Alatrofloxacin
Alatrofloxacin (Trovan IV) is a fluoroquinolone antibiotic developed by Pfizer, delivered as a mesylate salt.[1]
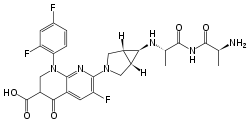 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a605016 |
| Routes of administration | Intravenous |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | N/A |
| Protein binding | 76% (trovafloxacin) |
| Metabolism | Quickly hydrolyzed to trovafloxacin |
| Elimination half-life | 9 to 12 hours (trovafloxacin) |
| Excretion | Fecal and renal (trovafloxacin) |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C26H25F3N6O5 |
| Molar mass | 558.518 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Trovafloxacin and alatrofloxacin were both withdrawn from the U.S. market in June 2006 due to hepatotoxicity leading to liver transplant or death.[2]
See also
References
- "Center for Drug Evaluation and Research – Application Number: 020759/020760 – Chemistry Review(s)" (PDF). Food and Drug Administration. Retrieved 29 August 2014.
- Qureshi ZP, Seoane-Vazquez E, Rodriguez-Monguio R, Stevenson KB, Szeinbach SL (July 2011). "Market withdrawal of new molecular entities approved in the United States from 1980 to 2009". Pharmacoepidemiology and Drug Safety. 20 (7): 772–777. doi:10.1002/pds.2155. PMID 21574210. S2CID 23821961.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.