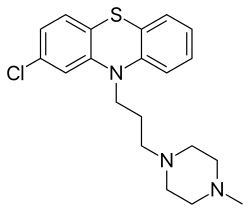
Prochlorperazine
Prochlorperazine, formerly[1] sold under the brand name Compazine among others, is a medication used to treat nausea, migraines and anxiety.[2][3][4][5] It is a less preferred medication for anxiety.[2] It may be taken by mouth, rectally, injection into a vein, or injection into a muscle.[2]
 | |
| Clinical data | |
|---|---|
| Trade names | Compazine, Stemetil, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682116 |
| License data | |
| Pregnancy category |
|
| Routes of administration | Oral administration, rectal administration, intramuscular injection, intravenous injection (IV) |
| Drug class | Typical antipsychotic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Unknown, but presumed substantial |
| Protein binding | 91–99% |
| Metabolism | Mainly Liver (CYP2D6 and/or CYP3A4) |
| Elimination half-life | 4–8 hours, differs with the method of administration |
| Excretion | Bile duct, (colored) inactive metabolites in urine |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.345 |
| Chemical and physical data | |
| Formula | C20H24ClN3S |
| Molar mass | 373.94 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
Common side effects include sleepiness, blurry vision, low blood pressure, and dizziness.[2] Serious side effects may include movement disorders including tardive dyskinesia and neuroleptic malignant syndrome.[2] Use in pregnancy and breastfeeding is generally not recommended.[6] It is a typical antipsychotic which is believed to work by reducing the action of dopamine in the brain.[2]
Prochlorperazine was approved for medical use in the United States in 1956.[2] It is available as a generic medication.[3] In 2019, it was the 313th most commonly prescribed medication in the United States, with more than 1 million prescriptions.[7]
Medical uses
Vomiting
Prochlorperazine is used to prevent vomiting caused by chemotherapy, radiation therapy and in the pre- and postoperative setting.[8] A 2015 Cochrane review found no differences in efficacy among drugs commonly used for this purpose in emergency rooms.[9]
Migraine
Prochlorperazine, generally by intravenous, is used to treat migraine.[10][11] Such use is recommended by The American Headache Society.[4] A 2019 systematic review found prochlorperazine was nearly three times as likely as metoclopramide to relieve headache within 60 minutes of administration.[10]
Labyrinthitis
In the UK prochlorperazine maleate has been used for labyrinthitis, which include not only nausea and vertigo, but spatial and temporal 'jerking' and distortion.[12]
Side effects
Sedation is very common, and extrapyramidal side effects are common and include restlessness, dystonic reactions, pseudoparkinsonism, and akathisia; the extrapyramidal symptoms can affect 2% of people at low doses, whereas higher doses may affect as many as 40% of people.[13][14]
Prochlorperazine can also cause a life-threatening condition called neuroleptic malignant syndrome (NMS). Some symptoms of NMS include high fever, stiff muscles, neck muscle spasm, confusion, irregular pulse or blood pressure, fast heart rate (tachycardia), sweating, abnormal heart rhythms (arrhythmias). Research from the Veterans Administration and United States Food and Drug Administration show injection site reactions. Adverse effects are similar in children.[8]
Warning
The FDA approved label for prochlorperazine includes a warning for increased risk of mortality in elderly patients with dementia related psychosis.[15]
Discontinuation
The British National Formulary recommends a gradual withdrawal when discontinuing antipsychotics to avoid acute withdrawal syndrome or rapid relapse.[16] Symptoms of withdrawal commonly include nausea, vomiting, and loss of appetite.[17] Other symptoms may include restlessness, increased sweating, and trouble sleeping.[17] Less commonly there may be a feeling of the world spinning, numbness, or muscle pains.[17] Symptoms generally resolve after a short period of time.[17]
There is tentative evidence that discontinuation of antipsychotics can result in psychosis.[18] It may also result in reoccurrence of the condition that is being treated.[19] Rarely tardive dyskinesia can occur when the medication is stopped.[17]
Pharmacology
| Site | Ki (nM) | Species | Ref |
|---|---|---|---|
| 5-HT1A | 5,888 | Human | [20] |
| 5-HT2A | 7.24 | Human | [20] |
| 5-HT2C | 123 | Human | [20] |
| 5-HT3 | 1349 | Human | [20] |
| 5-HT6 | 148 | Human | [20] |
| 5-HT7 | 6,760 | Human | [20] |
| α1 | 24 | Human | [21] |
| α2 | 1700 | Human | [21] |
| M1 | 776 | Human | [20] |
| M2 | 1413 | Human | [20] |
| D1 | 251 | Human | [20] |
| D2 | 2.24 | Human | [20] |
| D3 | 1.82 | Human | [20] |
| D4 | 5.37 | Human | [20] |
| H1 | 6.03 | Human | [22] |
| H3 | 17,378 | Human | [22] |
| H4 | 17783 | Human | [22] |
| DAT | 589 | Human | [20] |
| NET | 600 | Rat | [23] |
Prochlorperazine is thought to exert its antipsychotic effects by blocking dopamine receptors.[24]
Prochlorperazine is analogous to chlorpromazine; both of these agents antagonize dopaminergic D2 receptors in various pathways of the central nervous system. This D2 blockade results in antipsychotic, antiemetic and other effects. Hyperprolactinemia is a side effect of dopamine antagonists as blockade of D2 receptors within the tuberoinfundibular pathway results in increased plasma levels of prolactin due to increased secretion by lactotrophs in the anterior pituitary.
