Eukaryotic transcription
Eukaryotic transcription is the elaborate process that eukaryotic cells use to copy genetic information stored in DNA into units of transportable complementary RNA replica.[1] Gene transcription occurs in both eukaryotic and prokaryotic cells. Unlike prokaryotic RNA polymerase that initiates the transcription of all different types of RNA, RNA polymerase in eukaryotes (including humans) comes in three variations, each translating a different type of gene. A eukaryotic cell has a nucleus that separates the processes of transcription and translation. Eukaryotic transcription occurs within the nucleus where DNA is packaged into nucleosomes and higher order chromatin structures. The complexity of the eukaryotic genome necessitates a great variety and complexity of gene expression control.
| Part of a series on |
| Genetics |
|---|
 |
|
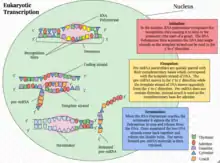
Eukaryotic transcription proceeds in three sequential stages: initiation, elongation, and termination.[1]
The RNAs transcribed serve diverse functions. For example, structural components of the ribosome are transcribed by RNA polymerase I. Protein coding genes are transcribed by RNA polymerase II into messenger RNAs (mRNAs) that carry the information from DNA to the site of protein synthesis.[1] More abundantly made are the so-called non-coding RNAs account for the large majority of the transcriptional output of a cell.[2] These non-coding RNAs perform a variety of important cellular functions.[2]
RNA polymerase
Eukaryotes have three nuclear RNA polymerases, each with distinct roles and properties.[3][4]
| Name | Location | Product |
| RNA Polymerase I (Pol I, Pol A) | nucleolus | larger ribosomal RNA (rRNA) (28S, 18S, 5.8S) |
| RNA Polymerase II (Pol II, Pol B) | nucleus | messenger RNA (mRNA), most small nuclear RNAs (snRNAs), small interfering RNA (siRNAs) and microRNA (miRNA). |
| RNA Polymerase III (Pol III, Pol C) | nucleus (and possibly the nucleolus-nucleoplasm interface) | transfer RNA (tRNA), other small RNAs (including the small 5S ribosomal RNA (5s rRNA), snRNA U6, signal recognition particle RNA (SRP RNA) and other stable short RNAs |
RNA polymerase I (Pol I) catalyses the transcription of all rRNA genes except 5S.[3][4] These rRNA genes are organised into a single transcriptional unit and are transcribed into a continuous transcript. This precursor is then processed into three rRNAs: 18S, 5.8S, and 28S. The transcription of rRNA genes takes place in a specialised structure of the nucleus called the nucleolus,[5] where the transcribed rRNAs are combined with proteins to form ribosomes.[6]
RNA polymerase II (Pol II) is responsible for the transcription of all mRNAs, some snRNAs, siRNAs, and all miRNAs.[3][4] Many Pol II transcripts exist transiently as single strand precursor RNAs (pre-RNAs) that are further processed to generate mature RNAs.[1] For example, precursor mRNAs (pre-mRNAs) are extensively processed before exiting into the cytoplasm through the nuclear pore for protein translation.
RNA polymerase III (Pol III) transcribes small non-coding RNAs, including tRNAs, 5S rRNA, U6 snRNA, SRP RNA, and other stable short RNAs such as ribonuclease P RNA.[7]
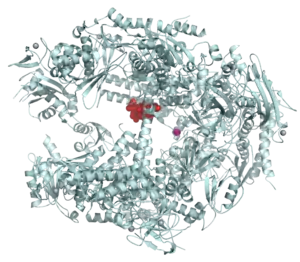
RNA Polymerases I, II, and III contain 14, 12, and 17 subunits, respectively.[8] All three eukaryotic polymerases have five core subunits that exhibit homology with the β, β’, αI, αII, and ω subunits of E. coli RNA polymerase. An identical ω-like subunit (RBP6) is used by all three eukaryotic polymerases, while the same α-like subunits are used by Pol I and III. The three eukaryotic polymerases share four other common subunits among themselves. The remaining subunits are unique to each RNA polymerase. The additional subunits found in Pol I and Pol III relative to Pol II, are homologous to Pol II transcription factors.[8]
Crystal structures of RNA polymerases I[9] and II[10] provide an opportunity to understand the interactions among the subunits and the molecular mechanism of eukaryotic transcription in atomic detail.
