Dirlotapide
Dirlotapide is a drug used to treat obesity in dogs.[1] It is manufactured by Pfizer and Zoetis and marketed under the brand name Slentrol.
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Slentrol |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | By mouth |
| ATCvet code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | High[1] |
| Metabolism | Liver |
| Elimination half-life | 5–18 hours (increased with repeated dosing) |
| Excretion | Feces, urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
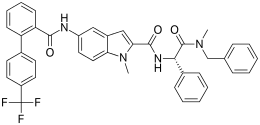
| Formula | C40H33F3N4O3 |
| Molar mass | 674.724 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
It works as a gut-selective microsomal triglyceride transfer protein (MTTP or MTP) inhibitor.[2] This blocks the assembly and release of lipoproteins into the bloodstream, thereby reducing fat absorption. It also elicits a satiety signal from lipid-filled cells lining the intestine.
Usage
It is supplied as an oral solution. It is not intended for use in humans, cats, birds, rodents, or other animals.
Dirlotapide is used to manage obesity in dogs and helps by reducing appetite. It is used as part of an overall weight control program that also includes proper diet and exercise, under the supervision of a veterinarian. Side effects may include vomiting, diarrhea, lethargy, drooling, or uncoordination. Allergic reaction to the medication may include, facial swelling, hives, scratching, sudden onset of diarrhea, vomiting, shock, seizures, pale gums, cold limbs, or coma.
Regulation and safety
On January 5, 2007, the U.S. Food and Drug Administration (FDA) approved Slentrol, the first time the FDA has approved a drug for obese dogs.[3][4]
However, concerns have since been raised, since 2010, about adverse effects[5] that might more strongly affect particular breeds.[6]
Dirlotapide (under the brand name Slentrol) was authorized for use in the EU by the European Medicines Agency for helping weight loss in dogs, but has since been withdrawn from the market in the EU.[7]
References
- "Slentrol (dirlotapide) Oral Solution (5 mg/ml, 1%) for Use in Dogs Only. Full Prescribing Information" (PDF). zoetisUS.com. Pfizer Animal Health. Div. of Pfizer Inc. NY, NY 10017. Archived from the original (PDF) on 8 December 2015. Retrieved 28 November 2015.
- US 20160002264, "Carbocyclic- and Heterocyclic-Substituted Hexahydropyrano[3,4-d][1,3]Thiazin-2-Amine Compounds", assigned to Pfizer, Inc.
- Klonoff DC (May 2007). "Dirlotapide, a U.S. Food and Drug Administration-approved first-in-class obesity drug for dogs-will humans be next?". Journal of Diabetes Science and Technology. 1 (3): 314–6. doi:10.1177/193229680700100301. PMC 2769592. PMID 19885086.
- Bridges A. "FDA approves 1st drug for obese dogs". Associated Press. Archived from the original on 2007-01-08. Retrieved January 6, 2007 – via Yahoo! News.
- Edwards J (15 October 2010). "Why Pfizer Doesn't Want the FDA Studying Its Diet Drug for Dogs". CBS News. Retrieved 2018-07-05.
- Hobson K (14 October 2010). "FDA Sniffing Around Pfizer's Doggy Weight-Loss Drug Slentrol". WSJ. Retrieved 2018-07-05.
- German AJ (October 2016). "Weight management in obese pets: the tailoring concept and how it can improve results". Acta Veterinaria Scandinavica. 58 (Suppl 1): 57. doi:10.1186/s13028-016-0238-z. PMC 5073926. PMID 27766974.