AL-1095
AL-1095,[1] is a centrally acting stimulant drug with comparable effects to amphetamine,[2] developed by Bristol in the 1970s.[3]
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
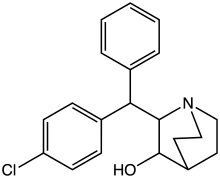
| Formula | C20H22ClNO |
| Molar mass | 327.85 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Synthesis
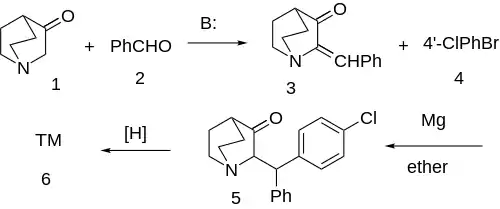
Correction:[4]
The first-step is a mixed-aldol condensation between 3-quinuclidinone [3731-38-2] (1) and benzaldehyde (2) gives 2-benzylidene-3-oxoquinuclidine [24123-89-5] (3). The conjugate addition of the Grignard reagent formed from 4-bromochlorobenzene [106-39-8] (4) to the enone gives the benzhydryl (5). MPV reduction of the carbonyl gives the syn stereoisomers, whereas borohydride gave trans. Both diastereoisomers are active but in only one of the enantiomers.
References
- US 3506673, Warawa ED, Mueller NJ, "2-(4'Halo)-Benzhydryl-3-Quinuclidinols", issued 14 April 1970, assigned to Aldrich Chemical Company
- Hedblom P, Grinspoon L (1975). The Speed Culture: Amphetamine Use and Abuse in America. Cambridge: Harvard University Press. p. 50. ISBN 0-674-83192-6.
- Warawa EJ, Mueller NJ (January 1975). "Quinuclidine Chemistry. 3. β-cis-2-(4'-Chlorobenzhydryl)-3-quinuclidinol, a New Central Nervous System Stimulant. Importance of the Benzhydryl Configuration". Journal of Medicinal Chemistry. 18 (1): 71–4. doi:10.1021/jm00235a016. PMID 803245.
- Warawa, E., Mueller, N., & Gylys, J. (1975). Additions and Corrections - Quinuclidine Chemistry. 3. β-cis-2-(4’-Chlorobenzhydryl)-3-quinuclidinol, a New Central Nervous System Stimulant. Importance of the Benzhydryl Configuration. Journal of Medicinal Chemistry, 18(12), 1275–1275. https://doi.org/10.1021/jm00246a600.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.