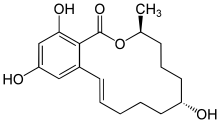
α-Zearalenol
α-Zearalenol is a nonsteroidal estrogen of the resorcylic acid lactone group related to mycoestrogens found in Fusarium spp.[1] It is the α epimer of β-zearalenol and along with β-zearalenol is a major metabolite of zearalenone formed mainly in the liver but also to a lesser extent in the intestines during first-pass metabolism.[2][3] A relatively low proportion of β-zearalenol is formed from zearalenone compared to α-zearalenol in humans.[3] α-Zearalenol is about 3- to 4-fold more potent as an estrogen relative to zearalenone.[1]
 | |
| Clinical data | |
|---|---|
| Other names | alpha-Zearalenol; trans-Zearalenol; 2,4-Dihydroxy-6-(6α,10-dihydroxy-trans-1-undecenyl)benzoic acid μ-lactone |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.264.264 |
| Chemical and physical data | |
| Formula | C18H24O5 |
| Molar mass | 320.385 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
See also
- Taleranol (β-zearalanol)
- Zeranol (α-zearalanol)
- Zearalanone
References
- Chelkowski J (28 June 2014). Fusarium: Mycotoxins, Taxonomy, Pathogenicity. Elsevier Science. pp. 85–. ISBN 978-1-4832-9785-9.
- Magan N, Olsen M (2004). Mycotoxins in Food: Detection and Control. Woodhead Publishing. pp. 356–. ISBN 978-1-85573-733-4.
- Eriksen GS (1998). Fusarium Toxins in Cereals: A Risk Assessment. Nordic Council of Ministers. pp. 61–. ISBN 978-92-893-0149-7.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.