Electric fish
An electric fish is any fish that can generate electric fields. Most electric fish are also electroreceptive, meaning that they can sense electric fields. The only exception is the stargazer family (Uranoscopidae). Electric fish, although a small minority of all fishes, include both oceanic and freshwater species, and both cartilaginous and bony fishes.
.jpg.webp)
Electric fish produce their electrical fields from an electric organ. This is made up of electrocytes, modified muscle or nerve cells, specialized for producing strong electric fields, used to locate prey, for defence against predators, and for signalling, such as in courtship. Electric organ discharges are two types, pulse and wave, and vary both by species and by function.
Electric fish have evolved many specialised behaviours. The predatory African sharptooth catfish eavesdrops on its weakly electric mormyrid prey to locate it when hunting, driving the prey fish to develop electric signals that are harder to detect. Bluntnose knifefishes produce an electric discharge pattern similar to the electrolocation pattern of the dangerous electric eel, probably a form of Batesian mimicry to dissuade predators. Glass knifefish that are using similar frequencies move their frequencies up or down in a jamming avoidance response; African knifefish have convergently evolved a nearly identical mechanism.
Evolution and phylogeny
All fish, indeed all vertebrates, use electrical signals in their nerves and muscles.[1] Cartilaginous fishes and some other basal groups use passive electrolocation with sensors that detect electric fields;[2] the platypus and echidna have separately evolved this ability. The knifefishes and elephantfishes actively electrolocate, generating weak electric fields to find prey. Finally, fish in several groups have the ability to deliver electric shocks powerful enough to stun their prey or repel predators. Among these, only the stargazers, a group of marine bony fish, do not also use electrolocation.[3][4]
In vertebrates, electroreception is an ancestral trait, meaning that it was present in their last common ancestor.[2] This form of ancestral electroreception is called ampullary electroreception, from the name of the receptive organs involved, ampullae of Lorenzini. These evolved from the mechanical sensors of the lateral line, and exist in cartilaginous fishes (sharks, rays, and chimaeras), lungfishes, bichirs, coelacanths, sturgeons, paddlefish, aquatic salamanders, and caecilians. Ampullae of Lorenzini were lost early in the evolution of bony fishes and tetrapods. Where electroreception does occur in these groups, it has secondarily been acquired in evolution, using organs other than and not homologous with ampullae of Lorenzini.[2][5] Most common bony fish are non-electric. There are some 350 species of electric fish.[6]
Electric organs have evolved eight times, four of these being organs powerful enough to deliver an electric shock. Each such group is a clade.[7][2] Most electric organs evolved from myogenic tissue (which forms muscle), however, one group of Gymnotiformes, the Apteronotidae, derived their electric organ from neurogenic tissue (which forms nerves).[8] In Gymnarchus niloticus (the African knifefish), the tail, trunk, hypobranchial, and eye muscles are incorporated into the organ, most likely to provide rigid fixation for the electrodes while swimming. In some other species, the tail fin is lost or reduced. This may reduce lateral bending while swimming, allowing the electric field to remain stable for electrolocation. There has been convergent evolution in these features among the mormyrids and gymnotids. Electric fish species that live in habitats with few obstructions, such as some bottom-living fish, display these features less prominently. This implies that convergence for electrolocation is indeed what has driven the evolution of the electric organs in the two groups.[9][10]
Actively electrolocating fish are marked on the phylogenetic tree with a small yellow lightning flash ![]() . Fish able to deliver electric shocks are marked with a red lightning flash
. Fish able to deliver electric shocks are marked with a red lightning flash ![]() . Non-electric and purely passively electrolocating species are not shown.[2][11][10]
. Non-electric and purely passively electrolocating species are not shown.[2][11][10]
