Carboprost
Carboprost (INN, trade names for the tromethamine salts Hemabate, Tham) is a synthetic prostaglandin analogue of PGF2α (specifically, it is 15-methyl-PGF2α) with oxytocic properties.
 | |
| Clinical data | |
|---|---|
| Trade names | Hemabate |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a600042 |
| Pregnancy category |
|
| Routes of administration | Intramuscular |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C21H36O5 |
| Molar mass | 368.514 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Carboprost's main use is in the obstetrical emergency of postpartum hemorrhage which reduces postpartum bleeding during these circumstances.
Indication
Used in postpartum hemorrhage caused by uterine atony not controlled by other methods. One study has shown that carboprost tromethamine is more effective than oxytocin in preventing postpartum hemorrhage in high-risk patients undergoing cesarean delivery.[2] Carboprost is also used for the termination of pregnancy in the 2nd trimester.[3]
Contraindication
Contraindicated in severe cardiovascular, renal, and hepatic disease. It is also contraindicated in acute pelvic inflammatory disease. Hypersensitivity to carboprost or any of its components is also a contraindication[3]
Precautions
- asthma
- anemia
- jaundice
- diabetes mellitus
- seizure disorders
- past uterine surgery
Adverse Effects
- diarrhea (most common, may be sudden in onset)
- flushing or hot flashes
- fever
- chills
- nausea/vomiting
Storage and Availability
Carboprost is supplied with its salt derivative tromethamine in 1 milliliter ampules containing a 250 microgram/milliliter solution of the active drug. The drug must be refrigerated at a temperature between 2 – 8 degrees Celsius.[3]
Synthesis
A significant deactivating metabolic transformation of natural prostaglandins is enzymatic oxidation of the C-15 hydroxyl to the corresponding ketone. This is prevented, with retention of activity, by methylation to give the C-15 tertiary carbinol series.
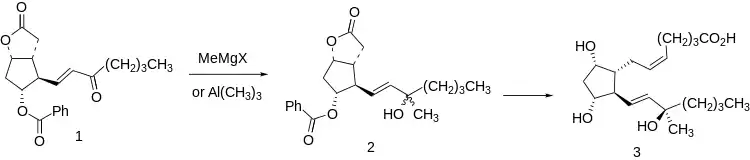
This molecular feature is readily introduced at the stage of the Corey lactone (1) by reaction with methyl Grignard reagent or trimethylaluminium. The resulting mixture of tertiary carbinols (2) is transformed to oxytocic carboprost (3) by standard transformations, including separation of diastereomers, so that the final product is the C-15 analogue. This diastereomer is reputably freeer of prostaglandin side effects than the C-15 (S) isomer.
References
- "Carboprost-REACH (Reach Pharmaceuticals Pty Ltd)". Therapeutic Goods Administration (TGA). 28 July 2023. Retrieved 10 September 2023.
- Bai J, Sun Q, Zhai H (January 2014). "A comparison of oxytocin and carboprost tromethamine in the prevention of postpartum hemorrhage in high-risk patients undergoing cesarean delivery". Experimental and Therapeutic Medicine. 7 (1): 46–50. doi:10.3892/etm.2013.1379. PMC 3861477. PMID 24348762.
- Hemabate [Package Insert]. New York, NY: Pharmacia and Upjohn Company; 2014.
- Yankee EW, Axen U, Bundy GL (September 1974). "Total synthesis of 15-methylprostaglandins". Journal of the American Chemical Society. 96 (18): 5865–76. doi:10.1021/ja00825a027. PMID 4416671.
- Bundy G, Lincoln F, Nelson N, Pike J, Schneider W (April 1971). "Novel prostaglandin syntheses". Annals of the New York Academy of Sciences. 180 (1): 76–90. Bibcode:1971NYASA.180...76B. doi:10.1111/j.1749-6632.1971.tb53186.x. PMID 5286110. S2CID 34735617.
- G. L. Bundy et al., DE 2121980, Gordon, Leonard; Pike, John Edward & Schneider, William Paul, "Verfahren zur Herstellung nueur Prostansäurederivate [Process for the production of new prostanoic acid derivatives]", published 1971-11-25, assigned to The Upjohn Co.
Further reading
- Indman PD (February 2004). "Use of carboprost to facilitate hysteroscopic resection of submucous myomas". The Journal of the American Association of Gynecologic Laparoscopists. 11 (1): 68–72. doi:10.1016/S1074-3804(05)60014-X. PMID 15104835.
- Vukelić J (2001). "Second trimester pregnancy termination in primigravidas by double application of dinoprostone gel and intramuscular administration of carboprost tromethamine". Medicinski Pregled. 54 (1–2): 11–6. PMID 11436877.
- Ippoliti C, Przepiorka D, Mehra R, Neumann J, Wood J, Claxton D, et al. (December 1995). "Intravesicular carboprost for the treatment of hemorrhagic cystitis after marrow transplantation". Urology. 46 (6): 811–5. doi:10.1016/S0090-4295(99)80349-5. PMID 7502421.