Risk factors for breast cancer
Risk factors for breast cancer may be divided into preventable and non-preventable. Their study belongs in the field of epidemiology. Breast cancer, like other forms of cancer, can result from multiple environmental and hereditary risk factors. The term "environmental", as used by cancer researchers, means any risk factor that is not genetically inherited.
For breast cancer, the list of environmental risk factors includes the individual person's development, exposure to microbes, "medical interventions, dietary exposures to nutrients, energy and toxicants, ionizing radiation, and chemicals from industrial and agricultural processes and from consumer products...reproductive choices, energy balance, adult weight gain, body fatness, voluntary and involuntary physical activity, medical care, exposure to tobacco smoke and alcohol, and occupational exposures, including shift work" as well as "metabolic and physiologic processes that modify the body's internal environment."[1] Some of these environmental factors are part of the physical environment, while others (such as diet and number of pregnancies) are primarily part of the social, cultural, or economic environment.[1]
Although many epidemiological risk factors have been identified, the cause of any individual breast cancer is most often unknowable. Epidemiological research informs the patterns of breast cancer incidence across certain populations, but not in a given individual. Approximately 5% of new breast cancers are attributable to hereditary syndromes, and well-established risk factors accounts for approximately 30% of cases.[2]
Age
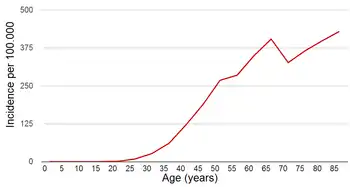
The risk of getting breast cancer increases with age. A woman is more than 100 times more likely to develop breast cancer in her 60s than in her 20s.[4] If all women lived to age 95, about one in eight would be diagnosed with breast cancer at some point during their lives.[5] However, the actual lifetime risk is lower than that, because 90% of women die before age 95, most commonly from heart attacks, strokes, or other forms of cancer.
The probability of breast cancer increases with age, but breast cancer tends to be more aggressive in younger people.
Sex
Male individuals have a much lower risk of developing breast cancer than females. In developed countries, about 99% of breast cancer cases are diagnosed in female patients; in a few African countries, which represent the highest incidence of male breast cancer, males account for 5–15% of cases.[4] The rate of male breast cancer appears to be rising somewhat.[6]
Male breast cancer patients tend to be older than female ones.[4] They are more likely to be diagnosed with hormone-receptor positive tumors, with about six out of seven cases being estrogen-receptor positive.[4] The overall prognosis is worse for male than for female patients.[4]
Heredity
United Kingdom being a member of the International Cancer Genome Consortium is leading efforts to map breast cancer's complete genome.
BRCA1 and BRCA2
In 5% of breast cancer cases, there is a strong inherited familial risk.[7]
Two autosomal dominant genes, BRCA1 and BRCA2, account for most of the cases of familial breast cancer. Women who carry a harmful BRCA mutation have a 60% to 80% risk of developing breast cancer in their lifetimes.[7] Other associated malignancies include ovarian cancer and pancreatic cancer. If a mother or a sister was diagnosed breast cancer, the risk of a hereditary BRCA1 or BRCA2 gene mutation is about 2-fold higher than those women without a familial history. Commercial testing for BRCA1 and BRCA2 gene mutations has been available in most developed countries since at least 2004.
In addition to the BRCA genes associated with breast cancer, the presence of NBR2, near breast cancer gene 1, has been discovered, and research into its contribution to breast cancer pathogenesis is ongoing.[8]
Other genes
Hereditary non-BRCA1 and non-BRCA2 breast tumors (and even some sporadic carcinomas) are believed to result from the expression of weakly penetrant but highly prevalent mutations in various genes. For instance, polymorphism has been identified in genes associated to the metabolism of estrogens and/or carcinogens (Cytochrome P450, family 1, member A1, CYP1B1, CYP17A1, CYP19, Catechol-O-methyltransferase, N-acetyltransferase 2, Glutathione S-transferase Mu 1, GSTP1, GSTT, . . . ), to estrogen, androgen and vitamin D action (ESR1, AR, VDR), to co-activation of gene transcription (AIB1), to DNA damage response pathways (CHEK2, HRAS1, XRCC1, XRCC3, XRCC5).[9] Sequence variants of these genes that are relatively common in the population may be associated with a small to moderate increased relative risk for breast cancer. Combinations of such variants could lead to multiplicative effects. Sporadic cancers likely result from the complex interplay between the expression of low penetrance genes (risk variants) and environmental factors. However, the suspected impact of most of these variants on breast cancer risk should, in most cases, be confirmed in large populations studies. Indeed, low penetrance genes cannot be easily tracked through families, as is true for dominant high-risk genes.[9]
Part of the hereditary non-BRCA1 and non-BRCA2 breast tumors may be associated to rare syndromes, of which breast cancer is only one component. Such syndromes result notably from mutations in TP53 (Li–Fraumeni syndrome), ATM (ataxia–telangiectasia), STK11/LKB1(Peutz–Jeghers syndrome), PTEN (Cowden syndrome).
RAB11FIP1,[10] TP53, PTEN and rs4973768 are also associated with increased risk of breast cancer. rs6504950 is associated with lower risk of breast cancer.[11]
Mutations in RAD51C confer an increased risk for breast and ovarian cancer.[12]
Prior cancers
People who have previously been diagnosed with breast, ovarian, uterine, or bowel cancer have a higher risk of developing breast cancer in the future.[4] Mothers of children with soft-tissue sarcoma may have an increased risk of breast cancer.[4] Men with prostate cancer may have an elevated risk of breast cancer, although the absolute risk remains low.
Dietary factors
Alcohol
In population studies that do not control for breast cancer screening incidence, drinkers of alcohol have a higher tendency to be diagnosed with breast cancer. For example, a study of more than one million middle-aged British women concluded that each daily alcoholic beverage was associated with an increased incidence of breast cancer of 11 cases per 1000 women.[13] This means that among a group of 1000 women who drink one alcoholic beverage per day, they will have 11 extra cases of breast cancer when compared to a group of women who drink less than one alcoholic beverage per week; a group of 1000 women who have four drinks per day will have an extra 44 cases of breast cancer compared to non-drinkers. One or two drinks each day increases the relative risk to 150% of normal, and six drinks per day increases the risk to 330% of normal.
