AT (June 2017). "Should I stay or should I go? Cadherin function and regulation in the neural crest". Genesis. 55 (6): e23028. doi:10.1002/dvg.23028. PMC 5468476. PMID 28253541.
- Roy, Frans (2013). The Molecular Biology of Cadherins. Elsevier. pp. 264–274. ISBN 978-0-12-394311-8.
- Reinhold WC, Reimers MA, Maunakea AK, Kim S, Lababidi S, Scherf U, et al. (February 2007). "Detailed DNA methylation profiles of the E-cadherin promoter in the NCI-60 cancer cells". Molecular Cancer Therapeutics. 6 (2): 391–403. doi:10.1158/1535-7163.MCT-06-0609. PMID 17272646.
- Beavon IR (August 2000). "The E-cadherin-catenin complex in tumour metastasis: structure, function and regulation". European Journal of Cancer. 36 (13 Spec No): 1607–20. doi:10.1016/S0959-8049(00)00158-1. PMID 10959047.
- Nives Pećina-Šlaus (2003). "Tumor suppressor gene E-cadherin and its role in normal and malignant cells". Cancer Cell Int. 3 (17): 17. doi:10.1186/1475-2867-3-17. PMC 270068. PMID 14613514.
- Morales CP, Souza RF, Spechler SJ (November 2002). "Hallmarks of cancer progression in Barrett's oesophagus". Lancet. 360 (9345): 1587–9. doi:10.1016/S0140-6736(02)11569-8. PMID 12443613. S2CID 22401564.
- Cavallaro U, Schaffhauser B, Christofori G (February 2002). "Cadherins and the tumour progression: is it all in a switch?". Cancer Letters. 176 (2): 123–8. doi:10.1016/S0304-3835(01)00759-5. PMID 11804738.
- Grigorian IY, Linkova NS, Polyakova VO, Paltseva EM, Kozlov KL (January 2016). "Signaling molecules of the endometrium: Gerontological and general pathological aspects". Advances in Gerontology. 6 (1): 36–43. doi:10.1134/S2079057016010045. S2CID 87472683.
- Harrison OJ, Jin X, Hong S, Bahna F, Ahlsen G, Brasch J, et al. (February 2011). "The extracellular architecture of adherens junctions revealed by crystal structures of type I cadherins". Structure. 19 (2): 244–56. doi:10.1016/j.str.2010.11.016. PMC 3070544. PMID 21300292.
- Offermanns S, Rosenthal W (2008). Encyclopedia of Molecular Pharmacology. Springer. pp. 306–. ISBN 978-3-540-38916-3. Retrieved 14 December 2010.
- Lodish H, Berk A, Kaiser C, Krieger M, Bretscher A, Ploegh H, Amon A (2013). Molecular Cell Biology (Seventh ed.). New York: Worth Publ. p. 934. ISBN 978-1-4292-3413-9.
Further reading
- Beavon IR (August 2000). "The E-cadherin-catenin complex in tumour metastasis: structure, function and regulation". European Journal of Cancer. 36 (13 Spec No): 1607–20. doi:10.1016/S0959-8049(00)00158-1. PMID 10959047.
- Berx G, Becker KF, Höfler H, van Roy F (1998). "Mutations of the human E-cadherin (CDH1) gene". Human Mutation. 12 (4): 226–37. doi:10.1002/(SICI)1098-1004(1998)12:4<226::AID-HUMU2>3.0.CO;2-D. PMID 9744472.
- Bryant DM, Stow JL (August 2004). "The ins and outs of E-cadherin trafficking". Trends in Cell Biology. 14 (8): 427–34. doi:10.1016/j.tcb.2004.07.007. PMID 15308209.
- Chun YS, Lindor NM, Smyrk TC, Petersen BT, Burgart LJ, Guilford PJ, Donohue JH (July 2001). "Germline E-cadherin gene mutations: is prophylactic total gastrectomy indicated?". Cancer. 92 (1): 181–7. doi:10.1002/1097-0142(20010701)92:1<181::AID-CNCR1307>3.0.CO;2-J. PMID 11443625. S2CID 11052015.