The carboxyl terminal domain (CTD) of RPB1, the largest subunit of RNA polymerase II, plays an important role in bringing together the machinery necessary for the synthesis and processing of Pol II transcripts.[11] Long and structurally disordered, the CTD contains multiple repeats of heptapeptide sequence YSPTSPS that are subject to phosphorylation and other posttranslational modifications during the transcription cycle. These modifications and their regulation constitute the operational code for the CTD to control transcription initiation, elongation and termination and to couple transcription and RNA processing.[11]
Initiation
The initiation of gene transcription in eukaryotes occurs in specific steps.[1] First, an RNA polymerase along with general transcription factors binds to the promoter region of the gene to form a closed complex called the preinitiation complex. The subsequent transition of the complex from the closed state to the open state results in the melting or separation of the two DNA strands and the positioning of the template strand to the active site of the RNA polymerase. Without the need of a primer, RNA polymerase can initiate the synthesis of a new RNA chain using the template DNA strand to guide ribonucleotide selection and polymerization chemistry.[1] However, many of the initiated syntheses are aborted before the transcripts reach a significant length (~10 nucleotides). During these abortive cycles, the polymerase keeps making and releasing short transcripts until it is able to produce a transcript that surpasses ten nucleotides in length. Once this threshold is attained, RNA polymerase passes the promoter and transcription proceeds to the elongation phase.[1]

Eukaryotic promoters and general transcription factors
Pol II-transcribed genes contain a region in the immediate vicinity of the transcription start site (TSS) that binds and positions the preinitiation complex. This region is called the core promoter because of its essential role in transcription initiation.[12][13] Different classes of sequence elements are found in the promoters. For example, the TATA box is the highly conserved DNA recognition sequence for the TATA box binding protein, TBP, whose binding initiates transcription complex assembly at many genes.
Eukaryotic genes also contain regulatory sequences beyond the core promoter. These cis-acting control elements bind transcriptional activators or repressors to increase or decrease transcription from the core promoter. Well-characterized regulatory elements include enhancers, silencers, and insulators. These regulatory sequences can be spread over a large genomic distance, sometimes located hundreds of kilobases from the core promoters.[1]
General transcription factors are a group of proteins involved in transcription initiation and regulation.[1] These factors typically have DNA-binding domains that bind specific sequence elements of the core promoter and help recruit RNA polymerase to the transcriptional start site. General transcription factors for RNA polymerase II include TFIID, TFIIA, TFIIB, TFIIF, TFIIE, and TFIIH.[1][14][15]
Assembly of preinitiation complex
The transcription, a complete set of general transcription factors and RNA polymerase need to be assembled at the core promoter to form the ~2.5 million Dalton preinitiation complex.[16] For example, for promoters that contain a TATA box near the TSS, the recognition of TATA box by the TBP subunit of TFIID initiates the assembly of a transcription complex. The next proteins to enter are TFIIA and TFIIB, which stabilize the DNA-TFIID complex and recruit Pol II in association with TFIIF and additional transcription factors. TFIIB serves as the bridge between the TATA-bound TBP and the RNA polymerase. It also helps to place the active centre of the polymerase in the correct position to initiate transcription. One of the last transcription factors to be recruited to the preinitiation complex is TFIIH, which plays an important role in promoter melting and escape.[17]

Promoter melting and open complex formation
For pol II-transcribed genes, and unlike bacterial RNA polymerase, promoter melting requires hydrolysis of ATP and is mediated by TFIIH.[17] TFIIH is a ten-subunit protein, including both ATPase and protein kinase activities.[18] While the upstream promoter DNA is held in a fixed position by TFIID, TFIIH pulls downstream double-stranded DNA into the cleft of the polymerase, driving the separation of DNA strands and the transition of the preinitiation complex from the closed to open state. TFIIB aids in open complex formation by binding the melted DNA and stabilizing the transcription bubble.
Abortive initiation
Once the initiation complex is open, the first ribonucleotide is brought into the active site to initiate the polymerization reaction in the absence of a primer.[1] This generates a nascent RNA chain that forms a hetero-duplex with the template DNA strand. However, before entering the elongation phase, polymerase may terminate prematurely and release a short, truncated transcript. This process is called abortive initiation.[19] Many cycles of abortive initiation may occur before the transcript grows to sufficient length to promote polymerase escape from the promoter. Throughout abortive initiation cycles, RNA polymerase remains bound to the promoter and pulls downstream DNA into its catalytic cleft in a scrunching-kind of motion.[19]
Promoter escape
When a transcript attains the threshold length of ten nucleotides, it enters the RNA exit channel.[1] The polymerase breaks its interactions with the promoter elements and any regulatory proteins associated with the initiation complex that it no longer needs.[20] Promoter escape in eukaryotes requires ATP hydrolysis and, in the case of Pol II-phosphorylation of the CTD. Meanwhile, the transcription bubble collapses down to 12-14 nucleotides, providing kinetic energy required for the escape.[1]
Elongation
After escaping the promoter and shedding most of the transcription factors for initiation, the polymerase acquires new factors for the next phase of transcription: elongation.[21][22] Transcription elongation is a processive process. Double stranded DNA that enters from the front of the enzyme is unzipped to avail the template strand for RNA synthesis. For every DNA base pair separated by the advancing polymerase, one hybrid RNA:DNA base pair is immediately formed. DNA strands and nascent RNA chain exit from separate channels; the two DNA strands reunite at the trailing end of the transcription bubble while the single strand RNA emerges alone.
