Bolandiol
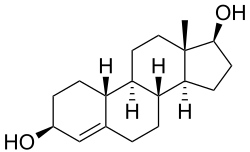
Bolandiol (INN, also known as 19-nor-4-androstenediol, estr-4-ene-3β,17β-diol, or 3β-dihydronandrolone) is an anabolic-androgenic steroid (AAS) that was never marketed.[1] However, a dipropionate ester derivative, bolandiol dipropionate, has been marketed.[1][2] Bolandiol and its dipropionate ester are unique among AASs in that they reportedly also possesses estrogenic and progestogenic activity.[3][4]
 | |
| Clinical data | |
|---|---|
| Other names | 19-Norandrostenediol; 19-Nor-4-androstene-3β,17β-diol; Estr-4-ene-3β,17β-diol; 3β-Dihydronandrolone |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C18H28O2 |
| Molar mass | 276.420 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Bolandiol is on the World Anti-Doping Agency's list of prohibited substances,[5] and is therefore banned from use in most major sports. It is a potential metabolic precursor to nandrolone.[6] However, several clinical studies have concluded that bolandiol does not alter strength, body composition, or exercise performance.[7][8]
See also
- 4-Androstenediol
- 19-Nor-5-androstenediol
- 19-Nor-5-androstenedione
- Bolandione (19-nor-4-androstenedione)
- Bolenol (17α-ethyl-19-nor-5-androstenol)
References
- Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 899–. ISBN 978-1-4757-2085-3.
- Hyde TE, Gengenbach MS (2007). Conservative Management of Sports Injuries. Jones & Bartlett Learning. pp. 1100–. ISBN 978-0-7637-3252-3.
- Action QA, ed. (9 January 2012). Hormones: Advances in Research and Application (2011 ed.). ScholarlyEditions. pp. 59–. ISBN 978-1-4649-2242-8.
- Attardi BJ, Page ST, Hild SA, Coss CC, Matsumoto AM (February 2010). "Mechanism of action of bolandiol (19-nortestosterone-3beta,17beta-diol), a unique anabolic steroid with androgenic, estrogenic, and progestational activities". The Journal of Steroid Biochemistry and Molecular Biology. 118 (3): 151–61. doi:10.1016/j.jsbmb.2009.11.008. PMC 2831543. PMID 19941958.
- "The World Anti-Doping Code: The 2012 Prohibited List" (PDF). World Anti-Doping Agency. Archived from the original (PDF) on 2012-05-13. Retrieved 2012-07-17.
- Dehennin L, Bonnaire Y, Plou P (January 2002). "Human nutritional supplements in the horse: comparative effects of 19-norandrostenedione and 19-norandrostenediol on the 19-norsteroid profile and consequences for doping control". Journal of Chromatography. B, Analytical Technologies in the Biomedical and Life Sciences. 766 (2): 257–263. doi:10.1016/S0378-4347(01)00506-0. PMID 11824814.
- Van Gammeren D, Falk D, Antonio J (May 2001). "The effects of supplementation with 19-nor-4-androstene-3,17-dione and 19-nor-4-androstene-3,17-diol on body composition and athletic performance in previously weight-trained male athletes". European Journal of Applied Physiology. 84 (5): 426–431. doi:10.1007/s004210100395. PMID 11417430. S2CID 13129479.
- van Gammeren D, Falk D, Antonio J (September 2002). "Effects of norandrostenedione and norandrostenediol in resistance-trained men". Nutrition. 18 (9): 734–737. doi:10.1016/s0899-9007(02)00834-1. PMID 12297208.
| ARTooltip Androgen receptor |
| ||||||
|---|---|---|---|---|---|---|---|
| GPRC6A |
| ||||||
| |||||||
| ERTooltip Estrogen receptor |
| ||||||
|---|---|---|---|---|---|---|---|
| GPERTooltip G protein-coupled estrogen receptor |
| ||||||
| |||||||
| PRTooltip Progesterone receptor |
| ||||||
|---|---|---|---|---|---|---|---|
| mPRTooltip Membrane progesterone receptor (PAQRTooltip Progestin and adipoQ receptor) |
| ||||||
| |||||||
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.