However, studies of mortality show that drinkers have no greater risk of dying from breast cancer. An analysis of various causes of death of middle aged and elderly Americans[14] found that, of the 251,420 women in the study, 0.3% of the zero and super-light drinkers died from breast cancer, over the 10 years of study observation. This proportion of 0.3% was exactly the same for moderate to heavy drinkers (1 to 4+ drinks per day). In another mortality study of 85,000 women,[15] the chance of death from breast cancer during the 12 year follow-up period was 0.4%, and again this was identical for zero-to-super-light drinkers as with moderate-to-heavy drinkers. This paradoxical difference between the results for diagnoses and those for mortality appears to be due to drinkers screening more for breast cancer.[16] Potential reasons for the higher rates of screening among drinkers are because they are wealthier, more urban, more health conscious, and closer to screening clinics. Studies that control for screening rates show no association between drinking and being diagnosed with breast cancer.[17]
Meta-analysis of the epidemiological studies looking at drinking and breast cancer mortality/survival after diagnosis shows no association between levels of drinking (before or after diagnosis) and risk of breast cancer death, nor reoccurrence of the cancer.[18] Two recent studies looking at patients already diagnosed with breast cancer both found that women who drank before their breast cancer diagnosis had no higher risk of dying from the cancer than the non-drinkers.[19][20] Similarly a large study with long follow-up of women with breast cancer [21] showed breast cancer patients had better chances of survival if they were regular drinkers before diagnosis. If they altered their drinking after diagnosis this did not alter their chance of dying from breast cancer. But an increase in drinking was associated with an overall improvement in life expectancy (largely due to substantially fewer heart disease deaths among those who increased their alcohol consumption).
Fat intake
Dietary influences have been examined for decades with conflicting results and have so far failed to confirm any significant link. One recent study suggests that low-fat diets may significantly decrease the risk of breast cancer as well as the recurrence of breast cancer.[22] Another study showed no contribution of dietary fat intake on the incidence of breast cancer in over 300,000 women.[23] A randomized, controlled study of the consequences of a low-fat diet, carried out as part of the Women's Health Initiative, failed to show a statistically significant reduction in breast cancer incidence in the group assigned a low-fat diet, although the authors did find evidence of a benefit in the subgroup of women who followed the low-fat diet in a strict manner.[24] A prospective cohort study, the Nurses' Health Study II, found increased breast cancer incidence in premenopausal women only, with higher intake of animal fat, but not vegetable fat. Taken as a whole, these results point to a possible association between dietary fat intake and breast cancer incidence, though these interactions are hard to measure in large groups of women.
Specific dietary fatty acids
Although many claims have been made in popular literature there is no solid evidence linking specific fats to breast cancer.
A study published in 2001 found higher levels of monounsaturated fatty acids MUFAs (especially oleic acid) in the erythrocyte membranes of postmenopausal women who developed breast cancer.[25]
That same study discussed that a diet high in MUFAs is not the major determinant of erythrocyte membrane MUFAs, where most oleic acid in mammalian tissue is derived from the saturated stearic acid residue. Where key conversion is controlled by the Delta9-desaturase, which also regulates the transformation of the other common saturated fatty acids (SFAs) (myristic and palmitic). The study discussed that fat content of the diet has an important effect on Delta9-d activity, while high levels of SFAs increase Delta9-d activity by twofold to threefold, whereas polyunsaturated fatty acids (PUFAs) decrease.[25]
This conclusion was partially contradicted by a latter study, which showed a direct relation between very high consumption of omega-6 fatty acids (PUFAs) and breast cancer in postmenopausal women.[26]
Polycyclic Aromatic Hydrocarbons
Polycyclic aromatic hydrocarbons (PAHs) are a known human carcinogens due to their mutagenic ability to damage DNA which causes cancer. A 2016 study found that sources of PAHs such as grilled and smoked meats (along with other sources such as cigarette smoking and indoor air pollution) were associated with 30–50% increase in breast cancer incidence.[27] High risk individuals were those who were active smokers, consumed greater than 55 servings of grilled/smoked meats a year, frequently used indoor fire places, and were exposed to large amounts of traffic pollution.[27] The study was a population-based case-control study conducted on Long island, New York (N=1,508 breast cancer cases/1,556 controls). The authors concluded that while PAHs are ubiquitous, breast cancer risk factors could be modified by dietary and lifestyle changes.
Phytoestrogens
Phytoestrogens have been extensively studied in animal and human in-vitro and epidemiological studies. Research failed to establish any noticeable benefit and some phytoestrogens may present a breast cancer risk.
The literature support the following conclusions:
Calcium
Some studies have found a relationship between calcium intake and lowered breast cancer risk.
- In the Nurse's Health Study, a high dietary intake of calcium showed 33% lower risk of breast cancer.[30]
- Cancer Prevention Study II Nutrition Cohort Concluded 20% lower risk of breast cancer with 1250 mg of calcium intake.[31]
- Women's Health Study shows an inverse association between total calcium intake and premenopausal breast cancer risk.[32]
- Another two studies, one in France[33] and another in Finland,[34] showed significant inverse relation between calcium intake and breast cancer.
- Hypotheses
- Calcium reduces cell proliferation and induces differentiation in mammary glands.
- High calcium intake decreases fat-induced epithelial hypoproliferation of mammary gland and chemically induced carcinogenesis.
- Breast density is positively associated with breast cancer. Dietary calcium intake reduces the breast density.
- High calcium intake is associated with a reduced risk of benign proliferative epithelial disorders which are thought to be precursors of breast cancer.
Vitamin D
Vitamin D is related to reduced risk of breast cancer and disease prognosis. A 2011 study done at the University of Rochester Medical Center found that low vitamin D levels among women with breast cancer correlate with more aggressive tumors and poorer prognosis. The study associated sub-optimal vitamin D levels with poor scores on every major biological marker that helps physicians predict a patient's breast cancer outcome. The lead researcher stated, "Based on these results, doctors should strongly consider monitoring vitamin D levels among breast cancer patients and correcting them as needed."[35]
- Hypotheses
- Vitamin D metabolites (25 (OH) D, 1, 25 (OH) 2 D) promote cellular differentiation and it is important for chemoprevention.
- Low circulating levels of 25 (OH) D in adolescence may be an important predisposing factor for breast cancer risk in later life.
