Foliglurax
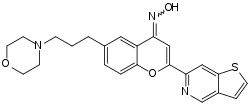
Foliglurax (developmental code names PXT-002331, DT2331) is a positive allosteric modulator of the metabotropic glutamate receptor 4 (mGluR4),[1] which is under development by Prexton Therapeutics for the treatment of Parkinson's disease.[2][3][4] It reached phase II clinical trials,[2] but while it was found to be safe and showed some signs of clinical improvement, it failed to sufficiently distingush itself from placebo to meet the study endpoints.[5][6]
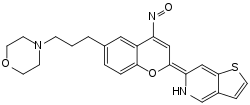
The other tautomeric form of foliglurax.
 | |
| Clinical data | |
|---|---|
| Other names | DT2331; PXT-002331; PXT-2331 |
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| Chemical and physical data | |
| Formula | C23H23N3O3S |
| Molar mass | 421.52 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
References
- Charvin D, Pomel V, Ortiz M, Frauli M, Scheffler S, Steinberg E, et al. (October 2017). "Discovery, Structure-Activity Relationship, and Antiparkinsonian Effect of a Potent and Brain-Penetrant Chemical Series of Positive Allosteric Modulators of Metabotropic Glutamate Receptor 4". Journal of Medicinal Chemistry. 60 (20): 8515–8537. doi:10.1021/acs.jmedchem.7b00991. PMID 28902994.
- "Foliglurax - Lundbeck". Adis Insight. Springer Nature Switzerland AG.
- Jankovic J, Aguilar LG (August 2008). "Current approaches to the treatment of Parkinson's disease". Neuropsychiatric Disease and Treatment. 4 (4): 743–57. doi:10.1016/j.bmcl.2017.07.075. PMC 2536542. PMID 19043519.
- Volpi C, Fallarino F, Mondanelli G, Macchiarulo A, Grohmann U (May 2018). "Opportunities and challenges in drug discovery targeting metabotropic glutamate receptor 4". Expert Opinion on Drug Discovery. 13 (5): 411–423. doi:10.1080/17460441.2018.1443076. PMID 29486616. S2CID 4572251.
- Gonzalez-Latapi P, Bhowmick SS, Saranza G, Fox SH (October 2020). "Non-Dopaminergic Treatments for Motor Control in Parkinson's Disease: An Update". CNS Drugs. 34 (10): 1025–1044. doi:10.1007/s40263-020-00754-0. PMID 32785890. S2CID 221111043.
- Rascol O, Medori R, Baayen C, Such P, Meulien D (February 2022). "A Randomized, Double-Blind, Controlled Phase II Study of Foliglurax in Parkinson's Disease". Movement Disorders. 37 (5): 1088–1093. doi:10.1002/mds.28970. PMC 9303267. PMID 35218231. S2CID 247129161.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.