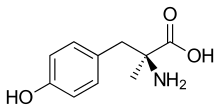
Metirosine
Metirosine (INN and BAN; α-Methyltyrosine, Metyrosine USAN, AMPT) is an antihypertensive drug. It inhibits the enzyme tyrosine hydroxylase and, therefore, catecholamine synthesis, which, as a consequence, depletes the levels of the catecholamines dopamine, adrenaline and noradrenaline in the body.
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Demser |
| Other names | Metyrosine (USAN US) |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| License data |
|
| Pregnancy category |
|
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 3.4–3.7 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.010.546 |
| Chemical and physical data | |
| Formula | C10H13NO3 |
| Molar mass | 195.218 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
It is available as a generic medication.[1]
Clinical use
Metirosine has been shown to suppress catecholamine synthesis and alleviate symptoms related to catecholamine excess, including hypertension, headache, tachycardia, constipation, and tremor.[2] Metirosine is primarily used to reduce these symptoms in patients with pheochromocytoma.[3] It is contraindicated for the treatment of essential hypertension.
Metirosine is used as an off-label treatment for DiGeorge syndrome.[4]
Metirosine is used in scientific research to investigate the effects of catecholamine depletion on behavior.[5] There is evidence that catecholamine depletion causes an increase in sleepiness that is more pronounced than sleep deprivation, and that the fatigue lingers after the drug is discontinued. Catecholamine depletion has also been linked to a negative mood, though this is reported less often than sleepiness.[6]
See also
References
- "Metyrosine: FDA-Approved Drugs". U.S. Food and Drug Administration. Retrieved 15 August 2020.
- Naruse M, Satoh F, Tanabe A, Okamoto T, Ichihara A, Tsuiki M, et al. (March 2018). "Efficacy and safety of metyrosine in pheochromocytoma/paraganglioma: a multi-center trial in Japan". Endocrine Journal. 65 (3): 359–371. doi:10.1507/endocrj.EJ17-0276. PMID 29353821.
- Green KN, Larsson SK, Beevers DG, Bevan PG, Hayes B (August 1982). "Alpha-methyltyrosine in the management of phaeochromocytoma". Thorax. 37 (8): 632–633. doi:10.1136/thx.37.8.632. PMC 459390. PMID 7179194.
- Talan J (30 April 2021). "Doctors said the boy was suffering from teenage psychosis. What he really had was a rare genetic condition". The Washington Post.
- O'Leary OF, Bechtholt AJ, Crowley JJ, Hill TE, Page ME, Lucki I (June 2007). "Depletion of serotonin and catecholamines block the acute behavioral response to different classes of antidepressant drugs in the mouse tail suspension test". Psychopharmacology. 192 (3): 357–371. doi:10.1007/s00213-007-0728-9. PMID 17318507. S2CID 24850438.
- McCann UD, Penetar DM, Shaham Y, Thorne DR, Sing HC, Thomas ML, et al. (June 1993). "Effects of catecholamine depletion on alertness and mood in rested and sleep deprived normal volunteers". Neuropsychopharmacology. 8 (4): 345–356. doi:10.1038/npp.1993.34. PMID 8099791.
External links
- "Metyrosine". Drug Information Portal. U.S. National Library of Medicine.