Brassica vegetables
In a study published in the Journal of the American Medical Association, biomedical investigators found that Brassicas vegetable intake (broccoli, cauliflower, cabbage, kale and Brussels sprouts) was inversely related to breast cancer development. The relative risk among women in the highest decile of Brassica vegetable consumption (median, 1.5 servings per day) compared to the lowest decile (virtually no consumption) was 0.58. That is, women who consumed around 1.5 servings of Brassica vegetables per day had 42% less risk of developing breast cancer than those who consumed virtually none.[36]
Country diet
A significant environmental effect is likely responsible for the different rates of breast cancer incidence between countries with different dietary customs. Researchers have long measured that breast cancer rates in an immigrant population soon come to resemble the rates of the host country after a few generations. The reason for this is speculated to be immigrant uptake of the host country diet. The prototypical example of this phenomenon is the changing rate of breast cancer after the arrival of Japanese immigrants to America.[37]
Mushrooms
In 2009, a case-control study of the eating habits of 2,018 women suggested that women who consumed mushrooms had an approximately 50% lower incidence of breast cancer. Women who consumed mushrooms and green tea had a 90% lower incidence of breast cancer.[38] A case control study of 362 Korean women also reported an association between mushroom consumption and decreased risk of breast cancer.[39]
Obesity and Lack of Exercise
Gaining weight after menopause can increase a woman's risk. A 2006 study found that putting on 9.9 kg (22 lbs) after menopause increased the risk of developing breast cancer by 18%.[43] Lack of exercise has been linked to breast cancer by the American Institute for Cancer Research.[44]
Obesity has been linked to an increased risk of developing breast cancer by many scientific studies.[45] There is evidence to suggest that excess body fat at the time of breast cancer diagnosis is associated with higher rates of cancer recurrence and death.[45] Furthermore, studies have shown that obese women are more likely to have large tumors, greater lymph node involvement, and poorer breast cancer prognosis with 30% higher risk of mortality.[46]
Weight gain after diagnosis has also been linked to higher rates of breast cancer recurrence or mortality although this finding is not consistent.[45] Weight gain is often less severe with newer chemotherapy treatments but one study found a significant risk of breast cancer mortality in women who gained weight compared to those who maintained their weight.[47] However, other cohort studies and recent clinical trials have not shown a significant relationship between weight gain after diagnosis and breast cancer mortality.[45][48]
Weight loss after diagnosis has not been shown to decrease the risk of breast cancer recurrence or mortality.[45] However, physical activity after breast cancer diagnosis has shown some associations with reducing breast cancer recurrence and mortality independent of weight loss.[49] Data for both weight loss and physical activity and the effect on breast cancer prognosis is still lacking.[45]
There is debate as to whether the higher rate of breast cancer associated with obesity is due to a biological difference in the cancer itself, or differences in other factors such as health screen practices.[50] It has been suggested that obesity may be a determinant for breast cancer screening by mammography. Seventeen scientific studies in the United States have found that as obesity increases in women over 40 years of age the rate of mammography reported decreases significantly.[51] When stratified by race (white vs. black) there was a stronger relationship between obesity and lack of mammography screening among white women.[51] Another study also found lower rates of mammography among those who were overweight and obese compared to those women who were of normal body mass index—this effect was only seen in white women.[52] Obese women are more likely to list pain associated with mammograms as a reason for not getting screened; however, leaner women also list this as a reason for avoiding mammograms.[53] Other reasons obese women may avoid mammography are due to lack of insurance, low income, or embarrassment at the procedure, although when these factors are accounted for, the effect of lower rates of screening are still significant.[53] In contrast, other studies have shown that mammography patterns did not differ among women who were obese compared to those at a healthy weight indicating that there may be biological differences in cancer presentation between these groups.[54]
Hormones
Persistently increased blood levels of estrogen are associated with an increased risk of breast cancer, as are increased levels of the androgens androstenedione and testosterone (which can be directly converted by aromatase to the estrogens estrone and estradiol, respectively). Increased blood levels of progesterone are associated with a decreased risk of breast cancer in premenopausal women.[55] A number of circumstances which increase exposure to endogenous estrogens including not having children, delaying first childbirth, not breastfeeding, early menarche (the first menstrual period) and late menopause are suspected of increasing lifetime risk for developing breast cancer.[56]
However, not only sex hormones, but also insulin levels are positively associated with the risk of breast cancer.[57]
Pregnancy, childbearing and breastfeeding
Lower age of first childbirth, compared to the average age of 24,[58] having more children (about 7% lowered risk per child), and breastfeeding (4.3% per breastfeeding year, with an average relative risk around 0.7[59][60]) have all been correlated to lowered breast cancer risk in premenopausal women, but not postmenopausal women, in large studies.[61] Women who give birth and breast-feed by the age of 20 may have even greater protection.[62] In contrast, for instance, having the first live birth after age 30 doubles the risk compared to having first live birth at age less than 25.[63] Never having children triples the risk.[63] The studies have found that these risk factors become less material as a woman reaches menopause, i.e. that they affect risk of breast cancer prior to menopause but not after it. In balancing premenopausal reductions in risk from childbirth and lactation, it is important also to consider the risks involved in having a child.
Hormonal contraception
Hormonal contraceptives may produce a slight increase in the risk of breast cancer diagnosis among current and recent users, but this appears to be a short-term effect. In 1996 the largest collaborative reanalysis of individual data on over 150,000 women in 54 studies of breast cancer found a relative risk (RR) of 1.24 of breast cancer diagnosis among current combined oral contraceptive pill users; 10 or more years after stopping, no difference was seen. Further, the cancers diagnosed in women who had ever used hormonal contraceptives were less advanced than those in nonusers, raising the possibility that the small excess among users was due to increased detection.[64][65] The relative risk of breast cancer diagnosis associated with current and recent use of hormonal contraceptives did not appear to vary with family history of breast cancer.[66] Some studies have suggested that women who began using hormonal contraceptives before the age of 20 or before their first full-term pregnancy are at increased risk for breast cancer, but it is not clear how much of the risk stems from early age at first use, and how much stems from use before the first full-term pregnancy.[67]
Hormone replacement therapy
Data exist from both observational and randomized clinical trials regarding the association between menopausal hormone replacement therapy (menopausal HRT) and breast cancer. The largest meta-analysis (1997) of data from 51 observational studies, indicated a relative risk of breast cancer of 1.35 for women who had used HRT for 5 or more years after menopause.[68] The estrogen-plus-progestin arm of the Women's Health Initiative (WHI), a randomized controlled trial, which randomized more than 16,000 postmenopausal women to receive combined hormone therapy or placebo, was halted early (2002) because health risks exceeded benefits. One of the adverse outcomes prompting closure was a significant increase in both total and invasive breast cancers (hazard ratio = 1.24) in women randomized to receive estrogen and progestin for an average of 5 years.[69] HRT-related breast cancers had adverse prognostic characteristics (more advanced stages and larger tumors) compared with cancers occurring in the placebo group, and HRT was also associated with a substantial increase in abnormal mammograms. Short-term use of hormones for treatment of menopausal symptoms appears to confer little or no breast cancer risk.[66] A correlation was found between the use of hormonal contraceptives and subsequent reliance on hormone replacement therapy.[67]
Oophorectomy and mastectomy
Prophylactic oophorectomy (removal of ovaries) and mastectomy in individuals with high-risk mutations of BRCA1 or BRCA2 genes reduces the risk of developing breast cancer as well as reducing the risk of developing ovarian cancer. Because of a complex balance of benefits and risks of a prophylactic surgery it is recommended only in very specific cases.
Hormonal therapy
Hormonal therapy has been used for chemoprevention in individuals at high risk for breast cancer. Overall it is recommended only in very special circumstances. In 2002, a clinical practice guideline by the US Preventive Services Task Force (USPSTF) recommended that "clinicians discuss chemoprevention with women at high risk for breast cancer and at low risk for adverse effects of chemoprevention" with a grade B recommendation.[70][71][72]
Selective estrogen receptor modulators (SERMs)
The guidelines were based on studies of SERMs from the MORE, BCPT P-1, and Italian trials. In the MORE trial, the relative risk reduction for raloxifene was 76%.[73] The P-1 preventative study demonstrated that tamoxifen can prevent breast cancer in high-risk individuals. The relative risk reduction was up to 50% of new breast cancers, though the cancers prevented were more likely estrogen-receptor positive (this is analogous to the effect of finasteride on the prevention of prostate cancer, in which only low-grade prostate cancers were prevented).[74][75] The Italian trial showed benefit from tamoxifen.[76]
Additional randomized controlled trials have been published since the guidelines. The IBIS trial found benefit from tamoxifen.[77] In 2006, the NSABP STAR trial demonstrated that raloxifene had equal efficacy in preventing breast cancer compared with tamoxifen, but that there were fewer side effects with raloxifene.[78] The RUTH Trial concluded that "benefits of raloxifene in reducing the risks of invasive breast cancer and vertebral fracture should be weighed against the increased risks of venous thromboembolism and fatal stroke".[79] On September 14, 2007, the US Food and Drug Administration approved raloxifene (Evista) to prevent invasive breast cancer in postmenopausal women.[80]
Endocrine disruptors
Many xenoestrogens (industrially made estrogenic compounds) and other endocrine disruptors are potential risk factors of breast cancer.