Elongation factors
Among the proteins recruited to polymerase are elongation factors, thus called because they stimulate transcription elongation.[23] There are different classes of elongation factors. Some factors can increase the overall rate of transcribing, some can help the polymerase through transient pausing sites, and some can assist the polymerase to transcribe through chromatin.[24] One of the elongation factors, P-TEFb, is particularly important.[25] P-TEFb phosphorylates the second residue (Ser-2) of the CTD repeats (YSPTSPS) of the bound Pol II. P-TEFb also phosphorylates and activates SPT5 and TAT-SF1. SPT5 is a universal transcription factor that helps recruit 5'-capping enzyme to Pol II with a CTD phosphorylated at Ser-5. TAF-SF1 recruits components of the RNA splicing machinery to the Ser-2 phosphorylated CTD. P-TEFb also helps suppress transient pausing of polymerase when it encounters certain sequences immediately following initiation.[25]
Transcription fidelity
Transcription fidelity is achieved through multiple mechanisms. RNA polymerases select correct nucleoside triphosphate (NTP) substrate to prevent transcription errors. Only the NTP which correctly base pairs with the coding base in the DNA is admitted to the active center.[26][27] RNA polymerase performs two known proof reading functions to detect and remove misincorporated nucleotides: pyrophosphorylytic editing and hydrolytic editing.[1] The former removes the incorrectly inserted ribonucleotide by a simple reversal of the polymerization reaction, while the latter involves backtracking of the polymerase and cleaving of a segment of error-containing RNA product. Elongation factor TFIIS (InterPro: IPR006289; TCEA1, TCEA2, TCEA3) stimulates an inherent ribonuclease activity in the polymerase, allowing the removal of misincorporated bases through limited local RNA degradation.[28] Note that all reactions (phosphodiester bond synthesis, pyrophosphorolysis, phosphodiester bond hydrolysis) are performed by RNA polymerase by using a single active center.[29]
Pausing, poising, and backtracking
Transcription elongation is not a smooth ride along double stranded DNA, as RNA polymerase undergoes extensive co-transcriptional pausing during transcription elongation.[30][31] In general, RNA polymerase II does not transcribe through a gene at a constant pace. Rather it pauses periodically at certain sequences, sometimes for long periods of time before resuming transcription.[32] This pausing is especially pronounced at nucleosomes, and arises in part through the polymerase entering a transcriptionally incompetent backtracked state.[30] The duration of these pauses ranges from seconds to minutes or longer, and exit from long-lived pauses can be promoted by elongation factors such as TFIIS.[33]
This pausing is also sometimes used for proofreading; here the polymerase backs up, erases some of the RNA it has already made and has another go at transcription.[1] In extreme cases, for example, when the polymerase encounters a damaged nucleotide, it comes to a complete halt. More often, an elongating polymerase is stalled near the promoter.[32] Promoter-proximal pausing during early elongation is a commonly used mechanism for regulating genes poised to be expressed rapidly or in a coordinated fashion. Pausing is mediated by a complex called NELF (negative elongation factor) in collaboration with DSIF (DRB-sensitivity-inducing factor containing SPT4/SPT5).[34] The blockage is released once the polymerase receives an activation signal, such as the phosphorylation of Ser-2 of CTD tail by P-TEFb. Other elongation factors such as ELL and TFIIS stimulate the rate of elongation by limiting the length of time that polymerase pauses.[1]
RNA processing
Elongating polymerase is associated with a set of protein factors required for various types of RNA processing.[1] mRNA is capped as soon as it emerges from the RNA-exit channel of the polymerase. After capping, dephosphorylation of Ser-5 within the CTD repeats may be responsible for dissociation of the capping machinery. Further phosphorylation of Ser-2 causes recruitment of the RNA splicing machinery that catalyzes the removal of non-coding introns to generate mature mRNA.[1] Alternative splicing expands the protein complements in eukaryotes. Just as with 5’-capping and splicing, the CTD tail is involved in recruiting enzymes responsible for 3’-polyadenylation, the final RNA processing event that is coupled with the termination of transcription.[1]
Termination
The last stage of transcription is termination, which leads to the dissociation of the complete transcript and the release of RNA polymerase from the template DNA.The process differs for each of the three RNA polymerases.[35] The mechanism of termination is the least understood of the three transcription stages.
Factor-dependent
The termination of transcription of pre-rRNA genes by polymerase Pol I is performed by a system that needs a specific transcription termination factor.[3] The mechanism used bears some resemblance to the rho-dependent termination in prokaryotes.[36] Eukaryotic cells contain hundreds of ribosomal DNA repeats, sometimes distributed over multiple chromosomes. Termination of transcription occurs in the ribosomal intergenic spacer region that contains several transcription termination sites upstream of a Pol I pausing site. Through a yet unknown mechanism, the 3’-end of the transcript is cleaved, generating a large primary rRNA molecule that is further processed into the mature 18S, 5.8S and 28S rRNAs.
