Gelsolin
Gelsolin is an actin-binding protein that is a key regulator of actin filament assembly and disassembly. Gelsolin is one of the most potent members of the actin-severing gelsolin/villin superfamily, as it severs with nearly 100% efficiency.[4][5]
Cellular gelsolin, found within the cytosol and mitochondria,[6] has a closely related secreted form, Plasma gelsolin, that contains an additional 24 AA N-terminal extension.[7][8] Plasma gelsolin's ability to sever actin filaments helps the body recover from disease and injury that leaks cellular actin into the blood. Additionally it plays important roles in host innate immunity, activating macrophages and localizing of inflammation.
Structure
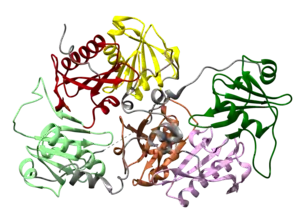
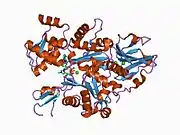
Gelsolin is an 82-kD protein with six homologous subdomains, referred to as S1-S6. Each subdomain is composed of a five-stranded β-sheet, flanked by two α-helices, one positioned perpendicular with respect to the strands and one positioned parallel. The β-sheets of the three N-terminal subdomains (S1-S3) join to form an extended β-sheet, as do the β-sheets of the C-terminal subdomains (S4-S6).[9]
Regulation
Among the lipid-binding actin regulatory proteins, gelsolin (like cofilin) preferentially binds polyphosphoinositide (PPI).[10] The binding sequences in gelsolin closely resemble the motifs in the other PPI-binding proteins.[10]
Gelsolin's activity is stimulated by calcium ions (Ca2+).[5] Although the protein retains its overall structural integrity in both activated and deactivated states, the S6 helical tail moves like a latch depending on the concentration of calcium ions.[11] The C-terminal end detects the calcium concentration within the cell. When there is no Ca2+ present, the tail of S6 shields the actin-binding sites on one of S2's helices.[9] When a calcium ion attaches to the S6 tail, however, it straightens, exposing the S2 actin-binding sites.[11] The N-terminal is directly involved in the severing of actin. S2 and S3 bind to the actin before the binding of S1 severs actin-actin bonds and caps the barbed end.[10]
Gelsolin can be inhibited by a local rise in the concentration of phosphatidylinositol (4,5)-bisphosphate (PIP2), a PPI. This is a two step process. Firstly, (PIP2) binds to S2 and S3, inhibiting gelsolin from actin side binding. Then, (PIP2) binds to gelsolin’s S1, preventing gelsolin from severing actin, although (PIP2) does not bind directly to gelsolin's actin-binding site.[10]
Gelsolin's severing of actin, in contrast to the severing of microtubules by katanin, does not require any extra energy input.
Cellular function
As an important actin regulator, gelsolin plays a role in podosome formation (along with Arp3, cortactin, and Rho GTPases).[12]
Gelsolin also inhibits apoptosis by stabilizing the mitochondria.[6] Prior to cell death, mitochondria normally lose membrane potential and become more permeable. Gelsolin can impede the release of cytochrome C, obstructing the signal amplification that would have led to apoptosis.[13]
Actin can be cross-linked into a gel by actin cross-linking proteins. Gelsolin can turn this gel into a sol, hence the name gelsolin.