Diethylstilbestrol (DES) is a synthetic form of estrogen. It has been used between the early 1940s and 1971. Pregnant women took DES to prevent certain pregnancy complications. However, it also increased their risk of breast cancer. It also increased the risk of breast cancer in the prenatally exposed daughters after they have reached an age 40 years.[81]
Furthermore, there is exposure to endocrine disruptors from the environment, in addition to phytoestrogens mentioned above in the diet section. See xenoestrogens in environmental factors below
Factors in the physical environment
According to a review, the main mechanisms by which environmental compounds increase breast cancer risk are acting like hormones, especially estrogen, or affecting susceptibility to carcinogenesis.[82] The evidence to date generally supports an association between breast cancer and polycyclic aromatic hydrocarbons (PAHs) and polychlorinated biphenyls (PCBs). Dioxins and organic solvents, on the other hand, have only shown an association in sparse and methodologically limited studies, but are suggestive of an association.[82] Overall, however, evidence is still based on a relatively small number of studies.[82]
Xenoestrogens
Many xenoestrogens (industrially made estrogenic compounds) are endocrine disruptors, and potential risk factors of breast cancer. Endocrine disruption is the hypothesis that some chemicals in the body, such as Bisphenol A, are capable of interfering with the production, processing, and transmission of hormones.[83]
A substantial and growing body of evidence indicates that exposures to certain toxic chemicals and hormone-mimicking compounds including chemicals used in pesticides, cosmetics and cleaning products contribute to the development of breast cancer.
The increasing prevalence of these substances in the environment may explain the increasing incidence of breast cancer, though direct evidence is sparse.
Bisphenol A
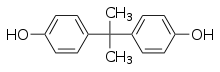
Bisphenol A (BPA) is a chemical compound used in the production of plastics found in numerous commercial products, including laptops, baby bottles, food containers, water main pipes, and laboratory and hospital equipment. BPA was first produced in 1891, but its estrogenic properties went undiscovered until the mid-1930s. Today it is considered a xenoestrogen, and it functions as an endocrine disruptor that interferes with hormones in the body and disrupts the normal functioning of the endocrine system. At very low levels the FDA has long considered BPA in food to be safe, but this has been challenged over the years as more information is discovered regarding the effects of the chemical.[85]
Rats exposed prenatally to environmentally relevant doses of BPA show an increased number of intraductal hyperplasias (precancerous lesions) in mammary glands that appear during adulthood, while high doses induce the development of carcinomas in breast tissue. Animals exposed to BPA during fetal life develop palpable tumors, and all studies show an increased susceptibility to mammary gland neoplasia that manifests during adulthood. Exposure of mouse dams to environmentally relevant levels of BPA during organogenesis results in considerable alterations in the mammary gland. It was concluded that perinatal exposure to low doses of BPA results in altered mammary gland morphogenesis, induction of precancerous lesions, and carcinoma in situ.[84]
A study sought to determine whether early exposure to BPA could accelerate mammary carcinogenesis in a dimethylbenzanthracene (DMBA) model of rodent mammary cancer. In the study, scientists exposed neonatal/prebubertal rats to BPA via lactation from nursing dams treated orally with 0, 25, and 250 µg BPA/kg body weight/day. For tumorigenesis studies, female offspring were exposed to 30 mg DMBA/kg body weight at 50 days of age. DMBA induces mammary tumors and allows chemicals that predispose for mammary cancer to increase the number of mammary adenocarcinomas. The results of the study showed that female rats in the control, BPA 25, and BPA 250 groups administered DMBA exhibited a BPA dose-dependent increase in mammary tumors. The groups had 2.84, 3.82, and 5.00 mammary tumors per rat respectively. Treatment with BPA also reduced tumor latency, with the median tumor latency of 65, 53, and 56.5 days for 0, BPA 25, and BPA 250 groups respectively. Maternal exposure to BPA during lactation decreased time to first tumor latency and increased the number of DMBA-induced mammary tumors in female offspring. If these effects found in rodents carry over to humans, even minimal exposure to BPA could cause an increased risk for breast cancer.[86]
The elevated incidence of breast cancer in women has been associated with prolonged exposure to high levels of estrogens. Xenoestrogens, such as BPA have the capacity to perturb normal hormonal actions. This study provides evidence of the estrogenic effects of BPA. In this study the human breast epithelial cells MCF-10F were treated with 10-3 M, 10-4 M, 10-5 M and 10-6 M BPA continuously for two weeks. The cells treated with 10-3 M BPA died on the second day of treatment. The concentration of 10-4 M BPA was also toxic for the breast epithelial cells, and they died on the fourth day of treatment. This data indicated that these concentrations of BPA are toxic for MCF-10F cells. After the two-week observation period it was seen that the cells formed a high percentage of duct like structures in collagen. MCF-10F cells treated with 10-5 M and 10-6 M BPA formed a high percentage of solid masses, 27% and 20% respectively. This data indicates that BPA is able to induce neoplastic transformation of human breast epithelial cells. Epigenetic changes are involved in the early stages of cancer initiation by altering ductulogenesis. BPA was able to induce transformation of human breast MCF-10F epithelial cells. After treatment with BPA, the cells produced fewer collagen tubules and more solid masses.[87]
Consumer groups recommend that people wishing to lower their exposure to bisphenol A avoid canned food and polycarbonate plastic containers (which shares resin identification code 7 with many other plastics) unless the packaging indicates the plastic is bisphenol A-free.[88] The National Toxicology Panel recommends avoiding microwaving food in plastic containers, putting plastics in the dishwasher, or using harsh detergents on plastics, to avoid leaching.[89]
Aromatic Amines
Aromatic amines are chemicals that are produced when products such as dyes, polyurethane products, and certain pesticides are made. They are also found in cigarette smoke, fuel exhaust, and in over cooked, burned meat. The three types of aromatic amines monocyclic, polycyclic, and heterocyclic have all been found in recent studies of breast health. Monocyclic amines have been found to cause mammary cancer in rats. Studies have shown that women who eat higher amounts of overcooked meat, meaning more exposure to heterocyclic amines, have also been diagnosed with more post-menopausal breast cancer. Heterocyclic amines also have the ability to copy estrogen and in laboratory studies have been found to encourage the growth of cancerous tumors on human tissue.[90]
Benzene
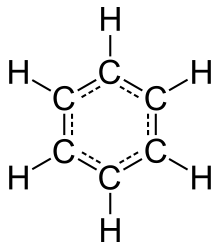
Benzene is a petrochemical solvent. Benzene exposure mostly originates from air pollution resulting from industrial burning, exhaust and gas fumes, as well as cigarette smoke. Petroleum, its distillates such as gasoline, auto and truck exhausts also contain benzene. The International Agency for Research on Cancer and the National Toxicology Program have labeled benzene as a definite human carcinogen. Multiple studies point to a correlation between benzene exposure and breast cancer risk. Laboratory studies on mice have shown that a high level of benzene exposure can lead to mammary cancer.[90]
DDT
