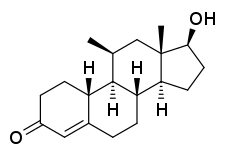
11β-Methyl-19-nortestosterone
11β-Methyl-19-nortestosterone (11β-MNT) is a synthetic and orally active anabolic–androgenic steroid (AAS) and a derivative of nandrolone (19-nortestosterone) which was developed by the Contraceptive Development Branch (CDB) of the National Institute of Child Health and Human Development (NICHD) and has not been marketed at this time.[1][2][3]
 | |
| Clinical data | |
|---|---|
| Other names | 11β-MNT; 11β-Methylestr-4-en-17β-ol-3-one |
| Routes of administration | By mouth |
| Drug class | Androgen; Anabolic steroid; Progestogen |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C19H28O2 |
| Molar mass | 288.431 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
The C17β dodecylcarbonate ester of 11β-MNT, 11β-methyl-19-nortestosterone 17β-dodecylcarbonate (11β-MNTDC) (CDB-4754), is a prodrug of 11β-MNT.[1][2][3] Along with the closely related AAS dimethandrolone (7α,11β-dimethyl-19-nortestosterone; CDB-1321) and its ester prodrug dimethandrolone undecanoate (CDB-4521), 11β-MNT and 11β-MNTDC are under investigation as potential male contraceptives and to treat male hypogonadism.[1][2][3]
Side effects
Pharmacology
Pharmacodynamics
11β-MNT does not undergo aromatization into the corresponding estrogenic metabolite 11β-methylestradiol, and for this reason, has no potential for estrogenic side effects such as gynecomastia.[2] In addition, unlike testosterone, 11β-MNT does not appear to undergo 5α-reduction into the corresponding 5α-dihydrogenated metabolite 5α-dihydro-11β-MNT (5α-DHMNT).[3] This conclusion is based on the fact that 5α-DHMNT is 4 to 8 times as potent as 11β-MNT in terms of androgenicity in animal bioassays, yet the co-administration of the 5α-reductase inhibitor dutasteride with 11β-MNT had no influence on its potency in assays using tissues that express 5α-reductase like the ventral prostate and seminal vesicles.[3] Due to lack of potentiation by 5α-reductase in androgenic tissues like the skin, hair follicles, and prostate gland, 11β-MNT may have a lower risk of certain side effects such as oily skin, acne, androgenic alopecia (pattern hair loss), prostate enlargement, and prostate cancer than testosterone and certain other AAS.[3]
Similarly to nandrolone, dimethandrolone, and other 19-nortestosterone derivatives, 11β-MNT has been found to possess progestogenic activity.[4] Because of its dual activity as an AAS and progestogen, 11β-MNT may have greater efficacy in suppression of spermatogenesis and hence male fertility than pure AAS like testosterone.[4]
Oral 11β-MNT has shown little to no potential for hepatotoxicity in animals, similarly to testosterone but unlike 17α-alkylated AAS like methyltestosterone.[5] The drug notably shows a much lower hepatotoxic potential than dimethandrolone and trestolone (7α-methyl-19-nortestosterone; MENT), which may have an increased risk due to their shared C7α methyl group (although a risk that is still significantly lower than that of 17α-alkylated AAS).[5]
Chemistry
11β-MNT, or 11β-methyl-19-nortestosterone, also known as 11β-methylestr-4-en-17β-ol-3-one, is a synthetic estrane steroid and a non-17α-alkylated derivative of nandrolone (19-nortestosterone).[4]
References
- Attardi BJ, Marck BT, Matsumoto AM, Koduri S, Hild SA (2011). "Long-term effects of dimethandrolone 17β-undecanoate and 11β-methyl-19-nortestosterone 17β-dodecylcarbonate on body composition, bone mineral density, serum gonadotropins, and androgenic/anabolic activity in castrated male rats". J. Androl. 32 (2): 183–92. doi:10.2164/jandrol.110.010371. PMID 20798389.
- Attardi BJ, Pham TC, Radler LC, Burgenson J, Hild SA, Reel JR (2008). "Dimethandrolone (7alpha,11beta-dimethyl-19-nortestosterone) and 11beta-methyl-19-nortestosterone are not converted to aromatic A-ring products in the presence of recombinant human aromatase". J. Steroid Biochem. Mol. Biol. 110 (3–5): 214–22. doi:10.1016/j.jsbmb.2007.11.009. PMC 2575079. PMID 18555683.
- Attardi BJ, Hild SA, Koduri S, Pham T, Pessaint L, Engbring J, Till B, Gropp D, Semon A, Reel JR (2010). "The potent synthetic androgens, dimethandrolone (7α,11β-dimethyl-19-nortestosterone) and 11β-methyl-19-nortestosterone, do not require 5α-reduction to exert their maximal androgenic effects". J. Steroid Biochem. Mol. Biol. 122 (4): 212–8. doi:10.1016/j.jsbmb.2010.06.009. PMC 2949447. PMID 20599615.
- Attardi BJ, Hild SA, Reel JR (2006). "Dimethandrolone undecanoate: a new potent orally active androgen with progestational activity". Endocrinology. 147 (6): 3016–26. doi:10.1210/en.2005-1524. PMID 16497801.
- Hild SA, Attardi BJ, Koduri S, Till BA, Reel JR (2010). "Effects of synthetic androgens on liver function using the rabbit as a model". J. Androl. 31 (5): 472–81. doi:10.2164/jandrol.109.009365. PMC 2943539. PMID 20378929.