Animal studies
Research in mice suggests that gelsolin, like other actin-severing proteins, is not expressed to a significant degree until after the early embryonic stage—approximately 2 weeks in murine embryos.[14] In adult specimens, however, gelsolin is particularly important in motile cells, such as blood platelets. Mice with null gelsolin-coding genes undergo normal embryonic development, but the deformation of their blood platelets reduced their motility, resulting in a slower response to wound healing.[14]
An insufficiency of gelsolin in mice has also been shown to cause increased permeability of the vascular pulmonary barrier, suggesting that gelsolin is important in the response to lung injury.[15]
Related proteins
| Gelsolin-like domain | |
|---|---|
 | |
| Identifiers | |
| Symbol | ? |
Sequence comparisons indicate an evolutionary relationship between gelsolin, villin, fragmin, and severin.[17] Six large repeating segments occur in gelsolin and villin, and 3 similar segments in severin and fragmin. The multiple repeats are related in structure (but barely in sequence) to the ADF-H domain, forming a superfamily (InterPro: IPR029006). The family appears to have evolved from an ancestral sequence of 120 to 130 amino acid residues.[17][4]
Asgard archaea encode many functional gelsolins.[18]
Interactions
Gelsolin is a cytoplasmic, calcium-regulated, actin-modulating protein that binds to the barbed ends of actin filaments, preventing monomer exchange (end-blocking or capping).[19] It can promote nucleation (the assembly of monomers into filaments), as well as sever existing filaments. In addition, this protein binds with high affinity to fibronectin. Plasma gelsolin and cytoplasmic gelsolin are derived from a single gene by alternate initiation sites and differential splicing.[7]
Gelsolin has been shown to interact with:
References
- GRCm38: Ensembl release 89: ENSMUSG00000026879 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Ghoshdastider U, Popp D, Burtnick LD, Robinson RC (November 2013). "The expanding superfamily of gelsolin homology domain proteins". Cytoskeleton. 70 (11): 775–95. doi:10.1002/cm.21149. PMID 24155256. S2CID 205643538.
- Sun HQ, Yamamoto M, Mejillano M, Yin HL (November 1999). "Gelsolin, a multifunctional actin regulatory protein". The Journal of Biological Chemistry. 274 (47): 33179–82. doi:10.1074/jbc.274.47.33179. PMID 10559185.
- Koya RC, Fujita H, Shimizu S, Ohtsu M, Takimoto M, Tsujimoto Y, Kuzumaki N (May 2000). "Gelsolin inhibits apoptosis by blocking mitochondrial membrane potential loss and cytochrome c release". The Journal of Biological Chemistry. 275 (20): 15343–9. doi:10.1074/jbc.275.20.15343. PMID 10809769.
- Kwiatkowski DJ, Stossel TP, Orkin SH, Mole JE, Colten HR, Yin HL (1986-10-02). "Plasma and cytoplasmic gelsolins are encoded by a single gene and contain a duplicated actin-binding domain". Nature. 323 (6087): 455–8. Bibcode:1986Natur.323..455K. doi:10.1038/323455a0. PMID 3020431. S2CID 4356162.
- Nag S, Larsson M, Robinson RC, Burtnick LD (July 2013). "Gelsolin: the tail of a molecular gymnast". Cytoskeleton. 70 (7): 360–84. doi:10.1002/cm.21117. PMID 23749648. S2CID 23646422.
- Kiselar JG, Janmey PA, Almo SC, Chance MR (April 2003). "Visualizing the Ca2+-dependent activation of gelsolin by using synchrotron footprinting". Proceedings of the National Academy of Sciences of the United States of America. 100 (7): 3942–7. Bibcode:2003PNAS..100.3942K. doi:10.1073/pnas.0736004100. PMC 153027. PMID 12655044.
- Yu FX, Sun HQ, Janmey PA, Yin HL (July 1992). "Identification of a polyphosphoinositide-binding sequence in an actin monomer-binding domain of gelsolin". The Journal of Biological Chemistry. 267 (21): 14616–21. doi:10.1016/S0021-9258(18)42086-8. PMID 1321812.
- Burtnick LD, Urosev D, Irobi E, Narayan K, Robinson RC (July 2004). "Structure of the N-terminal half of gelsolin bound to actin: roles in severing, apoptosis and FAF". The EMBO Journal. 23 (14): 2713–22. doi:10.1038/sj.emboj.7600280. PMC 514944. PMID 15215896.