- Georgolios A, Batistatou A, Manolopoulos L, Charalabopoulos K (March 2006). "Role and expression patterns of E-cadherin in head and neck squamous cell carcinoma (HNSCC)". Journal of Experimental & Clinical Cancer Research. 25 (1): 5–14. PMID 16761612.
- Hazan RB, Qiao R, Keren R, Badano I, Suyama K (April 2004). "Cadherin switch in tumor progression". Annals of the New York Academy of Sciences. 1014 (1): 155–63. Bibcode:2004NYASA1014..155H. doi:10.1196/annals.1294.016. PMID 15153430. S2CID 37486403.
- Moran CJ, Joyce M, McAnena OJ (April 2005). "CDH1 associated gastric cancer: a report of a family and review of the literature". European Journal of Surgical Oncology. 31 (3): 259–64. doi:10.1016/j.ejso.2004.12.010. PMID 15780560.
- Reynolds AB, Carnahan RH (December 2004). "Regulation of cadherin stability and turnover by p120ctn: implications in disease and cancer". Seminars in Cell & Developmental Biology. 15 (6): 657–63. doi:10.1016/j.semcdb.2004.09.003. PMID 15561585.
- Wang HD, Ren J, Zhang L (November 2004). "CDH1 germline mutation in hereditary gastric carcinoma". World Journal of Gastroenterology. 10 (21): 3088–93. doi:10.3748/wjg.v10.i21.3088. PMC 4611247. PMID 15457549.
- Wijnhoven BP, Dinjens WN, Pignatelli M (August 2000). "E-cadherin-catenin cell-cell adhesion complex and human cancer". The British Journal of Surgery. 87 (8): 992–1005. doi:10.1046/j.1365-2168.2000.01513.x. hdl:1765/56571. PMID 10931041. S2CID 3083613.
- Wilson PD (April 2001). "Polycystin: new aspects of structure, function, and regulation" (PDF). Journal of the American Society of Nephrology. 12 (4): 834–45. doi:10.1681/ASN.V124834. PMID 11274246.
- Renaud-Young M, Gallin WJ (October 2002). "In the first extracellular domain of E-cadherin, heterophilic interactions, but not the conserved His-Ala-Val motif, are required for adhesion". The Journal of Biological Chemistry. 277 (42): 39609–16. doi:10.1074/jbc.M201256200. PMID 12154084.
- Zaidel-Bar R (January 2013). "Cadherin adhesome at a glance". Journal of Cell Science. 126 (Pt 2): 373–8. doi:10.1242/jcs.111559. PMID 23547085.
- Chmelarova M, Baranova I, Ruszova E, Laco J, Hrochova K, Dvorakova E, Palicka V (October 2019). "Importance of Cadherins Methylation in Ovarian Cancer: a Next Generation Sequencing Approach". Pathology & Oncology Research. 25 (4): 1457–1465. doi:10.1007/s12253-018-0500-y. PMID 30368729. S2CID 53083365.
- Daw S, Law S (February 2021). "The functional interplay of transcription factors and cell adhesion molecules in experimental myelodysplasia including hematopoietic stem progenitor compartment". Molecular and Cellular Biochemistry. 476 (2): 535–551. doi:10.1007/s11010-020-03920-6. PMID 33011884. S2CID 222153001.
- Priest AV, Koirala R, Sivasankar S (December 2019). "Single-molecule studies of classical and desmosomal cadherin adhesion". Current Opinion in Biomedical Engineering. 12: 43–50. doi:10.1016/j.cobme.2019.08.006. PMC 6859941. PMID 31742239.
- Adu-Gyamfi EA, Czika A, Gorleku PN, Ullah A, Panhwar Z, Ruan LL, Ding YB, Wang YX (February 2021). "The Involvement of Cell Adhesion Molecules, Tight Junctions, and Gap Junctions in Human Placentation". Reproductive Sciences (Thousand Oaks, Calif.). 28 (2): 305–320. doi:10.1007/s43032-020-00364-7. PMID 33146876.
External links
- Proteopedia Cadherin - view cadherin structure in interactive 3D
- Cadherin domain in PROSITE
- The cadherin family
- The Cadherin Resource
- InterPro: IPR002126