As Pol II reaches the end of a gene, two protein complexes carried by the CTD, CPSF (cleavage and polyadenylation specificity factor) and CSTF (cleavage stimulation factor), recognize the poly-A signal in the transcribed RNA.[35] Poly-A-bound CPSF and CSTF recruit other proteins to carry out RNA cleavage and then polyadenylation. Poly-A polymerase adds approximately 200 adenines to the cleaved 3’ end of the RNA without a template.[35] The long poly-A tail is unique to transcripts made by Pol II.
In the process of terminating transcription by Pol I and Pol II, the elongation complex does not dissolve immediately after the RNA is cleaved. The polymerase continues to move along the template, generating a second RNA molecule associated with the elongation complex.[1] Two models have been proposed to explain how termination is achieved at last.[35] The allosteric model states that when transcription proceeds through the termination sequence, it causes disassembly of elongation factors and/or an assembly of termination factors that cause conformational changes of the elongation complex.[36][37] The torpedo model suggests that a 5' to 3' exonuclease degrades the second RNA as it emerges from the elongation complex. Polymerase is released as the highly processive exonuclease overtakes it. It is proposed that an emerging view will express a merge of these two models.[37]
Factor-independent
RNA polymerase III can terminate transcription efficiently without the involvement of additional factors. The Pol III termination signal consists of a stretch of thymines (on the nontemplate strand) located within 40bp downstream from the 3' end of mature RNAs.[35] The poly-T termination signal pauses Pol III
Eukaryotic transcriptional control
The regulation of gene expression in eukaryotes is achieved through the interaction of several levels of control that acts both locally to turn on or off individual genes in response to a specific cellular need and globally to maintain a chromatin-wide gene expression pattern that shapes cell identity.[1][38] Because eukaryotic genome is wrapped around histones to form nucleosomes and higher-order chromatin structures, the substrates for transcriptional machinery are in general partially concealed.[1] Without regulatory proteins, many genes are expressed at low level or not expressed at all. Transcription requires displacement of the positioned nucleosomes to enable the transcriptional machinery to gain access of the DNA.[39]
All steps in the transcription are subject to some degree of regulation.[1] Transcription initiation in particular is the primary level at which gene expression is regulated. Targeting the rate-limiting initial step is the most efficient in terms of energy costs for the cell. Transcription initiation is regulated by cis-acting elements (enhancers, silencers, isolators) within the regulatory regions of the DNA, and sequence-specific trans-acting factors that act as activators or repressors.[1] Gene transcription can also be regulated post-initiation by targeting the movement of the elongating polymerase.[40]
Global control and epigenetic regulation
The eukaryotic genome is organized into a compact chromatin structure that allows only regulated access to DNA. The chromatin structure can be globally "open" and more transcriptionally permissive, or globally "condensed" and transcriptionally inactive. The former (euchromatin) is lightly packed and rich in genes under active transcription. The latter (heterochromatin) includes gene-poor regions such as telomeres and centromeres but also regions with normal gene density but transcriptionally silenced. Transcription can be silenced by histone modification (deacetylation and methylation), RNA interference, and/or DNA methylation.[41]
The gene expression patterns that define cell identity are inherited through cell division.[1] This process is called epigenetic regulation. DNA methylation is reliably inherited through the action of maintenance methylases that modify the nascent DNA strand generated by replication.[1] In mammalian cells, DNA methylation is the primary marker of transcriptionally silenced regions. Specialized proteins can recognize the marker and recruit histone deacetylases and methylases to re-establish the silencing. Nucleosome histone modifications could also be inherited during cell division, however, it is not clear whether it can work independently without the direction by DNA methylation.[1]
Gene-specific activation
The two main tasks of transcription initiation are to provide RNA polymerase with an access to the promoter and to assemble general transcription factors with polymerase into a transcription initiation complex. Diverse mechanisms of initiating transcription by overriding inhibitory signals at the gene promoter have been identified.[1] Eukaryotic genes have acquired extensive regulatory sequences that encompass a large number of regulator-binding sites and spread overall kilobases (sometimes hundreds of kilobases) from the promoter–-both upstream and downstream.[1] The regulator binding sites are often clustered together into units called enhancers. Enhancers can facilitate highly cooperative action of several transcription factors (which constitute enhanceosomes). Remote enhancers allow transcription regulation at a distance. Insulators situated between enhancers and promoters help define the genes that an enhancer can or cannot influence.
Eukaryotic transcriptional activators have separate DNA-binding and activating functions.[1] Upon binding to its cis-element, an activator can recruit polymerase directly or recruit other factors needed by the transcriptional machinery. An activator can also recruit nucleosome modifiers that alter chromatin in the vicinity of the promoter and thereby help initiation. Multiple activators can work together, either by recruiting a common or two mutually dependent components of the transcriptional machinery, or by helping each other bind to their DNA sites.[1] These interactions can synergize multiple signaling inputs and produce intricate transcriptional responses to address cellular needs.
