Colestilan
Colestilan (INN, trade name BindRen) is a medication that acts as a phosphate binder[1] and bile acid sequestrant.[2] It is an ion-exchange resin, is an orally administered bile acid sequestrant that is being developed by Mitsubishi Tanabe Pharma Corporation for the treatment of hypercholesterolaemia and hyperphosphataemia. It has been launched in Japan for hypercholesterolaemia. For the treatment of hyperphosphataemia, it is launched in Austria, Germany, the Czech Republic, Portugal and the United Kingdom, is registered in the EU. Phase III development in paediatric patients with hyperphosphataemia associated with chronic kidney disease was underway in the UK and Germany. However, the company discontinued the development. In addition, the phase II development in type-2 diabetes mellitus and phase I development in hyperphosphataemia, in Japan, was also discontinued by the company.[3]
 | |
| Clinical data | |
|---|---|
| Trade names | BindRen |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Not absorbed |
| Protein binding | NA |
| Metabolism | Not absorbed |
| Excretion | Gut |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
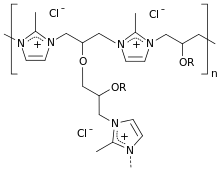
| Formula | (C4H5ClN2)m(C3H6O)n |
| | |
Phase III development for hyperphosphataemia was previously underway in the US. However, Mitsubishi Tanabe Pharma discontinued development in this market. Colestilan was also investigated in phase III trials in Europe and Asia for hypercholesterolaemia. However, as of March 2015, no recent reports of development have been identified for the drug in this indication[3]
Clinical use
Colestilan is used for the treatment of hyperphosphataemia (too high phosphate concentrations in the blood serum) in patients undergoing dialysis, including peritoneal dialysis.[1][4]
Contraindications
Colestilan is contraindicated in patients with bowel obstruction.[4]
Interactions
The substance can inhibit the resorption of other drugs, as well as fat soluble vitamins (A, D, E, K) and folate, from the gut.[1] Resulting lower blood levels can be clinically problematic with immunosuppressant and antiepileptic drugs.[4]
Adverse effects
Adverse effects include gastrointestinal problems such as constipation, as well as vitamin and calcium deficiency. Vitamin K deficiency sometimes causes gastrointestinal bleeding.[1][4]
Chemistry and mechanism of action
Colestilan is a cross-linked copolymer of 2-methylimidazole and epichlorohydrin and works as an anion exchanger resin with affinity to phosphate, bile acid anions and urate. It binds these anions in the gut and removes them from the enterohepatic circulation. Colestilan is not absorbed from the gut, but is excreted together with the bound anions.[1]

Notes and references
- Klement A (11 November 2013). "Dialysepflichtig – weniger Phosphat mit BindRen". Österreichische Apothekerzeitung (in German) (23/2013): 28f.
- Handelsman Y (May 2011). "Role of bile acid sequestrants in the treatment of type 2 diabetes". Diabetes Care. 34 Suppl 2: S244-50. doi:10.2337/dc11-s237. PMC 3632187. PMID 21525463.
- "Drug Profile:Colestilan". Adis Insight. Springer Nature Switzerland AG. December 2, 2015. Retrieved December 16, 2015.
- Haberfeld H, ed. (2013). Austria-Codex (in German). Vienna: Österreichischer Apothekerverlag. BindRen 1 g Filmtabletten.