Nandrolone propionate
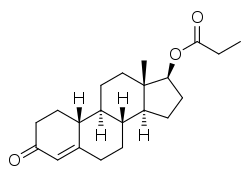
Nandrolone propionate (brand names Anabolicus, Nor-Anabol, Nortesto, Norybol-19, Pondus, Testobolin), or nandrolone propanoate, also known as 19-nortestosterone 17β-propionate, is a synthetic androgen and anabolic steroid and nandrolone ester that is or has been marketed in Spain.[1][2][3][4][5]
| Compound | PRTooltip Progesterone receptor | ARTooltip Androgen receptor | ERTooltip Estrogen receptor | GRTooltip Glucocorticoid receptor | MRTooltip Mineralocorticoid receptor | SHBGTooltip Sex hormone-binding globulin | CBGTooltip Corticosteroid-binding globulin |
|---|---|---|---|---|---|---|---|
| Nandrolone | 20 | 154–155 | <0.1 | 0.5 | 1.6 | 1–16 | 0.1 |
| Testosterone | 1.0–1.2 | 100 | <0.1 | 0.17 | 0.9 | 19–82 | 3–8 |
| Estradiol | 2.6 | 7.9 | 100 | 0.6 | 0.13 | 8.7–12 | <0.1 |
| Notes: Values are percentages (%). Reference ligands (100%) were progesterone for the PRTooltip progesterone receptor, testosterone for the ARTooltip androgen receptor, estradiol for the ERTooltip estrogen receptor, dexamethasone for the GRTooltip glucocorticoid receptor, aldosterone for the MRTooltip mineralocorticoid receptor, dihydrotestosterone for SHBGTooltip sex hormone-binding globulin, and cortisol for CBGTooltip corticosteroid-binding globulin. Sources: See template. | |||||||
Not to be confused with Nandrolone phenylpropionate.
 | |
| Clinical data | |
|---|---|
| Trade names | Anabolicus, Nor-Anabol, Nortesto, Norybol-19, Pondus, Testobolin |
| Other names | Nandrolone propanoate; 19-Nortestosterone 17β-propionate |
| Routes of administration | Intramuscular injection |
| Drug class | Androgen; Anabolic steroid; Androgen ester; Progestogen |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.027.807 |
| Chemical and physical data | |
| Formula | C21H30O3 |
| Molar mass | 330.468 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
References
- Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 660–. ISBN 978-1-4757-2085-3.
- Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 716–717. ISBN 978-3-88763-075-1.
- Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. ISBN 978-94-011-4439-1.
- Negwer M, Scharnow HG (2001). Organic-chemical drugs and their synonyms: (an international survey). Wiley-VCH. p. 2177. ISBN 978-3-527-30247-5.
- Mozayani A, Raymon L (15 October 2003). Handbook of Drug Interactions: A Clinical and Forensic Guide. Springer Science & Business Media. pp. 501–. ISBN 978-1-59259-654-6.
| Progestogens (and progestins) |
| ||||
|---|---|---|---|---|---|
| Antiprogestogens |
| ||||
| |||||
| ARTooltip Androgen receptor |
| ||||||
|---|---|---|---|---|---|---|---|
| GPRC6A |
| ||||||
| |||||||
| PRTooltip Progesterone receptor |
| ||||||
|---|---|---|---|---|---|---|---|
| mPRTooltip Membrane progesterone receptor (PAQRTooltip Progestin and adipoQ receptor) |
| ||||||
| |||||||
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.