Gene-specific repression
Eukaryotic transcription repressors share some of the mechanisms used by their prokaryotic counterparts. For example, by binding to a site on DNA that overlaps with the binding site of an activator, a repressor can inhibit binding of the activator. But more frequently, eukaryotic repressors inhibit the function of an activator by masking its activating domain, preventing its nuclear localization, promoting its degradation, or inactivating it through chemical modifications.[1] Repressors can directly inhibit transcription initiation by binding to a site upstream of a promoter and interacting with the transcriptional machinery. Repressors can indirectly repress transcription by recruiting histone modifiers (deacetylases and methylases) or nucleosome remodeling enzymes that affect the accessibility of the DNA.[1] Repressing histone and DNA modifications are also the basis of transcriptional silencing that can spread along the chromatin and switch off multiple genes.[42]
Elongation and termination control
The elongation phase starts once assembly of the elongation complex has been completed, and progresses until a termination sequence is encountered.[1] The post-initiation movement of RNA polymerase is the target of another class of important regulatory mechanisms. For example, the transcriptional activator Tat affects elongation rather than initiation during its regulation of HIV transcription.[43] In fact, many eukaryotic genes are regulated by releasing a block to transcription elongation called promoter-proximal pausing.[44] Pausing can influence chromatin structure at promoters to facilitate gene activity and lead to rapid or synchronous transcriptional responses when cells are exposed to an activation signal.[32] Pausing is associated with the binding of two negative elongation factors, DSIF (SPT4/SPT5) and NELF, to the elongation complex. Other factors can also influence the stability and duration of the paused polymerase.[45] Pause release is triggered by the recruitment of the P-TEFb kinase.[40]
Transcription termination has also emerged as an important area of transcriptional regulation. Termination is coupled with the efficient recycling of polymerase.[46] The factors associated with transcription termination can also mediate gene looping and thereby determine the efficiency of re-initiation.
Transcription-coupled DNA repair
When transcription is arrested by the presence of a lesion in the transcribed strand of a gene, DNA repair proteins are recruited to the stalled RNA polymerase to initiate a process called transcription-coupled repair.[47] Central to this process is the general transcription factor TFIIH that has ATPase activity. TFIIH causes a conformational change in the polymerase, to expose the transcription bubble trapped inside, in order for the DNA repair enzymes to gain access to the lesion.[48] Thus, RNA polymerase serves as damage-sensing protein in the cell to target repair enzymes to genes that are being actively transcribed.
Comparisons between prokaryotic and eukaryotic transcription
Eukaryotic transcription is more complex than prokaryotic transcription. For instance, in eukaryotes the genetic material (DNA), and therefore transcription, is primarily localized to the nucleus, where it is separated from the cytoplasm (in which translation occurs) by the nuclear membrane. This allows for the temporal regulation of gene expression through the sequestration of the RNA in the nucleus, and allows for selective transport of mature RNAs to the cytoplasm. Bacteria do not have a distinct nucleus that separates DNA from ribosome and mRNA is translated into protein as soon as it is transcribed. The coupling between the two processes provides an important mechanism for prokaryotic gene regulation.[1]
At the level of initiation, RNA polymerase in prokaryotes (bacteria in particular) binds strongly to the promoter region and initiates a high basal rate of transcription. No ATP hydrolysis is needed for the close-to-open transition, promoter melting is driven by binding reactions that favor the melted conformation. Chromatin greatly impedes transcription in eukaryotes. Assembly of large multi-protein preinitiation complex is required for promoter-specific initiation. Promoter melting in eukaryotes requires hydrolysis of ATP. As a result, eukaryotic RNA polymerases exhibit a low basal rate of transcription initiation.[42]
Regulation of transcription in cancer
In vertebrates, the majority of gene promoters contain a CpG island with numerous CpG sites.[49] When many of a gene's promoter CpG sites are methylated the gene becomes silenced.[50] Colorectal cancers typically have 3 to 6 driver mutations and 33 to 66 hitchhiker or passenger mutations.[51] However, transcriptional silencing may be of more importance than mutation in causing progression to cancer. For example, in colorectal cancers about 600 to 800 genes are transcriptionally silenced by CpG island methylation (see regulation of transcription in cancer). Transcriptional repression in cancer can also occur by other epigenetic mechanisms, such as altered expression of microRNAs.[52] In breast cancer, transcriptional repression of BRCA1 may occur more frequently by over-expressed microRNA-182 than by hypermethylation of the BRCA1 promoter (see Low expression of BRCA1 in breast and ovarian cancers).
See also
References
- Watson J, Baker TA, Bell SP, Gann A, Levine M, Losik R, Harrison SC (2014). Molecular Biology of the Gene (7th ed.). Benjamin-Cummings Publishing Company. ISBN 978-0-321-76243-6.
- Mattick JS (November 2001). "Non-coding RNAs: the architects of eukaryotic complexity". EMBO Reports. 2 (11): 986–91. doi:10.1093/embo-reports/kve230. PMC 1084129. PMID 11713189.