Although the pesticide DDT was banned over 20 years ago, studies have shown that there are still trace amounts found in certain agricultural products, as well as in human and animal milk.[91] While individual studies have come to conflicting conclusions, the most recent reviews of all the evidence conclude that exposure to DDT before puberty increases the risk of breast cancer later in life.[92][93]
Ethylene oxide
Ethylene oxide is a chemical that can be found in some personal care products, mainly in the form of fragrance. It is also used for the sterilization of various medical objects. The National Toxicology Program has labeled ethylene oxide as a definite human and animal carcinogen. A study done by the National Institute for Occupational Safety and Health including 7,576 women found a direct correlation between breast cancer rates and exposure to ethylene oxide during medical sterilization processes. Also, human breast cells put into contact with small amounts of ethylene oxide in a laboratory can lead to DNA damage of the breast tissue.[90]
Polycyclic aromatic hydrocarbons
Polycyclic aromatic hydrocarbons are chemical products of combustion from coal burners, fuel, cigarette smoke, and various other sources. PAH's are often found in the air and are breathed into the body. PAH's bioaccumulate easily and can copy the estrogen hormone. PAH's can also be genotoxic, meaning they have the ability to harm DNA.[90]
Vinyl chloride
Vinyl chloride is produced when PVC or polyvinyl chloride is made. PVC is found in plastic packaging, outerwear, plastic toys and other plastic products. Vinyl chloride can be found in cigarette smoke and the air around garbage and land fills. It can also be found in the wastewater when PVC is made. The National Toxicology Program and the International Agency for Research on Cancer have both labeled vinyl chloride as a definite human carcinogen.[90]
Tobacco
Until recently, most studies had not found an increased risk of breast cancer from active tobacco smoking. Beginning in the mid-1990s, a number of studies suggested an increased risk of breast cancer in both active smokers and those exposed to secondhand smoke compared to women who reported no exposure to secondhand smoke.[94] By 2005 enough evidence had accumulated for the [California Environmental Protection Agency] to conclude that breathing secondhand smoke causes breast cancer in younger, primarily premenopausal women.[95] The Agency concluded that the risk was increased by 70%, based on epidemiological studies and the fact that there are many mammary carcinogens in secondhand smoke. The following year (2006) the US Surgeon General[96] identified the same risk increase and concluded that the evidence is "suggestive," one step below causal. There is some evidence that exposure to tobacco smoke is most problematic between puberty and first childbirth. The reason is that breast tissue appears most sensitive to chemical carcinogens breast cells not fully differentiated until lactation.[97] The likely reason that the older studies of active smoking did not detect risks associated with smoking was that they compared active smokers to all nonsmokers (which includes many passive smokers). The newer studies, which exclude passive smokers from the control group, generally show elevated risks associated with active as well as passive smoking.
Passive smoking
Breathing secondhand smoke increases breast cancer risk by 70% in younger, primarily pre-menopausal women. The California Environmental Protection Agency has concluded that passive smoking causes breast cancer[95] and the US Surgeon General[96] has concluded that the evidence is suggestive, one step below causal. There is some evidence that exposure to tobacco smoke is most problematic between puberty and first childbirth. The reason that breast tissue appears most sensitive to chemical carcinogens in this phase is that breast cells are not fully differentiated until lactation.[97]
Radiation
Women who have received high-dose ionizing radiation to the chest (for example, as treatments for other cancers) have a relative risk of breast cancer between 2.1 and 4.0.[94] The risk increases with increased dose. In addition, the risk is higher in women irradiated before age 30, when there is still breast development.[63]
Dioxins
Dioxins (most notably the polychlorinated dibenzodioxins) are chemicals that are produced when chlorinated products are burned, such as polyvinyl chloride (PVC). This occurs when chlorinated products are used in certain manufacturing industries. Dioxins are also added to the air when gasoline and diesel fuels break down. Dioxins are able to bioaccumulate, meaning that they settle and stay in human and animal fat for long periods of time. There are many different types of dioxins and only a few of them have been labeled by the Environmental Protection Agency as definite human carcinogens and endocrine hormone disruptors. Although dioxins are floating in the air, they eventually settle on plants and other vegetation surfaces. These plants and vegetation are them eaten by cows and other animals. Humans end up eating the produce, milk, eggs, and meat produced by these animals that have consumed dioxin covered vegetation. Dioxins are more harmful when ingested this way. Multiple studies have led to the idea that increased dioxin levels can increase one's risk for breast cancer. A study done in 1976 after a chemical plant explosion in Seveso, Italy concluded that high dioxin level exposure in a woman's body correlated with a more than double chance of developing breast cancer.[90]
Light at night and disturbance of circadian rhythm
In 1978 Cohen et al. proposed that reduced production of the hormone melatonin might increase the risk of breast cancer and citing "environmental lighting" as a possible causal factor.[98] Researchers at the National Cancer Institute (NCI) and National Institute of Environmental Health Sciences conducted a study in 2005 that suggests that artificial light during the night can be a factor for breast cancer by disrupting melatonin levels.[99] According to a research in 2008, a reduced melatonin level in postmenopausal women is linked to a higher risk of breast cancer.[100]
In 2007, "shiftwork that involves circadian disruption" was listed as a probable carcinogen by the World Health Organization's International Agency for Research on Cancer. (IARC Press release No. 180).[101] Multiple studies have documented a link between night shift work and the increased incidence of breast cancer.[102][103][104][105] A review of current knowledge of the health consequences of exposure to artificial light at night including the increased incidence of breast cancer and an explanation of the causal mechanisms has been published in the Journal of Pineal Research in 2007.[106]
Ethnicity-related and socioeconomic factors
Incidence and mortality vary with ethnic background and social status. Incidence rises with improving economic situation, while mortality is tied to low economic status. In the US incidence is significantly lower and mortality higher among black women and this difference appears to persist even after adjustment for economic status. It is currently unclear if significant ethnic differences in incidence and mortality persist after adjustment for economic status between women of white, Hispanic and Asian origin in the US.[107]
Several studies have found that black women in the U.S. are more likely to die from breast cancer even though white women are more likely to be diagnosed with the disease. Even after diagnosis, black women are less likely to get treatment compared to white women.[108][109][110] Scholars have advanced several theories for the disparities, including inadequate access to screening, reduced availability of the most advanced surgical and medical techniques, or some biological characteristic of the disease in the African American population.[111] Some studies suggest that the racial disparity in breast cancer outcomes may reflect cultural biases more than biological disease differences.[112] However, the lack of diversity in clinical trials for breast cancer treatment may contribute to these disparities, with recent research indicating that black women are more likely to have estrogen receptor negative breast cancers, which are not responsive to hormone treatments that are effective for most white women.[113] Research is currently ongoing to define the contribution of both biological and cultural factors.[109][114]