| Vertebrates |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Amp. of Lorenzini |
Weakly electric fish

Weakly electric fish generate a discharge that is typically less than one volt. These are too weak to stun prey and instead are used for navigation, electrolocation in conjunction with electroreceptors in their skin, and electrocommunication with other electric fish. The major groups of weakly electric fish are the Osteoglossiformes, which include the Mormyridae (elephantfishes) and the African knifefish Gymnarchus, and the Gymnotiformes (South American knifefishes). These two groups have evolved convergently, with similar behaviour and abilities but different types of electroreceptors and differently sited electric organs.[2][11]
Strongly electric fish
Strongly electric fish, namely the electric eels, the electric catfishes, the electric rays, and the stargazers, have an electric organ discharge powerful enough to stun prey or be used for defence,[14] and navigation.[15][9][16] The electric eel, even when very small in size, can deliver substantial electric power, and enough current to exceed many species' pain threshold.[17] Electric eels sometimes leap out of the water to electrify possible predators directly, as has been tested with a human arm.[17]
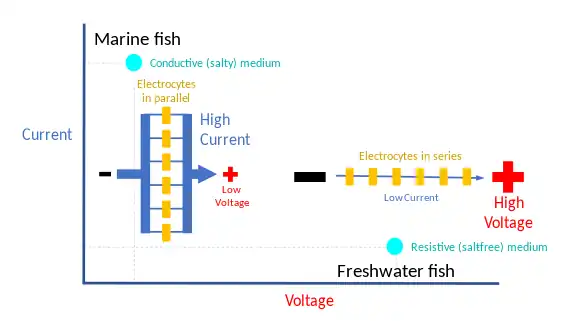
The amplitude of the electrical output from these fish can range from 10 to 860 volts with a current of up to 1 ampere, according to the surroundings, for example different conductances of salt and freshwater. To maximize the power delivered to the surroundings, the impedances of the electric organ and the water must be matched:[13]
- Strongly electric marine fish give low voltage, high current electric discharges. In salt water, a small voltage can drive a large current limited by the internal resistance of the electric organ. Hence, the electric organ consists of many electrocytes in parallel.
- Freshwater fish have high voltage, low current discharges. In freshwater, the power is limited by the voltage needed to drive the current through the large resistance of the medium. Hence, these fish have numerous cells in series.[13]
Electric organ
Anatomy
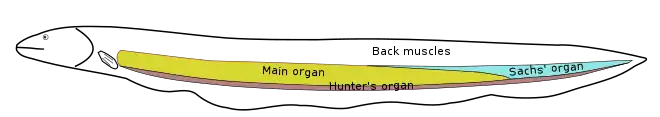
Electric organs vary widely among electric fish groups. They evolved from excitable, electrically active tissues that make use of action potentials for their function: most derive from muscle tissue, but in some groups the organ derives from nerve tissue.[18] The organ may lie along the body's axis, as in the electric eel and Gymnarchus; it may be in the tail, as in the elephantfishes; or it may be in the head, as in the electric rays and the stargazers.[3][8][19]
Physiology
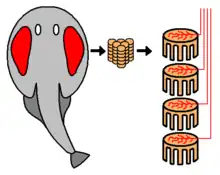
Electric organs are made up of electrocytes, large, flat cells that create and store electrical energy, awaiting discharge. The anterior ends of these cells react to stimuli from the nervous system and contain sodium channels. The posterior ends contain sodium–potassium pumps. Electrocytes become polar when triggered by a signal from the nervous system. Neurons release the neurotransmitter acetylcholine; this triggers acetylcholine receptors to open and sodium ions to flow into the electrocytes.[15] The influx of positively charged sodium ions causes the cell membrane to depolarize slightly. This in turn causes the gated sodium channels at the anterior end of the cell to open, and a flood of sodium ions enters the cell. Consequently, the anterior end of the electrocyte becomes highly positive, while the posterior end, which continues to pump out sodium ions, remains negative. This sets up a potential difference (a voltage) between the ends of the cell. After the voltage is released, the cell membranes go back to their resting potentials until they are triggered again.[15]
Discharge patterns
Electric organ discharges (EODs) need to vary with time for electrolocation, whether with pulses, as in the Mormyridae, or with waves, as in the Torpediniformes and Gymnarchus, the African knifefish.[19][20][21] Many electric fishes also use EODs for communication, while strongly electric species use them for hunting or defence.[20] Their electric signals are often simple and stereotyped, the same on every occasion.[19]
Electrocommunication
Weakly electric fish can communicate by modulating the electrical waveform they generate. They may use this to attract mates and in territorial displays.[22]
