Ciclazindol
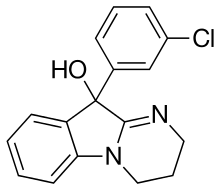
Ciclazindol (WY-23409) is an antidepressant and anorectic drug of the tetracyclic chemical class that was developed in the mid to late 1970s, but was never marketed.[2][3] It acts as a norepinephrine reuptake inhibitor, and to a lesser extent as a dopamine reuptake inhibitor.[2][4] Ciclazindol has no effects on the SERT, 5-HT receptors, mACh receptors, or α-adrenergic receptors, and has only weak affinity for the H1 receptor.[4][5][6] As suggested by its local anesthetic properties,[5] ciclazindol may also inhibit sodium channels. It is known to block potassium channels as well.[7][8]
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Pharmacokinetic data | |
| Metabolism | Renal[1] |
| Elimination half-life | ~32 hours[1] |
| Excretion | Urine, feces[1] |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C17H15ClN2O |
| Molar mass | 298.77 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
See also
References
- Swaisland AJ, Franklin RA, Southgate PJ, Coleman AJ (February 1977). "The pharmacokinetics of ciclazindol (Wy 23409) in human volunteers". British Journal of Clinical Pharmacology. 4 (1): 61–5. doi:10.1111/j.1365-2125.1977.tb00668.x. PMC 1428987. PMID 843425.
- Ghose K, Rama Rao VA, Bailey J, Coppen A (April 1978). "Antidepressant activity and pharmacological interactions of ciclazindol". Psychopharmacology. 57 (1): 109–14. doi:10.1007/BF00426966. PMID 96461. S2CID 12961802.
- Levine S (1979). "A controlled comparative trial of a new antidepressant, ciclazindol". The Journal of International Medical Research. 7 (1): 1–6. doi:10.1177/030006057900700101. PMID 369921. S2CID 28112402.
- Oh VM, Ehsanullah RS, Leighton M, Kirby MJ (January 1979). "Influence of ciclazindol on monoamine uptake and CNS function in normal subjects". Psychopharmacology. 60 (2): 177–81. doi:10.1007/BF00432290. PMID 106428. S2CID 24199961.
- Waterfall JF, Smith MA, Gaston WH, Maher J, Warburton G (July 1979). "Cardiovascular and autonomic actions of ciclazindol and tricyclic antidepressants". Archives Internationales de Pharmacodynamie et de Thérapie. 240 (1): 116–36. PMID 507990.
- Gardner CR, Wilford AE (January 1980). "The effects of mianserine, amitriptyline, ciclazindol and viloxazine on presynaptic alpha-receptors in isolated rat atria [proceedings]". British Journal of Pharmacology. 68 (1): 184P–185P. doi:10.1111/j.1476-5381.1980.tb10705.x. PMC 2044122. PMID 6244029.
- Noack T, Edwards G, Deitmer P, et al. (May 1992). "The involvement of potassium channels in the action of ciclazindol in rat portal vein". British Journal of Pharmacology. 106 (1): 17–24. doi:10.1111/j.1476-5381.1992.tb14286.x. PMC 1907450. PMID 1504725.
- Lee K, Khan RN, Rowe IC, et al. (April 1996). "Ciclazindol inhibits ATP-sensitive K+ channels and stimulates insulin secretion in CR1-G1 insulin-secreting cells". Molecular Pharmacology. 49 (4): 715–20. PMID 8609901.
| Central |
| ||||||
|---|---|---|---|---|---|---|---|
| Peripheral |
| ||||||
| |||||||
| DAT (DRIs) |
| ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NET (NRIs) |
| ||||||||||||||
| SERT (SRIs) |
| ||||||||||||||
| VMATs |
| ||||||||||||||
| Others |
| ||||||||||||||
See also: Receptor/signaling modulators • Monoamine releasing agents • Adrenergics • Dopaminergics • Serotonergics • Monoamine metabolism modulators • Monoamine neurotoxins | |||||||||||||||
| Calcium |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Potassium |
| ||||||||||||||||||||||||
| Sodium |
| ||||||||||||||||||||||||
| Chloride |
| ||||||||||||||||||||||||
| Others |
| ||||||||||||||||||||||||
See also: Receptor/signaling modulators • Transient receptor potential channel modulators | |||||||||||||||||||||||||
| Classes |
|
|---|---|
| Antidepressants (TCAs and TeCAs) |
|
| Antihistamines |
|
| Antipsychotics |
|
| Anticonvulsants | |
| Others |
|
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.