- Lodish H, Berk A, Kaiser CA, Krieger M, Bretscher A, Ploegh H, Amon A, Scott MP (2012-05-02). Molecular cell biology (7th ed.). New York: W.H. Freeman and Co. ISBN 9781429234139.
- Cramer P, Armache KJ, Baumli S, Benkert S, Brueckner F, Buchen C, Damsma GE, Dengl S, Geiger SR, Jasiak AJ, Jawhari A, Jennebach S, Kamenski T, Kettenberger H, Kuhn CD, Lehmann E, Leike K, Sydow JF, Vannini A (2008). "Structure of eukaryotic RNA polymerases". Annual Review of Biophysics. 37: 337–52. doi:10.1146/annurev.biophys.37.032807.130008. hdl:11858/00-001M-0000-0015-7BF0-E. PMID 18573085. S2CID 8818814.
- Sirri V, Urcuqui-Inchima S, Roussel P, Hernandez-Verdun D (January 2008). "Nucleolus: the fascinating nuclear body". Histochemistry and Cell Biology. 129 (1): 13–31. doi:10.1007/s00418-007-0359-6. PMC 2137947. PMID 18046571.
- Fromont-Racine M, Senger B, Saveanu C, Fasiolo F (August 2003). "Ribosome assembly in eukaryotes". Gene. 313: 17–42. doi:10.1016/S0378-1119(03)00629-2. PMID 12957375.
- Dieci G, Fiorino G, Castelnuovo M, Teichmann M, Pagano A (December 2007). "The expanding RNA polymerase III transcriptome". Trends in Genetics. 23 (12): 614–22. doi:10.1016/j.tig.2007.09.001. PMID 17977614.
- Carter R, Drouin G (May 2010). "The increase in the number of subunits in eukaryotic RNA polymerase III relative to RNA polymerase II is due to the permanent recruitment of general transcription factors". Molecular Biology and Evolution. 27 (5): 1035–43. doi:10.1093/molbev/msp316. PMID 20026480.
- Fernández-Tornero C, Moreno-Morcillo M, Rashid UJ, Taylor NM, Ruiz FM, Gruene T, Legrand P, Steuerwald U, Müller CW (October 2013). "Crystal structure of the 14-subunit RNA polymerase I". Nature. 502 (7473): 644–9. Bibcode:2013Natur.502..644F. doi:10.1038/nature12636. PMID 24153184. S2CID 205235881.
- Cramer P, Bushnell DA, Kornberg RD (June 2001). "Structural basis of transcription: RNA polymerase II at 2.8 angstrom resolution" (PDF). Science. 292 (5523): 1863–76. Bibcode:2001Sci...292.1863C. doi:10.1126/science.1059493. hdl:11858/00-001M-0000-0015-8729-F. PMID 11313498. S2CID 4993438.
- Corden JL (November 2013). "RNA polymerase II C-terminal domain: Tethering transcription to transcript and template". Chemical Reviews. 113 (11): 8423–55. doi:10.1021/cr400158h. PMC 3988834. PMID 24040939.
- Butler JE, Kadonaga JT (October 2002). "The RNA polymerase II core promoter: a key component in the regulation of gene expression". Genes & Development. 16 (20): 2583–92. doi:10.1101/gad.1026202. PMID 12381658.
- Lenhard B, Sandelin A, Carninci P (March 2012). "Metazoan promoters: emerging characteristics and insights into transcriptional regulation". Nature Reviews. Genetics. 13 (4): 233–45. doi:10.1038/nrg3163. PMID 22392219. S2CID 39948639.
- Orphanides G, Lagrange T, Reinberg D (November 1996). "The general transcription factors of RNA polymerase II". Genes & Development. 10 (21): 2657–83. doi:10.1101/gad.10.21.2657. PMID 8946909.
- Roeder RG (September 1996). "The role of general initiation factors in transcription by RNA polymerase II". Trends in Biochemical Sciences. 21 (9): 327–35. doi:10.1016/S0968-0004(96)10050-5. PMID 8870495.
- He Y, Fang J, Taatjes DJ, Nogales E (March 2013). "Structural visualization of key steps in human transcription initiation". Nature. 495 (7442): 481–6. Bibcode:2013Natur.495..481H. doi:10.1038/nature11991. PMC 3612373. PMID 23446344.
- Holstege FC, Fiedler U, Timmers HT (December 1997). "Three transitions in the RNA polymerase II transcription complex during initiation". The EMBO Journal. 16 (24): 7468–80. doi:10.1093/emboj/16.24.7468. PMC 1170346. PMID 9405375.
- Svejstrup JQ, Vichi P, Egly JM (September 1996). "The multiple roles of transcription/repair factor TFIIH". Trends in Biochemical Sciences. 21 (9): 346–50. doi:10.1016/S0968-0004(96)10046-3. PMID 8870499.
- Revyakin A, Liu C, Ebright RH, Strick TR (November 2006). "Abortive initiation and productive initiation by RNA polymerase involve DNA scrunching". Science. 314 (5802): 1139–43. Bibcode:2006Sci...314.1139R. doi:10.1126/science.1131398. PMC 2754787. PMID 17110577.