Sexual behaviour
In sexually dimorphic signalling, as in the brown ghost knifefish (Apteronotus leptorhynchus), the electric organ produces distinct signals to be received by individuals of the same or other species.[23] The electric organ fires to produce an EOD with a certain frequency, along with short modulations termed "chirps" and "gradual frequency rises", both varying widely between species and differing between the sexes.[24][20] For example, in the glass knifefish genus Eigenmannia, females produce a nearly pure sine wave with few harmonics, males produce a far sharper non-sinusoidal waveform with strong harmonics.[25]
Male bluntnose knifefishes (Brachyhypopomus) produce a continuous electric "hum" to attract females; this consumes 11–22% of their total energy budget, whereas female electrocommunication consumes only 3%. Large males produced signals of larger amplitude, and these are preferred by the females. The cost to males is reduced by a circadian rhythm, with more activity coinciding with night-time courtship and spawning, and less at other times.[26]
Antipredator behaviour
Electric catfish (Malapteruridae) frequently use their electric discharges to ward off other species from their shelter sites, whereas with their own species they have ritualized fights with open-mouth displays and sometimes bites, but rarely use electric organ discharges.[27]
The electric discharge pattern of bluntnose knifefishes is similar to the low voltage electrolocative discharge of the electric eel. This is thought to be a form of bluffing Batesian mimicry of the powerfully protected electric eel.[28]
Fish that prey on electrolocating fish may "eavesdrop"[29] on the discharges of their prey to detect them. The electroreceptive African sharptooth catfish (Clarias gariepinus) may hunt the weakly electric mormyrid, Marcusenius macrolepidotus in this way.[30] This has driven the prey, in an evolutionary arms race, to develop more complex or higher frequency signals that are harder to detect.[31]
Jamming avoidance response
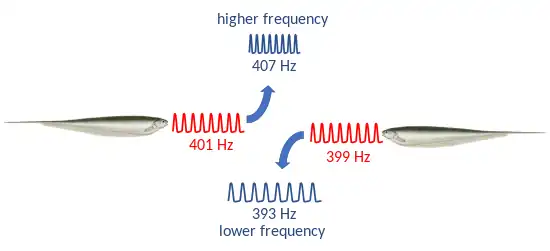
It had been theorized as early as the 1950s that electric fish near each other might experience some type of interference. In 1963, Akira Watanabe and Kimihisa Takeda discovered the jamming avoidance response in Eigenmannia.[32] When two fish are approaching one another, their electric fields interfere.[33] This sets up a beat with a frequency equal to the difference between the discharge frequencies of the two fish.[33] The jamming avoidance response comes into play when fish are exposed to a slow beat. If the neighbour's frequency is higher, the fish lowers its frequency, and vice versa.[32][25] A similar jamming avoidance response was discovered in the distantly related Gymnarchus niloticus, the African knifefish, by Walter Heiligenberg in 1975, in a further example of convergent evolution between the electric fishes of Africa and South America.[34] Both the neural computational mechanisms and the behavioural responses are nearly identical in the two groups.[35]
See also
References
- Belanger, J. H. (1 August 2005). "Contrasting Tactics in Motor Control by Vertebrates and Arthropods". Integrative and Comparative Biology. 45 (4): 672–678. doi:10.1093/icb/45.4.672. PMID 21676816.
- Bullock, Theodore H.; Bodznick, D. A.; Northcutt, R. G. (1983). "The phylogenetic distribution of electroreception: Evidence for convergent evolution of a primitive vertebrate sense modality" (PDF). Brain Research Reviews. 6 (1): 25–46. doi:10.1016/0165-0173(83)90003-6. hdl:2027.42/25137. PMID 6616267. S2CID 15603518.
- Alves-Gomes, J. (2001). "The evolution of electroreception and bioelectrogenesis in teleost fish: a phylogenietic perspective". Journal of Fish Biology. 58 (6): 1489–1511. doi:10.1111/j.1095-8649.2001.tb02307.x.
- Berry, Frederick H.; Anderson, William W. (1961). "Stargazer fishes from the western north Atlantic (Family Uranoscopidae)" (PDF). Proceedings of the United States National Museum. 1961.
- King, Benedict; Hu, Yuzhi; Long, John A. (11 February 2018). "Electroreception in early vertebrates: survey, evidence and new information". Palaeontology. 61 (3): 325–358. doi:10.1111/pala.12346.
- Rubega, Margaret (December 1999). "Vertebrate Life. F. Harvey Pough , Christine M. Janis , John B. Heiser". The Quarterly Review of Biology. 74 (4): 478–479. doi:10.1086/394168.
