Camps quinoline synthesis
The Camps quinoline synthesis (also known as the Camps cyclization) is a chemical reaction whereby an o-acylaminoacetophenone is transformed into two different hydroxyquinolines (products A and B) using hydroxide ion.[1][2][3][4]
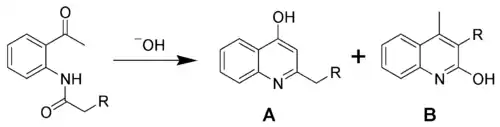
The Camps quinoline synthesis
| Camps quinoline synthesis | |
|---|---|
| Named after | Rudolph Camps |
| Reaction type | Ring forming reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000524 |
The relative proportions of the hydroxyquinolines (A and B) produced are dependent upon the reaction conditions and structure of the starting material. Although the reaction product is commonly depicted as a quinoline (the enol form), it is believed that the keto form predominates in both the solid state and in solution, making the compound a quinolone.[5]
An example of the Camps reaction is given below:[5]
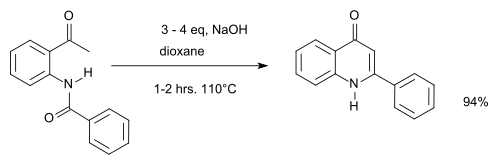
Camps quinoline synthesis
The amides of 1,3-enaminoketones react to form pyridinones-2 under similar conditions. [6]
References
- Camps, R.; Ber. 1899, 22, 3228.
- Camps, R.; Arch. Pharm. 1899, 237, 659.
- Camps, R.; Arch. Pharm. 1901, 239, 591.
- Manske, R. H. F.; Chem. Rev. 1942, 30, 127. (Review)
- Sequential Cu-Catalyzed Amidation-Base-Mediated Camps Cyclization: A Two-Step Synthesis of 2-Aryl-4-quinolones from o-Halophenones Jones, C. P.; Anderson, K. W.; Buchwald, S. L. J. Org. Chem.; (Article); 2007; 72(21); 7968-7973. doi:10.1021/jo701384n
- Camps Reaction and Related Cyclizations. A. S. Fisyuk, A. S. Kostyuchenko, D. S. Goncharov. Russ. J. Org. Chem., 2020, 56, (11), 1649–1679 (Review). doi:10.1134/s1070428020110019
See also
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.