Following intramuscular injection, the antiemetic action is evident within 5 to 10 minutes and lasts for 3 to 4 hours. Rapid action is also noted after buccal treatment. With oral dosing, the start of action is delayed but the duration somewhat longer (approximately 6 hours).
Society and culture
In the UK, prochlorperazine is available for the treatment of nausea caused by migraine as a tablet dissolved in the mouth, and in Australia as a tablet swallowed whole. It is sold as a "pharmacy medicine", meaning it does not require a prescription but is only available after talking with a pharmacist.[25][26]
Marketing
Prochlorperazine is available as tablets, suppositories, and in an injectable form.[27]
As of September 2017 it was marketed under the trade names Ametil, Antinaus, Buccastem, Bukatel, Chlormeprazine, Chloropernazine, Compazine, Compro, Daolin, Dhaperazine, Emedrotec, Emetiral, Eminorm, Lotamin, Mitil, Mormal, Nautisol, Novamin, Novomit, Proazine, Procalm, Prochlorperazin, Prochlorperazine, Prochlorpérazine, Prochlorperazinum, Prochlozine, Proclorperazina, Promat, Promin, Promtil, Roumin, Scripto-metic, Seratil, Stemetil, Steremal, Vergon, Vestil, and Volimin.[27][28]
It was also marketed at that time as a combination drug for humans with paracetamol as Vestil-A, as a combination drug for veterinary use, with isopropamide as Darbazine.[27]
Research
Alexza Pharmaceuticals studied an inhaled form of prochlorperazine for the treatment of migraine through Phase II trials under the development name AT-001; development was discontinued in 2011.[29]
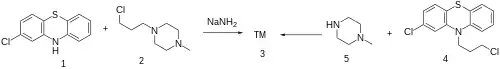
Synthesis
The alkylation of 2-chlorophenothiazine (1) and 1-(3-Chloropropyl)-4-methylpiperazine [104-16-5] (2) in the presence of sodamide gives Prochlorperazine (3); or by alkylation of 2-Chloro-10-(3-chloropropyl)phenothiazine [2765-59-5] (4) and 1-methylpiperazine (5).
References
- "Side Effects of Compazine (Prochlorperazine), Warnings, Uses". RX List. Retrieved 14 April 2021.
- "Prochlorperazine Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 3 March 2019.
- British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 385–386. ISBN 9780857113382.
- Orr SL, Friedman BW, Christie S, Minen MT, Bamford C, Kelley NE, Tepper D (June 2016). "Management of Adults With Acute Migraine in the Emergency Department: The American Headache Society Evidence Assessment of Parenteral Pharmacotherapies". Headache. 56 (6): 911–940. doi:10.1111/head.12835. PMID 27300483.
- Din L, Preuss CV (March 2022). "Prochlorperazine". StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. PMID 30725768.
- "Prochlorperazine Use During Pregnancy". Drugs.com. Retrieved 3 March 2019.
- "Prochlorperazine - Drug Usage Statistics". ClinCalc. Retrieved 16 October 2021.
- Lau Moon Lin M, Robinson PD, Flank J, Sung L, Dupuis LL (June 2016). "The Safety of Prochlorperazine in Children: A Systematic Review and Meta-Analysis". Drug Safety. 39 (6): 509–516. doi:10.1007/s40264-016-0398-9. PMID 26884326. S2CID 39349233.
- Furyk JS, Meek RA, Egerton-Warburton D (September 2015). "Drugs for the treatment of nausea and vomiting in adults in the emergency department setting". The Cochrane Database of Systematic Reviews (9): CD010106. doi:10.1002/14651858.CD010106.pub2. PMC 6517141. PMID 26411330.
- Golikhatir I, Cheraghmakani H, Bozorgi F, Jahanian F, Sazgar M, Montazer SH (May 2019). "The Efficacy and Safety of Prochlorperazine in Patients With Acute Migraine: A Systematic Review and Meta-Analysis". Headache. 59 (5): 682–700. doi:10.1111/head.13527. PMID 30990883. S2CID 119544256.