- Kirschbaum, Frank (2019). "Structure and Function of Electric Organs". The Histology of Fishes. Boca Raton, Florida: CRC Press. pp. 75–87. doi:10.1201/9780429113581-5. ISBN 9780429113581. S2CID 216572032.
- Bullock, Theodore H.; Hopkins, Carl D.; Popper, Arthur N.; Fay, Richard R., eds. (2005). "Electroreception". Springer Handbook of Auditory Research. 21. doi:10.1007/0-387-28275-0. ISBN 978-0-387-23192-1.
- Lissmann, H. W. (1958-03-01). "On the Function and Evolution of Electric Organs in Fish". Journal of Experimental Biology. 35 (1): 156–191. doi:10.1242/jeb.35.1.156.
- Lavoué, Sébastien; Miya, Masaki; Arnegard, Matthew E.; Sullivan, John P.; Hopkins, Carl D.; Nishida, Mutsumi (14 May 2012). "Comparable Ages for the Independent Origins of Electrogenesis in African and South American Weakly Electric Fishes". PLOS One. 7 (5): e36287. Bibcode:2012PLoSO...736287L. doi:10.1371/journal.pone.0036287. PMC 3351409. PMID 22606250.
- Lavoué, Sébastien; Miya, Masaki; Arnegard, Matthew E.; Sullivan, John P.; Hopkins, Carl D.; Nishida, Mutsumi (2012-05-14). Murphy, William J. (ed.). "Comparable Ages for the Independent Origins of Electrogenesis in African and South American Weakly Electric Fishes". PLOS ONE. 7 (5): e36287. Bibcode:2012PLoSO...736287L. doi:10.1371/journal.pone.0036287. PMC 3351409. PMID 22606250.
- Von der Emde, G. (1999). "Active electrolocation of objects in weakly electric fish". Journal of Experimental Biology, 202 (10): 1205–1215. Full text
- Kramer, Bernd (2008). "Electric Organ Discharge". In Marc D. Binder; Nobutaka Hirokawa; Uwe Windhorst (eds.). Encyclopedia of Neuroscience. Berlin, Heidelberg: Springer. pp. 1050–1056. ISBN 978-3-540-23735-8. Retrieved 2012-03-25.
- Macesic, Laura J.; Kajiura, Stephen M. (November 2009). "Electric organ morphology and function in the lesser electric ray, Narcine brasiliensis". Zoology. 112 (6): 442–450. doi:10.1016/j.zool.2009.02.002. PMID 19651501.
- Traeger, Lindsay L.; Sabat, Grzegorz; Barrett-Wilt, Gregory A.; Wells, Gregg B.; Sussman, Michael R. (July 2017). "A tail of two voltages: Proteomic comparison of the three electric organs of the electric eel". Science Advances. 3 (7): e1700523. Bibcode:2017SciA....3E0523T. doi:10.1126/sciadv.1700523. PMC 5498108. PMID 28695212.
- Nelson, Mark. "What IS an electric fish?". Retrieved 10 August 2014.
- Catania, Kenneth C. (2017-09-25). "Power Transfer to a Human during an Electric Eel's Shocking Leap". Current Biology. 27 (18): 2887–2891.e2. doi:10.1016/j.cub.2017.08.034. PMID 28918950. S2CID 13359966.
- Albert, J. S.; Crampton, William G. R. (2006). "Electroreception and electrogenesis". In Evans, David H.; Claiborne, James B. (eds.). The Physiology of Fishes (3rd ed.). CRC Press. pp. 431–472. ISBN 978-0-8493-2022-4.
- Crampton, William G. R. (2019-02-05). "Electroreception, electrogenesis and electric signal evolution". Journal of Fish Biology. 95 (1): 92–134. doi:10.1111/jfb.13922. PMID 30729523. S2CID 73442571.
- Nagel, Rebecca; Kirschbaum, Frank; Hofmann, Volker; Engelmann, Jacob; Tiedemann, Ralph (December 2018). "Electric pulse characteristics can enable species recognition in African weakly electric fish species". Scientific Reports. 8 (1): 10799. Bibcode:2018NatSR...810799N. doi:10.1038/s41598-018-29132-z. PMC 6050243. PMID 30018286.