Part of the differences in incidence that is attributable to race and economic status may be explained by past use of hormone replacement therapy[115]
Factors with inconclusive research
Tea
One research published in 2009 has shown that moderate green tea or black tea consumption (three or more cups per day) can reduce breast cancer risk by 37% in women younger than 50 years old, comparing with women who drank no tea at all. But no association was found for overall women.[116] However that study has been criticized for inaccuracy[117] and another study found no substantial association between breast cancer and tea consumption in the overall, but found a weak inverse association between caffeine-containing beverages and risk of post-menopausal breast cancer.[118]
Specifically about green tea, one study has found significant inverse association between risk of breast cancer and green tea intake in Asian women who were low soy consumers.[119]
1,3-Butadiene
1,3-Butadiene is an environmental factor that can be found in air pollution and can be produced by combustion engines, as well as petroleum refineries. It is found in cigarette smoke and is also used in the making and processing of certain synthetic rubber products and fungicides. The National Toxicology Program has labeled 1,3-Butadiene as definite human carcinogen. The EPA has stated that people are mainly put in contact with this chemical through the means of simple inhalation.[90]
Mammographic density
Mammographic density refers to the relative proportions of radiodense area compared to the radiolucent area on a mammogram, which is basically an x-ray of the breast. The radiodense area on a mammogram is white and is associated with ductal and lobular epithelium, connective tissue and fluid in the breast. The radiolucent area is dark gray or black and is associated with fat in the breast. High mammographic density is associated with a higher risk of developing breast cancer, but the reasons for this link are not certain and are being studied.[120][121] Conversely, patients with very low mammographic breast density were found to hold a poorer prognosis irrespective of age, BMI and menopausal status.[122]
Red No. 3
Red No. 3 is a coloring agent used in some foods. In laboratory tests human breast cell DNA was found positive for damage when put into contact with Red No. 3, which means that it is genotoxic.[123]
Viruses
Several kinds of viruses with oncogenic potential are suspected to play a role or cause breast cancer. Among the three most commonly studied are the human papilloma virus (HPV),[124] mouse mammary tumour virus[125](MMTV) and the Epstein-Barr virus (EBV).[125] A study published in 2011, reviewing 85 original molecular research investigations on the presence of one or more of these three viruses found that only seven of the studies convincingly demonstrated the presence of an oncogenic virus biomarker, while twenty-five of the studies were able to show the absence of the virus studied, and the remaining studies were excluded due to shortcomings. Thus, the data from these investigations do not justify a conclusion as to whether HPV, MMTV, or EBV play a causal role in human breast cancer development.[126]
Humans are not the only mammals susceptible to breast cancer. Some strains of mice, namely the house mouse (Mus domesticus) are prone to breast cancer which is caused by infection with the mouse mammary tumour virus (MMTV or "Bittner virus" for its discoverer John Joseph Bittner), by random insertional mutagenesis. It is the only animal breast cancer with a known etiology.[126] These findings are taken to mean that a viral origin of human breast cancer is at least possible, though there is no definitive evidence to support the claim that MMTV causes human breast cancer. For example, there may be critical differences between cancer pathogenesis in mice and people. A human homologue of the mammary virus has been described in 1971 and linked to human breast cancer in several small epidemiologic studies.[127][128]
Factors with minimal or no impact
There is no significant association between first-trimester abortion and breast cancer risk.[129] There is no scientific evidence to prove that any kind of brassiere can cause cancer.[130][131] The myth that breast cancer is linked with deodorant use has been widely circulated, and appears to originate from a spam email sent in 1999.[132] There is however no evidence to support the existence of such a link.[133] There is no persuasive connection between fertility medications and breast cancer.[134]
History
In past centuries, the development of breast cancer was most commonly seen as divine punishment or a trial. From ancient Greek medicine until the end of the 17th century, the dominant medical explanation was an imbalance of the four humors.[135] By the start of the 18th century, humoralism had generally been rejected. Many other theories were put forward, often related to sexual activity: In 1713, Bernardino Ramazzini said that nuns developed breast cancer at a higher rate than married women because they did not engage in sexual intercourse, and the "unnatural" lack of sexual activity caused instability of the breasts; others countered that the cause was frequently too much sexual activity.[136] Other theories from the 18th century included various sorts of problems with the movement of body fluids, such as lymphatic blockages, curdled breast milk or the transformation of pus left after an infection.[136]
In modern times, women are more likely to blame themselves, perhaps deciding that their diet, childbearing history, decision not to breastfeed, or level of exercise is the cause.[135]
See also
- Carcinogen#IARC classification of carcinogens
- Epidemiology of breast cancer
- Silent Spring Institute
- Xenoestrogen
- Continuous Individualized Risk Index (CIRI)
- Your Disease Risk
- Mouse models of breast cancer metastasis
- Gynecologic cancer disparities in the United States
References
- Institute of Medicine (2012). Breast Cancer and the Environment: A Life Course Approach (Institute of Medicine). Washington, D.C: National Academies Press. pp. 52–53. doi:10.17226/13263. ISBN 978-0-309-22069-9.
- Madigan MP, Ziegler RG, Benichou J, Byrne C, Hoover RN (November 1995). "Proportion of breast cancer cases in the United States explained by well-established risk factors". Journal of the National Cancer Institute. 87 (22): 1681–5. doi:10.1093/jnci/87.22.1681. PMID 7473816.
- Excel chart for Figure 1.1: Breast Cancer (C50), Average Number of New Cases per Year and Age-Specific Incidence Rates, UK, 2006-2008 at Breast cancer - UK incidence statistics Archived 2012-05-14 at the Wayback Machine at Cancer Research UK. Section updated 18/07/11.
- Margolese RG, Fisher B, Hortobagyi GN, Bloomer WD (2000). "118". In Bast RC, Kufe DW, Pollock RE, et al. (eds.). Cancer Medicine (5th ed.). Hamilton, Ontario: B.C. Decker. ISBN 978-1-55009-113-7. Retrieved 27 January 2011.
- Olson JS (2002). Bathsheba's Breast: Women, Cancer and History. Baltimore: The Johns Hopkins University Press. pp. 199–200. ISBN 978-0-8018-6936-5. OCLC 186453370.
- Giordano SH, Cohen DS, Buzdar AU, Perkins G, Hortobagyi GN (July 2004). "Breast carcinoma in men: a population-based study". Cancer. 101 (1): 51–7. doi:10.1002/cncr.20312. PMID 15221988. S2CID 972345.
- Malone KE, Daling JR, Thompson JD, O'Brien CA, Francisco LV, Ostrander EA (March 1998). "BRCA1 mutations and breast cancer in the general population: analyses in women before age 35 years and in women before age 45 years with first-degree family history". JAMA. 279 (12): 922–9. doi:10.1001/jama.279.12.922. PMID 9544766.
