Igmesine
Igmesine (JO-1,784) is a sigma receptor agonist (IC50 = 39 nM (rat brain)).[1][2] It has neuroprotective and antidepressant-like effects in animal studies,[3][4] as well as nootropic effects in models of age-related cognitive decline.[5] In two phase II clinical trials, igmesine was found to be effective in the treatment of depression and was as active as the comparator fluoxetine.[2] However, in a large phase III clinical trial, igmesine failed to show significant effectiveness for depression.[6] The drug has not been developed further.[7]
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C23H29N |
| Molar mass | 319.492 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Synthesis
Use patent designated in conjunction with SSRI:[8]
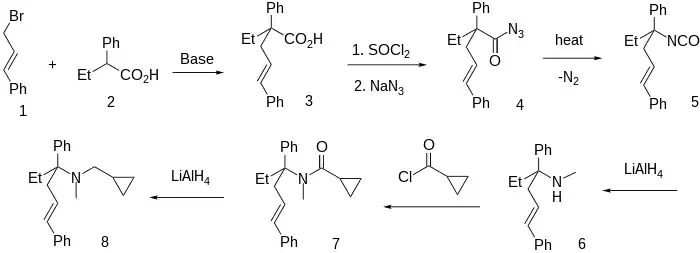
The first step involves a base catalysed alkylation between 2-phenylbutyric acid [90-27-7] (1) and cinnamyl bromide [4392-24-9] (2) giving 2-ethyl-2,5-diphenylpent-4-enoic acid, CID:53783865 (3). Stepwise treatment of the carboxylic acid with thionyl chloride and sodium azide followed by heating of this compound in an aprotic solvent causes the Curtius rearrangement of the acyl azide (4) to the isocyanate intermediate (5). The reduction of the isocyanate was carried out with lithium aluminium hydride affording the corresponding N-methylamine, i.e. N-Methyl-3,6-diphenylhex-5-en-3-amine, CID:53758312 (6). Schotten-Baumann reaction with cyclopropanoyl chloride [4023-34-1] gives the corresponding amide (7). A second step with lithium aluminium hydride reduces the amide carbonyl completing the synthesis of igmesine (8).
References
- Roman FJ, Pascaud X, Martin B, Vauché D, Junien JL (June 1990). "JO 1784, a potent and selective ligand for rat and mouse brain sigma-sites". The Journal of Pharmacy and Pharmacology. 42 (6): 439–40. doi:10.1111/j.2042-7158.1990.tb06588.x. PMID 1979628. S2CID 94116431.
- Fishback JA, Robson MJ, Xu YT, Matsumoto RR (2010). "Sigma receptors: potential targets for a new class of antidepressant drug". Pharmacol. Ther. 127 (3): 271–82. doi:10.1016/j.pharmthera.2010.04.003. PMC 3993947. PMID 20438757.
- O'Neill M, Caldwell M, Earley B, Canney M, O'Halloran A, Kelly J, Leonard BE, Junien JL (September 1995). "The sigma receptor ligand JO 1784 (igmesine hydrochloride) is neuroprotective in the gerbil model of global cerebral ischaemia". European Journal of Pharmacology. 283 (1–3): 217–25. doi:10.1016/0014-2999(95)00356-P. PMID 7498313.
- Akunne HC, Zoski KT, Whetzel SZ, Cordon JJ, Brandon RM, Roman F, Pugsley TA (July 2001). "Neuropharmacological profile of a selective sigma ligand, igmesine: a potential antidepressant". Neuropharmacology. 41 (1): 138–49. doi:10.1016/S0028-3908(01)00049-1. PMID 11445194. S2CID 25003111.
- Maurice T, Roman FJ, Su TP, Privat A (September 1996). "Beneficial effects of sigma agonists on the age-related learning impairment in the senescence-accelerated mouse (SAM)". Brain Research. 733 (2): 219–30. doi:10.1016/0006-8993(96)00565-3. PMID 8891305. S2CID 9306218.
- Hayashi T, Tsai SY, Mori T, Fujimoto M, Su TP (2011). "Targeting ligand-operated chaperone sigma-1 receptors in the treatment of neuropsychiatric disorders". Expert Opin. Ther. Targets. 15 (5): 557–77. doi:10.1517/14728222.2011.560837. PMC 3076924. PMID 21375464.
- Volz HP, Stoll KD (November 2004). "Clinical trials with sigma ligands". Pharmacopsychiatry. 37 Suppl 3: S214–20. doi:10.1055/s-2004-832680. PMID 15547788.
- Thomas Gary Heffner, et al. WO 2000041684 (Eli Lilly and Co, Warner Lambert Co LLC).
- Gilbert G. Aubard, 10 More », U.S. Patent 5,034,419 (1991 to L'oreal).
- Bagutski, V., Elford, T. G., Aggarwal, V. K. (1 February 2011). "Synthesis of Highly Enantioenriched C-Tertiary Amines From Boronic Esters: Application to the Synthesis of Igmesine". Angewandte Chemie International Edition. 50 (5): 1080–1083. doi:10.1002/anie.201006037.