- Patniyot IR, Gelfand AA (January 2016). "Acute Treatment Therapies for Pediatric Migraine: A Qualitative Systematic Review". Headache. 56 (1): 49–70. doi:10.1111/head.12746. PMID 26790849. S2CID 25893066.
- Coatesworth AP (November 2000). "Assessment and treatment of dizziness". Journal of Neurology, Neurosurgery, and Psychiatry. 69 (5): 706–707. doi:10.1136/jnnp.69.5.706. PMC 1763384. PMID 11184241.
- Brown TM, Stoudemire A (1998). "Antipsychotics". Psychiatric Side Effects of Prescription and Over-The-Counter Medications. American Psychiatric Publishing. p. 1946. ISBN 9780880488686.
- "Procot Side Effects in Detail". Drugs.com.
- "Prochlorperazine Maleate Tablets, USP". dailymed.nlm.nih.gov. Retrieved 2022-02-02.
- Joint Formulary Committee, BMJ, ed. (March 2009). "4.2.1". British National Formulary (57 ed.). United Kingdom: Royal Pharmaceutical Society of Great Britain. p. 192. ISBN 978-0-85369-845-6.
Withdrawal of antipsychotic drugs after long-term therapy should always be gradual and closely monitored to avoid the risk of acute withdrawal syndromes or rapid relapse.
- Haddad P, Haddad PM, Dursun S, Deakin B (2004). Adverse Syndromes and Psychiatric Drugs: A Clinical Guide. OUP Oxford. pp. 207–216. ISBN 9780198527480.
- Moncrieff J (July 2006). "Does antipsychotic withdrawal provoke psychosis? Review of the literature on rapid onset psychosis (supersensitivity psychosis) and withdrawal-related relapse". Acta Psychiatrica Scandinavica. 114 (1): 3–13. doi:10.1111/j.1600-0447.2006.00787.x. PMID 16774655. S2CID 6267180.
- Sacchetti E, Vita A, Siracusano A, Fleischhacker W (2013). Adherence to Antipsychotics in Sc. Springer Science & Business Media. p. 85. ISBN 9788847026797.
- Silvestre JS, Prous J (June 2005). "Research on adverse drug events. I. Muscarinic M3 receptor binding affinity could predict the risk of antipsychotics to induce type 2 diabetes". Methods and Findings in Experimental and Clinical Pharmacology. 27 (5): 289–304. doi:10.1358/mf.2005.27.5.908643. PMID 16082416.
- Richelson E, Nelson A (August 1984). "Antagonism by neuroleptics of neurotransmitter receptors of normal human brain in vitro". European Journal of Pharmacology. 103 (3–4): 197–204. doi:10.1016/0014-2999(84)90478-3. PMID 6149136.
- Appl H, Holzammer T, Dove S, Haen E, Strasser A, Seifert R (February 2012). "Interactions of recombinant human histamine H1R, H2R, H3R, and H4R receptors with 34 antidepressants and antipsychotics". Naunyn-Schmiedeberg's Archives of Pharmacology. 385 (2): 145–170. doi:10.1007/s00210-011-0704-0. PMID 22033803. S2CID 14274150.
- Richelson E, Pfenning M (September 1984). "Blockade by antidepressants and related compounds of biogenic amine uptake into rat brain synaptosomes: most antidepressants selectively block norepinephrine uptake". European Journal of Pharmacology. 104 (3–4): 277–286. doi:10.1016/0014-2999(84)90403-5. PMID 6499924.
- Ebadi MS (2007). Desk Reference of Clinical Pharmacology.
- "Buccastem M - Summary of Product Characteristics (SPC) - (eMC)". UK Electronic Medicines Compendium. 16 February 2016. Retrieved 19 September 2017.
- "Medicines information". NHS Choices. Retrieved 19 September 2017.
- "Prochlorperazine international brands". Drugs.com. Retrieved 19 September 2017.
- "Compazine (Prochlorperazine) Patient Information: Side Effects and Drug Images at RxList". RxList.
- Chua AL, Silberstein S (September 2016). "Inhaled drug therapy development for the treatment of migraine". Expert Opinion on Pharmacotherapy. 17 (13): 1733–1743. doi:10.1080/14656566.2016.1203901. PMID 27416108. S2CID 11724776.
- J. Gen. Chem. USSR (Engl. Transl.), vol. 32, p. 1915 - 1919,1892 – 1895.
- GB780193 idem Horclois Raymond Jacques, U.S. Patent 2,902,484 (1959 to Rhone Poulenc Sa).