- Auriol E, Billard LM, Magdinier F, Dante R (2005). "Specific binding of the methyl binding domain protein 2 at the BRCA1-NBR2 locus". Nucleic Acids Research. 33 (13): 4243–54. doi:10.1093/nar/gki729. PMC 1181861. PMID 16052033.
- Lacroix M, Leclercq G (February 2005). "The "portrait" of hereditary breast cancer". Breast Cancer Research and Treatment. 89 (3): 297–304. doi:10.1007/s10549-004-2172-4. PMID 15754129. S2CID 23327569.
- Zhang J, Liu X, Datta A, Govindarajan K, Tam WL, Han J, et al. (August 2009). "RCP is a human breast cancer-promoting gene with Ras-activating function". The Journal of Clinical Investigation (Free full text). 119 (8): 2171–83. doi:10.1172/JCI37622. PMC 2719918. PMID 19620787.
- "Breast cancer gene 'could reduce risk'". The Daily Telegraph. London. 2009-03-30. Archived from the original on 2009-04-03. Retrieved 2010-05-22.
- Meindl A, Hellebrand H, Wiek C, Erven V, Wappenschmidt B, Niederacher D, et al. (May 2010). "Germline mutations in breast and ovarian cancer pedigrees establish RAD51C as a human cancer susceptibility gene". Nature Genetics. 42 (5): 410–4. doi:10.1038/ng.569. PMID 20400964. S2CID 23842635.
- Allen NE, Beral V, Casabonne D, Kan SW, Reeves GK, Brown A, Green J (March 2009). "Moderate alcohol intake and cancer incidence in women". Journal of the National Cancer Institute. 101 (5): 296–305. doi:10.1093/jnci/djn514. PMID 19244173.
- Thun MJ, Peto R, Lopez AD, Monaco JH, Henley SJ, Heath CW, et al. (December 1997). "Alcohol consumption and mortality among middle-aged and elderly U.S. adults". The New England Journal of Medicine. 337 (24): 1705–14. doi:10.1056/NEJM199712113372401. PMID 9392695.
- Fuchs CS, Stampfer MJ, Colditz GA, Giovannucci EL, Manson JE, Kawachi I, et al. (May 1995). "Alcohol consumption and mortality among women". The New England Journal of Medicine. 332 (19): 1245–50. doi:10.1056/NEJM199505113321901. PMID 7708067.
- Mu L, Mukamal KJ (February 2016). "Alcohol consumption and rates of cancer screening: Is cancer risk overestimated?". Cancer Causes & Control. 27 (2): 281–9. doi:10.1007/s10552-015-0692-3. PMID 26590914. S2CID 366681.
- Land SR, Liu Q, Wickerham DL, Costantino JP, Ganz PA (May 2014). "Cigarette smoking, physical activity, and alcohol consumption as predictors of cancer incidence among women at high risk of breast cancer in the NSABP P-1 trial". Cancer Epidemiology, Biomarkers & Prevention. 23 (5): 823–32. doi:10.1158/1055-9965.EPI-13-1105-T. PMC 4011972. PMID 24569437.
- Gou YJ, Xie DX, Yang KH, Liu YL, Zhang JH, Li B, He XD (2013). "Alcohol Consumption and Breast Cancer Survival: A Meta- analysis of Cohort Studies". Asian Pacific Journal of Cancer Prevention. 14 (8): 4785–90. doi:10.7314/APJCP.2013.14.8.4785. PMID 24083744.
- Lowry SJ, Kapphahn K, Chlebowski R, Li CI (2016). "Alcohol Use and Breast Cancer Survival among Participants in the Women's Health Initiative". Cancer Epidemiology, Biomarkers & Prevention. 25 (8): 1268–73. doi:10.1158/1055-9965.EPI-16-0151. PMC 4970888. PMID 27197280.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Din N, Allen IE, Satariano WA, Demb J, Braithwaite D (July 2016). "Alcohol consumption and mortality after breast cancer diagnosis: The health and functioning in women study". Breast Disease. 36 (2–3): 77–89. doi:10.3233/bd-150202. PMID 27662274.
- Newcomb Polly A, Kampman E, Trentham-Dietz A, Egan KM, Titus LJ, Baron JA, Hampton JM, Passarelli MN, Willett WC (2013). "Alcohol Consumption Before and After Breast Cancer Diagnosis: Associations With Survival From Breast Cancer, Cardiovascular Disease, and Other Causes". Journal of Clinical Oncology. 31 (16): 1939–1946. doi:10.1200/JCO.2012.46.5765. PMC 3661933. PMID 23569314.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Chlebowski RT, Blackburn GL, Thomson CA, Nixon DW, Shapiro A, Hoy MK, et al. (December 2006). "Dietary fat reduction and breast cancer outcome: interim efficacy results from the Women's Intervention Nutrition Study". Journal of the National Cancer Institute. 98 (24): 1767–76. doi:10.1093/jnci/djj494. PMID 17179478.
- Hunter DJ, Spiegelman D, Adami HO, Beeson L, van den Brandt PA, Folsom AR, et al. (February 1996). "Cohort studies of fat intake and the risk of breast cancer--a pooled analysis". The New England Journal of Medicine. 334 (6): 356–61. doi:10.1056/NEJM199602083340603. PMID 8538706.
- Prentice RL, Caan B, Chlebowski RT, Patterson R, Kuller LH, Ockene JK, et al. (February 2006). "Low-fat dietary pattern and risk of invasive breast cancer: the Women's Health Initiative Randomized Controlled Dietary Modification Trial". JAMA. 295 (6): 629–42. doi:10.1001/jama.295.6.629. PMID 16467232.
- Pala V, Krogh V, Muti P, Chajès V, Riboli E, Micheli A, et al. (July 2001). "Erythrocyte membrane fatty acids and subsequent breast cancer: a prospective Italian study". Journal of the National Cancer Institute. 93 (14): 1088–95. doi:10.1093/jnci/93.14.1088. PMID 11459870.
- Sonestedt E, Ericson U, Gullberg B, Skog K, Olsson H, Wirfält E (October 2008). "Do both heterocyclic amines and omega-6 polyunsaturated fatty acids contribute to the incidence of breast cancer in postmenopausal women of the Malmö diet and cancer cohort?". International Journal of Cancer. 123 (7): 1637–43. doi:10.1002/ijc.23394. PMID 18636564. S2CID 40268547.
- White AJ, Bradshaw PT, Herring AH, Teitelbaum SL, Beyea J, Stellman SD, et al. (April 2016). "Exposure to multiple sources of polycyclic aromatic hydrocarbons and breast cancer incidence". Environment International. 89–90: 185–92. doi:10.1016/j.envint.2016.02.009. PMC 4818720. PMID 26878284.
- Rice S, Whitehead SA (December 2006). "Phytoestrogens and breast cancer--promoters or protectors?". Endocrine-Related Cancer. 13 (4): 995–1015. doi:10.1677/erc.1.01159. PMID 17158751.
