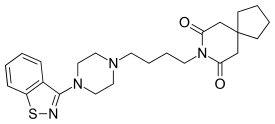
Tiospirone
Tiospirone (BMY-13,859), also sometimes called tiaspirone or tiosperone, is an atypical antipsychotic of the azapirone class.[1] It was investigated as a treatment for schizophrenia in the late 1980s and was found to have an effectiveness equivalent to those of typical antipsychotics in clinical trials but without causing extrapyramidal side effects.[2][3][4][5] However, development was halted and it was not marketed. Perospirone, another azapirone derivative with antipsychotic properties, was synthesized and assayed several years after tiospirone.[6] It was found to be both more potent and more selective in comparison and was commercialized instead.[6]
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Elimination half-life | 1.4 hours |
| Excretion | Urine |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C24H32N4O2S |
| Molar mass | 440.61 g·mol−1 |
Pharmacology
Pharmacodynamics
Tiospirone acts as a 5-HT1A receptor partial agonist, 5-HT2A, 5-HT2C, and 5-HT7 receptor inverse agonist, and D2, D4, and α1-adrenergic receptor antagonist.[7][8][9][10][11][12]
Binding profile[13]
| Receptor | Ki (nM) |
|---|---|
| 5-HT2A | 0.06 |
| 5-HT2C | 9.73 |
| 5-HT6 | 950 |
| 5-HT7 | 0.64 |
| M1 | 630 |
| M2 | 180 |
| M3 | 1290 |
| M4 | 480 |
| M5 | 3900 |
| D2 | 0.5 |
| D4 | 13.6 |
Synthesis
Note that the piperazine used is the same as in the synthesis of Ziprasidone as well as Perospirone & 1192U90 [155289-31-9].
Halogenation of 2,2'-dithiosalicylic acid [119-80-2] (1) with thionyl chloride gives 2,2'-dithiodibenzoyl chloride [19602-82-5] (2). Treatment with molecular chlorine then gives 2-(chlorothio)-benzoyl chloride [3950-02-5] (3). Treatment with ammonium hydroxide gives benzisothiazolone [2634-33-5] (4). Halogenation of the lactam with phosphoryl chloride gives 3-chloro-1,2-benzisothiazole [7716-66-7] (5). Alkylation of this with piperazine then yields 3-(1-piperazinyl)-1,2-benzisothiazole [87691-87-0] (6). The reaction of this with tetramethylenedibromide [110-52-1] (7) gave rise to CID:13232499 (8). The last step in the procedure is the reaction with azaspirodecanedione [1075-89-4] (9), thus completing the synthesis of tiaspirone (10).
See also
References
- Yevich JP, New JS, Smith DW, et al. (March 1986). "Synthesis and biological evaluation of 1-(1,2-benzisothiazol-3-yl)- and (1,2-benzisoxazol-3-yl)piperazine derivatives as potential antipsychotic agents". Journal of Medicinal Chemistry. 29 (3): 359–69. doi:10.1021/jm00153a010. PMID 2869146.
- Jain AK, Kelwala S, Moore N, Gershon S (April 1987). "A controlled clinical trial of tiaspirone in schizophrenia". International Clinical Psychopharmacology. 2 (2): 129–33. doi:10.1097/00004850-198704000-00006. PMID 2885367.
- Moore NC, Meyendorff E, Yeragani V, LeWitt PA, Gershon S (April 1987). "Tiaspirone in schizophrenia". Journal of Clinical Psychopharmacology. 7 (2): 98–101. doi:10.1097/00004714-198704000-00010. PMID 3294920.
- Borison RL, Sinha D, Haverstock S, McLarnon MC, Diamond BI (1989). "Efficacy and safety of tiospirone vs. haloperidol and thioridazine in a double-blind, placebo-controlled trial". Psychopharmacology Bulletin. 25 (2): 190–3. PMID 2574893.