- Dvir A (September 2002). "Promoter escape by RNA polymerase II". Biochimica et Biophysica Acta (BBA) - Gene Structure and Expression. 1577 (2): 208–223. doi:10.1016/S0167-4781(02)00453-0. PMID 12213653.
- Pokholok DK, Hannett NM, Young RA (April 2002). "Exchange of RNA polymerase II initiation and elongation factors during gene expression in vivo". Molecular Cell. 9 (4): 799–809. doi:10.1016/S1097-2765(02)00502-6. PMID 11983171.
- Wade JT, Struhl K (April 2008). "The transition from transcriptional initiation to elongation". Current Opinion in Genetics & Development. 18 (2): 130–6. doi:10.1016/j.gde.2007.12.008. PMC 2563432. PMID 18282700.
- Saunders A, Core LJ, Lis JT (August 2006). "Breaking barriers to transcription elongation". Nature Reviews. Molecular Cell Biology. 7 (8): 557–67. doi:10.1038/nrm1981. PMID 16936696. S2CID 29242830.
- Arndt KM, Kane CM (October 2003). "Running with RNA polymerase: eukaryotic transcript elongation". Trends in Genetics. 19 (10): 543–50. doi:10.1016/j.tig.2003.08.008. PMID 14550628.
- Brès V, Yoh SM, Jones KA (June 2008). "The multi-tasking P-TEFb complex". Current Opinion in Cell Biology. 20 (3): 334–40. doi:10.1016/j.ceb.2008.04.008. PMC 2628440. PMID 18513937.
- Westover KD, Bushnell DA, Kornberg RD (November 2004). "Structural basis of transcription: nucleotide selection by rotation in the RNA polymerase II active center". Cell. 119 (4): 481–9. doi:10.1016/j.cell.2004.10.016. PMID 15537538. S2CID 11068662.
- Wang D, Bushnell DA, Westover KD, Kaplan CD, Kornberg RD (December 2006). "Structural basis of transcription: role of the trigger loop in substrate specificity and catalysis". Cell. 127 (5): 941–54. doi:10.1016/j.cell.2006.11.023. PMC 1876690. PMID 17129781.
- Wang D, Bushnell DA, Huang X, Westover KD, Levitt M, Kornberg RD (May 2009). "Structural basis of transcription: backtracked RNA polymerase II at 3.4 angstrom resolution". Science. 324 (5931): 1203–6. Bibcode:2009Sci...324.1203W. doi:10.1126/science.1168729. PMC 2718261. PMID 19478184.
- Sosunov V, Sosunova E, Mustaev A, Bass I, Nikiforov V, Goldfarb A (May 2003). "Unified two-metal mechanism of RNA synthesis and degradation by RNA polymerase". The EMBO Journal. 22 (9): 2234–44. doi:10.1093/emboj/cdg193. PMC 156065. PMID 12727889.
- Hodges, Courtney; Bintu, Lacramioara; Lubkowska, Lucyna; Kashlev, Mikhail; Bustamante, Carlos (2009-07-31). "Nucleosomal fluctuations govern the transcription dynamics of RNA polymerase II". Science. 325 (5940): 626–628. Bibcode:2009Sci...325..626H. doi:10.1126/science.1172926. ISSN 1095-9203. PMC 2775800. PMID 19644123.
- Churchman, L. Stirling; Weissman, Jonathan S. (2011-01-20). "Nascent transcript sequencing visualizes transcription at nucleotide resolution". Nature. 469 (7330): 368–373. Bibcode:2011Natur.469..368C. doi:10.1038/nature09652. ISSN 1476-4687. PMC 3880149. PMID 21248844.
- Adelman K, Lis JT (October 2012). "Promoter-proximal pausing of RNA polymerase II: emerging roles in metazoans". Nature Reviews. Genetics. 13 (10): 720–31. doi:10.1038/nrg3293. PMC 3552498. PMID 22986266.
- Galburt, Eric A.; Grill, Stephan W.; Wiedmann, Anna; Lubkowska, Lucyna; Choy, Jason; Nogales, Eva; Kashlev, Mikhail; Bustamante, Carlos (2007-04-12). "Backtracking determines the force sensitivity of RNAP II in a factor-dependent manner". Nature. 446 (7137): 820–823. Bibcode:2007Natur.446..820G. doi:10.1038/nature05701. ISSN 1476-4687. PMID 17361130. S2CID 4310108.
- Missra A, Gilmour DS (June 2010). "Interactions between DSIF (DRB sensitivity inducing factor), NELF (negative elongation factor), and the Drosophila RNA polymerase II transcription elongation complex". Proceedings of the National Academy of Sciences of the United States of America. 107 (25): 11301–6. Bibcode:2010PNAS..10711301M. doi:10.1073/pnas.1000681107. PMC 2895096. PMID 20534440.
