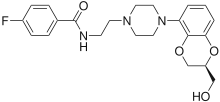
Flesinoxan
Flesinoxan (DU-29,373) is a potent and selective 5-HT1A receptor partial/near-full agonist of the phenylpiperazine class.[1][2][3] Originally developed as a potential antihypertensive drug,[1][2][4] flesinoxan was later found to possess antidepressant and anxiolytic effects in animal tests.[5][6] As a result, it was investigated in several small human pilot studies for the treatment of major depressive disorder, and was found to have robust effectiveness and very good tolerability.[7][8] However, due to "management decisions", the development of flesinoxan was stopped and it was not pursued any further.[9]
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C22H26FN3O4 |
| Molar mass | 415.465 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
In patients, flesinoxan enhances REM sleep latency, decreases body temperature, and increases ACTH, cortisol, prolactin, and growth hormone secretion.[2][8]
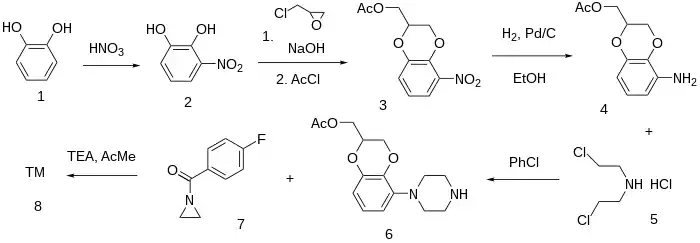
Synthesis
- See also for Halopemide (same sidechain).
The nitration of Catechol (1) with nitric acid gave 2-Nitrocatechol [3316-09-4] (2). The treatment with epichlorohydrin/NaOH (not glycerol) gave #. Acetylation with acetyl chloride gave (5-nitro-2,3-dihydro-1,4-benzodioxin-2-yl)methyl acetate [145124-52-3] (3). The reduction of the nitro group by catalytic hydrogenation led to (5-amino-2,3-dihydrobenzo[b][1,4]dioxin-2-yl)methyl acetate [145124-55-6] (4). Piperazine formation with Bis(2-chloroethyl)amine hydrochloride [821-48-7] (5) gave [145124-53-4] (6). Amide formation with 1-(4-fluorobenzoyl)aziridine [15257-81-5] (7) completed the synthesis of Flesinoxan (8).
See also
References
- Schoeffter P, Hoyer D (November 1988). "Centrally acting hypotensive agents with affinity for 5-HT1A binding sites inhibit forskolin-stimulated adenylate cyclase activity in calf hippocampus". British Journal of Pharmacology. 95 (3): 975–85. doi:10.1111/j.1476-5381.1988.tb11728.x. PMC 1854240. PMID 3207999.
- Pitchot W, Wauthy J, Legros JJ, Ansseau M (March 2004). "Hormonal and temperature responses to flesinoxan in normal volunteers: an antagonist study". European Neuropsychopharmacology. 14 (2): 151–5. doi:10.1016/S0924-977X(03)00108-1. PMID 15013031. S2CID 19082134.
- Hadrava V, Blier P, Dennis T, Ortemann C, de Montigny C (October 1995). "Characterization of 5-hydroxytryptamine1A properties of flesinoxan: in vivo electrophysiology and hypothermia study". Neuropharmacology. 34 (10): 1311–26. doi:10.1016/0028-3908(95)00098-Q. PMID 8570029. S2CID 27967032.
- Wouters W, Tulp MT, Bevan P (May 1988). "Flesinoxan lowers blood pressure and heart rate in cats via 5-HT1A receptors". European Journal of Pharmacology. 149 (3): 213–23. doi:10.1016/0014-2999(88)90651-6. PMID 2842163.
- van Hest A, van Drimmelen M, Olivier B (1992). "Flesinoxan shows antidepressant activity in a DRL 72-s screen". Psychopharmacology. 107 (4): 474–9. doi:10.1007/BF02245258. PMID 1351303. S2CID 6258207.
- Rodgers RJ, Cole JC, Davies A (August 1994). "Antianxiety and behavioral suppressant actions of the novel 5-HT1A receptor agonist, flesinoxan". Pharmacology, Biochemistry, and Behavior. 48 (4): 959–63. doi:10.1016/0091-3057(94)90205-4. PMID 7972301. S2CID 39446719.
- Grof P, Joffe R, Kennedy S, Persad E, Syrotiuk J, Bradford D (1993). "An open study of oral flesinoxan, a 5-HT1A receptor agonist, in treatment-resistant depression". International Clinical Psychopharmacology. 8 (3): 167–72. doi:10.1097/00004850-199300830-00005. PMID 8263314. S2CID 33325915.
- Ansseau M, Pitchot W, Moreno AG, Wauthy J, Papart P (2004). "Pilot study of flesinoxan, a 5-HT1A agonist, in major depression: Effects on sleep REM latency and body temperature". Human Psychopharmacology: Clinical and Experimental. 8 (4): 279–283. doi:10.1002/hup.470080407. S2CID 145758823.
- Celada P, Bortolozzi A, Artigas F (September 2013). "Serotonin 5-HT1A receptors as targets for agents to treat psychiatric disorders: rationale and current status of research". CNS Drugs. 27 (9): 703–16. doi:10.1007/s40263-013-0071-0. PMID 23757185. S2CID 31931009.
- Ennis, Michael D.; Ghazal, Nabil B. (1992). "The synthesis of (+)-and (−)-flesinoxan: Application of enzymatic resolution methodology". Tetrahedron Letters 33 (42): 6287–6290. doi:10.1016/S0040-4039(00)60954-1.
- EP0138280 idem Jan Hartog, Wouter Wouters, Lneke van Wijngaarden, U.S. Patent 4,833,142 (1989 to Duphar International Research B.V.).
External links
 Media related to Flesinoxan at Wikimedia Commons
Media related to Flesinoxan at Wikimedia Commons