- Kawasaki, M. (2011). "Detection and generation of electric signals". Encyclopedia of Fish Physiology. Elsevier. pp. 398–408. doi:10.1016/b978-0-12-374553-8.00136-2.
- Hopkins, C. D. (1999). "Design features for electric communication". Journal of Experimental Biology. 202 (Pt 10): 1217–1228. doi:10.1242/jeb.202.10.1217. PMID 10210663.
- Fukutomi, Matasaburo; Carlson, Bruce A. (2020-08-12). "Signal Diversification Is Associated with Corollary Discharge Evolution in Weakly Electric Fish". The Journal of Neuroscience. 40 (33): 6345–6356. doi:10.1523/JNEUROSCI.0875-20.2020. PMC 7424872. PMID 32661026.
- Ho, Winnie W.; Fernandes, Cristina Cox; Alves-Gomes, José A.; Smith, G. Troy (2010-07-11). "Sex Differences in the Electrocommunication Signals of the Electric Fish Apteronotus bonapartii". Ethology. 116 (11): 1050–1064. doi:10.1111/j.1439-0310.2010.01823.x. PMC 2953865. PMID 20953311.
- Kramer, Berndt (15 May 1999). "Waveform discrimination, phase sensitivity and jamming avoidance in a wave-type electric fish". Journal of Experimental Biology. 202 (10): 1387–1398. doi:10.1242/jeb.202.10.1387. PMID 10210679.
- Salazar, Vielka L.; Stoddard, Philip K. (15 March 2008). "Sex differences in energetic costs explain sexual dimorphism in the circadian rhythm modulation of the electrocommunication signal of the gymnotiform fish Brachyhypopomus pinnicaudatus". Journal of Experimental Biology. 211 (6): 1012–1020. doi:10.1242/jeb.014795. PMID 18310126. S2CID 14310938.
- Rankin, Catharine H.; Moller, Peter (26 April 2010). "Social Behavior of the African Electric Catfish, Malapterurus electricus, during Intra- and Interspecific Encounters". Ethology. Wiley. 73 (3): 177–190. doi:10.1111/j.1439-0310.1986.tb00909.x.
- Stoddard, P. K. (1999). "Predation enhances complexity in the evolution of electric fish signals". Nature. 400 (6741): 254–256. Bibcode:1999Natur.400..254S. doi:10.1038/22301. PMID 10421365. S2CID 204994529.
- Falk, Jay J.; ter Hofstede, Hannah M.; Jones, Patricia L.; et al. (7 June 2015). "Sensory-based niche partitioning in a multiple predator–multiple prey community". Proceedings of the Royal Society B: Biological Sciences. 282 (1808). doi:10.1098/rspb.2015.0520. PMC 4455811. PMID 25994677.
- Merron, G. S. (1993). "Pack-hunting in two species of catfish, Clavias gariepinus and C. ngamensis, in the Okavango Delta, Botswana". Journal of Fish Biology. 43 (4): 575–584. doi:10.1111/j.1095-8649.1993.tb00440.x.
- Stoddard, P. K. (2002). "The evolutionary origins of electric signal complexity". Journal of Physiology - Paris. 96 (5–6): 485–491. doi:10.1016/S0928-4257(03)00004-4. PMID 14692496. S2CID 6240530.
- Bullock, Theodore H.; Hamstra, R. Jr.; Scheich, H. (1972). "The jamming avoidance response of high frequency electric fish". Journal of Comparative Physiology (77): 1–22.
- Shifman, Aaron R.; Lewis, John E. (2018-01-04). "The complexity of high-frequency electric fields degrades electrosensory inputs: implications for the jamming avoidance response in weakly electric fish". Journal of the Royal Society Interface. 15 (138): 20170633. doi:10.1098/rsif.2017.0633. PMC 5805966. PMID 29367237.
- Heiligenberg, Walter (1975). "Electrolocation and jamming avoidance in the electric fishGymnarchus niloticus (Gymnarchidae, Mormyriformes)". Journal of Comparative Physiology A. 103 (1): 55–67. doi:10.1007/bf01380044. S2CID 42982465.
- Kawasaki, M. (1975). "Independently evolved jamming avoidance responses in Gymnotid and Gymnarchid electric fish: a case of convergent evolution of behavior and its sensory basis". Journal of Comparative Physiology (103): 97–121.