- Nasrallah, Henry A.; Shriqui, Christian L (1995). Contemporary issues in the treatment of schizophrenia. Washington, DC: American Psychiatric Press. p. 313. ISBN 0-88048-681-3.
- Ishizumi K, Kojima A, Antoku F, Saji I, Yoshigi M (December 1995). "Succinimide derivatives. II. Synthesis and antipsychotic activity of N-[4-[4-(1,2-benzisothiazol-3-yl)-1-piperazinyl]butyl]-1,2-cis- cyclohexanedicarboximide (SM-9018) and related compounds". Chemical & Pharmaceutical Bulletin. 43 (12): 2139–51. doi:10.1248/cpb.43.2139. PMID 8582016.
- Sumiyoshi T, Suzuki K, Sakamoto H, et al. (February 1995). "Atypicality of several antipsychotics on the basis of in vivo dopamine-D2 and serotonin-5HT2 receptor occupancy". Neuropsychopharmacology. 12 (1): 57–64. doi:10.1016/0893-133X(94)00064-7. PMID 7766287.
- Roth BL, Tandra S, Burgess LH, Sibley DR, Meltzer HY (August 1995). "D4 dopamine receptor binding affinity does not distinguish between typical and atypical antipsychotic drugs". Psychopharmacology. 120 (3): 365–8. doi:10.1007/BF02311185. PMID 8524985. S2CID 13549491.
- Weiner DM, Burstein ES, Nash N, et al. (October 2001). "5-hydroxytryptamine2A receptor inverse agonists as antipsychotics". The Journal of Pharmacology and Experimental Therapeutics. 299 (1): 268–76. PMID 11561089.
- Herrick-Davis K, Grinde E, Teitler M (October 2000). "Inverse agonist activity of atypical antipsychotic drugs at human 5-hydroxytryptamine2C receptors". The Journal of Pharmacology and Experimental Therapeutics. 295 (1): 226–32. PMID 10991983.
- Rauly-Lestienne I, Boutet-Robinet E, Ailhaud MC, Newman-Tancredi A, Cussac D (October 2007). "Differential profile of typical, atypical and third generation antipsychotics at human 5-HT7a receptors coupled to adenylyl cyclase: detection of agonist and inverse agonist properties". Naunyn-Schmiedeberg's Archives of Pharmacology. 376 (1–2): 93–105. doi:10.1007/s00210-007-0182-6. PMID 17786406. S2CID 29337002.
- Newman-Tancredi A, Assié MB, Leduc N, Ormière AM, Danty N, Cosi C (September 2005). "Novel antipsychotics activate recombinant human and native rat serotonin 5-HT1A receptors: affinity, efficacy and potential implications for treatment of schizophrenia". The International Journal of Neuropsychopharmacology. 8 (3): 341–56. doi:10.1017/S1461145704005000. PMID 15707540.
- Roth, BL; Driscol, J (12 January 2011). "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Archived from the original on 8 November 2013. Retrieved 3 December 2013.
- Yevich, Joseph P.; New, James S.; Smith, David W.; Lobeck, Walter G.; Catt, John D.; Minielli, Joseph L.; Eison, Michael S.; Taylor, Duncan P.; Riblet, Leslie A.; Temple, Davis L. (1986). "Synthesis and biological evaluation of 1-(1,2-benzisothiazol-3-yl)- and (1,2-benzisoxazol-3-yl)piperazine derivatives as potential antipsychotic agents". Journal of Medicinal Chemistry. 29 (3): 359–369. doi:10.1021/jm00153a010.
- Davis L. Temple, Jr. & Joseph P. Yevich, U.S. Patent 4,411,901 (1983 to Mead Johnson and Co).
- Davis L. Temple, Jr. & Joseph P. Yevich, U.S. Patent 4,452,799 (1984 to Mead Johnson and Co LLC).
- Ramon Merce-Vidal, et al. U.S. Patent 5,227,486 (1993 to Esteve Pharmaceuticals SA).