- Varon C, Tatin F, Moreau V, Van Obberghen-Schilling E, Fernandez-Sauze S, Reuzeau E, et al. (May 2006). "Transforming growth factor beta induces rosettes of podosomes in primary aortic endothelial cells". Molecular and Cellular Biology. 26 (9): 3582–94. doi:10.1128/MCB.26.9.3582-3594.2006. PMC 1447430. PMID 16611998.
- Kusano H, Shimizu S, Koya RC, Fujita H, Kamada S, Kuzumaki N, Tsujimoto Y (October 2000). "Human gelsolin prevents apoptosis by inhibiting apoptotic mitochondrial changes via closing VDAC". Oncogene. 19 (42): 4807–14. doi:10.1038/sj.onc.1203868. PMID 11039896.
- Witke W, Sharpe AH, Hartwig JH, Azuma T, Stossel TP, Kwiatkowski DJ (April 1995). "Hemostatic, inflammatory, and fibroblast responses are blunted in mice lacking gelsolin". Cell. 81 (1): 41–51. doi:10.1016/0092-8674(95)90369-0. PMID 7720072.
- Becker PM, Kazi AA, Wadgaonkar R, Pearse DB, Kwiatkowski D, Garcia JG (April 2003). "Pulmonary vascular permeability and ischemic injury in gelsolin-deficient mice". American Journal of Respiratory Cell and Molecular Biology. 28 (4): 478–84. doi:10.1165/rcmb.2002-0024OC. PMID 12654637.
- Wang H, Chumnarnsilpa S, Loonchanta A, Li Q, Kuan YM, Robine S, et al. (August 2009). "Helix straightening as an activation mechanism in the gelsolin superfamily of actin regulatory proteins". The Journal of Biological Chemistry. 284 (32): 21265–9. doi:10.1074/jbc.M109.019760. PMC 2755850. PMID 19491107.
- Way M, Weeds A (October 1988). "Nucleotide sequence of pig plasma gelsolin. Comparison of protein sequence with human gelsolin and other actin-severing proteins shows strong homologies and evidence for large internal repeats". Journal of Molecular Biology. 203 (4): 1127–33. doi:10.1016/0022-2836(88)90132-5. PMID 2850369.
- Akıl C, Tran LT, Orhant-Prioux M, Baskaran Y, Manser E, Blanchoin L, Robinson RC (August 2020). "Insights into the evolution of regulated actin dynamics via characterization of primitive gelsolin/cofilin proteins from Asgard archaea". Proceedings of the National Academy of Sciences of the United States of America. 117 (33): 19904–19913. Bibcode:2020PNAS..11719904A. doi:10.1073/pnas.2009167117. PMC 7444086. PMID 32747565.
- Weeds AG, Gooch J, Pope B, Harris HE (November 1986). "Preparation and characterization of pig plasma and platelet gelsolins". European Journal of Biochemistry. 161 (1): 69–76. doi:10.1111/j.1432-1033.1986.tb10125.x. PMID 3023087.
- Chauhan VP, Ray I, Chauhan A, Wisniewski HM (May 1999). "Binding of gelsolin, a secretory protein, to amyloid beta-protein". Biochemical and Biophysical Research Communications. 258 (2): 241–6. doi:10.1006/bbrc.1999.0623. PMID 10329371.
- Nishimura K, Ting HJ, Harada Y, Tokizane T, Nonomura N, Kang HY, et al. (August 2003). "Modulation of androgen receptor transactivation by gelsolin: a newly identified androgen receptor coregulator". Cancer Research. 63 (16): 4888–94. PMID 12941811.
- Wang Q, Xie Y, Du QS, Wu XJ, Feng X, Mei L, et al. (February 2003). "Regulation of the formation of osteoclastic actin rings by proline-rich tyrosine kinase 2 interacting with gelsolin". The Journal of Cell Biology. 160 (4): 565–75. doi:10.1083/jcb.200207036. PMC 2173747. PMID 12578912.
External links
- Gelsolin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)