- Richard P, Manley JL (June 2009). "Transcription termination by nuclear RNA polymerases". Genes & Development. 23 (11): 1247–69. doi:10.1101/gad.1792809. PMC 2763537. PMID 19487567.
- Clancey S (2008). "DNA transcription". Nature Education. 1 (41). Retrieved 27 November 2013.
- Rosonina E, Kaneko S, Manley JL (May 2006). "Terminating the transcript: breaking up is hard to do". Genes & Development. 20 (9): 1050–6. doi:10.1101/gad.1431606. PMID 16651651.
- Shandilya J, Roberts SG (May 2012). "The transcription cycle in eukaryotes: from productive initiation to RNA polymerase II recycling". Biochimica et Biophysica Acta (BBA) - Gene Regulatory Mechanisms. 1819 (5): 391–400. doi:10.1016/j.bbagrm.2012.01.010. PMID 22306664.
- Kulaeva OI, Gaykalova DA, Studitsky VM (May 2007). "Transcription through chromatin by RNA polymerase II: histone displacement and exchange". Mutation Research. 618 (1–2): 116–29. doi:10.1016/j.mrfmmm.2006.05.040. PMC 1924643. PMID 17313961.
- Peterlin BM, Price DH (August 2006). "Controlling the elongation phase of transcription with P-TEFb". Molecular Cell. 23 (3): 297–305. doi:10.1016/j.molcel.2006.06.014. PMID 16885020.
- Bannister AJ, Kouzarides T (March 2011). "Regulation of chromatin by histone modifications". Cell Research. 21 (3): 381–95. doi:10.1038/cr.2011.22. PMC 3193420. PMID 21321607.
- Brown TA (2002). Genomes. New York: Wiley-Liss. ISBN 978-0-471-25046-3.
- Kao SY, Calman AF, Luciw PA, Peterlin BM (3 December 1987). "Anti-termination of transcription within the long terminal repeat of HIV-1 by tat gene product". Nature. 330 (6147): 489–93. Bibcode:1987Natur.330..489K. doi:10.1038/330489a0. PMID 2825027. S2CID 4311235.
- Lis J (1998). "Promoter-associated pausing in promoter architecture and postinitiation transcriptional regulation". Cold Spring Harbor Symposia on Quantitative Biology. 63: 347–56. doi:10.1101/sqb.1998.63.347. PMID 10384299.
- Cheng B, Li T, Rahl PB, Adamson TE, Loudas NB, Guo J, Varzavand K, Cooper JJ, Hu X, Gnatt A, Young RA, Price DH (January 2012). "Functional association of Gdown1 with RNA polymerase II poised on human genes". Molecular Cell. 45 (1): 38–50. doi:10.1016/j.molcel.2011.10.022. PMC 3259526. PMID 22244331.
- Feige MJ, Hendershot LM (April 2011). "Disulfide bonds in ER protein folding and homeostasis". Current Opinion in Cell Biology. 23 (2): 167–75. doi:10.1016/j.ceb.2010.10.012. PMC 3078216. PMID 21144725.
- Mellon I, Spivak G, Hanawalt PC (October 1987). "Selective removal of transcription-blocking DNA damage from the transcribed strand of the mammalian DHFR gene". Cell. 51 (2): 241–9. doi:10.1016/0092-8674(87)90151-6. PMID 3664636. S2CID 2879958.
- Sarker AH, Tsutakawa SE, Kostek S, Ng C, Shin DS, Peris M, Campeau E, Tainer JA, Nogales E, Cooper PK (October 2005). "Recognition of RNA polymerase II and transcription bubbles by XPG, CSB, and TFIIH: insights for transcription-coupled repair and Cockayne Syndrome". Molecular Cell. 20 (2): 187–98. doi:10.1016/j.molcel.2005.09.022. PMID 16246722.
- Saxonov S, Berg P, Brutlag DL (January 2006). "A genome-wide analysis of CpG dinucleotides in the human genome distinguishes two distinct classes of promoters". Proceedings of the National Academy of Sciences of the United States of America. 103 (5): 1412–7. Bibcode:2006PNAS..103.1412S. doi:10.1073/pnas.0510310103. PMC 1345710. PMID 16432200.
- Bird A (January 2002). "DNA methylation patterns and epigenetic memory". Genes & Development. 16 (1): 6–21. doi:10.1101/gad.947102. PMID 11782440.
- Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA, Kinzler KW (March 2013). "Cancer genome landscapes". Science. 339 (6127): 1546–58. Bibcode:2013Sci...339.1546V. doi:10.1126/science.1235122. PMC 3749880. PMID 23539594.
- Tessitore A, Cicciarelli G, Del Vecchio F, Gaggiano A, Verzella D, Fischietti M, Vecchiotti D, Capece D, Zazzeroni F, Alesse E (2014). "MicroRNAs in the DNA Damage/Repair Network and Cancer". International Journal of Genomics. 2014: 820248. doi:10.1155/2014/820248. PMC 3926391. PMID 24616890.