- Messina M, McCaskill-Stevens W, Lampe JW (September 2006). "Addressing the soy and breast cancer relationship: review, commentary, and workshop proceedings". Journal of the National Cancer Institute. 98 (18): 1275–84. doi:10.1093/jnci/djj356. PMID 16985246.
- Shin MH, Holmes MD, Hankinson SE, Wu K, Colditz GA, Willett WC (September 2002). "Intake of dairy products, calcium, and vitamin d and risk of breast cancer". Journal of the National Cancer Institute. 94 (17): 1301–11. doi:10.1093/jnci/94.17.1301. PMID 12208895.
- McCullough ML, Rodriguez C, Diver WR, Feigelson HS, Stevens VL, Thun MJ, et al. (December 2005). "Dairy, calcium, and vitamin D intake and postmenopausal breast cancer risk in the Cancer Prevention Study II Nutrition Cohort". Cancer Epidemiology, Biomarkers & Prevention. 14 (12): 2898–904. doi:10.1158/1055-9965.EPI-05-0611. PMID 16365007. S2CID 1714581.
- Lin J, Manson JE, Lee IM, Cook NR, Buring JE, Zhang SM (May 2007). "Intakes of calcium and vitamin D and breast cancer risk in women". Archives of Internal Medicine. 167 (10): 1050–9. doi:10.1001/archinte.167.10.1050. PMID 17533208.
- Ann Nutr metab 2007; 51:139-45
- Knekt P, Järvinen R, Seppänen R, Pukkala E, Aromaa A (March 1996). "Intake of dairy products and the risk of breast cancer". British Journal of Cancer. 73 (5): 687–91. doi:10.1038/bjc.1996.119. PMC 2074353. PMID 8605108.
- "Aggressive Breast Tumors Linked to Vitamin D Deficiency".
- Terry P, Wolk A, Persson I, Magnusson C (June 2001). "Brassica vegetables and breast cancer risk". JAMA. 285 (23): 2975–7. doi:10.1001/jama.285.23.2975. PMID 11410091.
- Nelson NJ (April 2006). "Migrant studies aid the search for factors linked to breast cancer risk". Journal of the National Cancer Institute. 98 (7): 436–8. doi:10.1093/jnci/djj147. PMID 16595777.
- Zhang M, Huang J, Xie X, Holman CD (March 2009). "Dietary intakes of mushrooms and green tea combine to reduce the risk of breast cancer in Chinese women". International Journal of Cancer. International Journal of Cancer (Online). 124 (6): 1404–8. doi:10.1002/ijc.24047. PMID 19048616. S2CID 22781113.
- Hong SA, Kim K, Nam SJ, Kong G, Kim MK (February 2008). "A case-control study on the dietary intake of mushrooms and breast cancer risk among Korean women". International Journal of Cancer. 122 (4): 919–23. doi:10.1002/ijc.23134. PMID 17943725. S2CID 22185468.
- Venturi S (October 2001). "Is there a role for iodine in breast diseases?". Breast. 10 (5): 379–82. doi:10.1054/brst.2000.0267. PMID 14965610.
- Aceves C, Anguiano B, Delgado G (April 2005). "Is iodine a gatekeeper of the integrity of the mammary gland?". Journal of Mammary Gland Biology and Neoplasia. 10 (2): 189–96. doi:10.1007/s10911-005-5401-5. PMID 16025225. S2CID 16838840.
- Stoddard FR, Brooks AD, Eskin BA, Johannes GJ (July 2008). "Iodine alters gene expression in the MCF7 breast cancer cell line: evidence for an anti-estrogen effect of iodine". International Journal of Medical Sciences. 5 (4): 189–96. doi:10.7150/ijms.5.189. PMC 2452979. PMID 18645607.
- BBC report Weight link to breast cancer risk
- Nelson Mya (3 November 2011) New Research: Getting Up From Your Desk Can Put the "Breaks" on Cancer. Experts Urge Americans to Rethink Outdated Notions of Physical Activity American Institute for Cancer Research, Press release from AICR 2011 annual meeting, Retrieved 7 December 2011
- Ligibel J (October 2011). "Obesity and breast cancer". Oncology. 25 (11): 994–1000. PMID 22106549.
- Protani M, Coory M, Martin JH (October 2010). "Effect of obesity on survival of women with breast cancer: systematic review and meta-analysis" (PDF). Breast Cancer Research and Treatment. 123 (3): 627–35. doi:10.1007/s10549-010-0990-0. PMID 20571870. S2CID 22281435.
- Kroenke CH, Chen WY, Rosner B, Holmes MD (March 2005). "Weight, weight gain, and survival after breast cancer diagnosis". Journal of Clinical Oncology. 23 (7): 1370–8. doi:10.1200/JCO.2005.01.079. PMID 15684320.
- Caan BJ, Emond JA, Natarajan L, Castillo A, Gunderson EP, Habel L, et al. (September 2006). "Post-diagnosis weight gain and breast cancer recurrence in women with early stage breast cancer". Breast Cancer Research and Treatment. 99 (1): 47–57. doi:10.1007/s10549-006-9179-y. PMID 16541317. S2CID 23361085.
- Holmes MD, Chen WY, Feskanich D, Kroenke CH, Colditz GA (May 2005). "Physical activity and survival after breast cancer diagnosis". JAMA. 293 (20): 2479–86. doi:10.1001/jama.293.20.2479. PMID 15914748.
- Komen, Susan G. "Cure, Facts for Life Racial and Ethnic Differences" (PDF).
- Maruthur NM, Bolen S, Brancati FL, Clark JM (May 2009). "Obesity and mammography: a systematic review and meta-analysis". Journal of General Internal Medicine. 24 (5): 665–77. doi:10.1007/s11606-009-0939-3. PMC 2669867. PMID 19277790.
- Wee CC, McCarthy EP, Davis RB, Phillips RS (April 2004). "Obesity and breast cancer screening". Journal of General Internal Medicine. 19 (4): 324–31. doi:10.1111/j.1525-1497.2004.30354.x. PMC 1492197. PMID 15061741.
- Feldstein AC, Perrin N, Rosales AG, Schneider J, Rix MM, Glasgow RE (January 2011). "Patient Barriers to Mammography Identified During a Reminder Program". Journal of Women's Health. 20 (3): 421–428. doi:10.1089/jwh.2010.2195. PMC 3117308. PMID 21275649.
- Kerlikowske K, Walker R, Miglioretti DL, Desai A, Ballard-Barbash R, Buist DS (December 2008). "Obesity, mammography use and accuracy, and advanced breast cancer risk". Journal of the National Cancer Institute. 100 (23): 1724–33. doi:10.1093/jnci/djn388. PMC 2734114. PMID 19033562.
- Yager JD, Davidson NE (January 2006). "Estrogen carcinogenesis in breast cancer". The New England Journal of Medicine. 354 (3): 270–82. doi:10.1056/NEJMra050776. PMID 16421368.
- "What Are the Risk Factors for Breast Cancer?". American Cancer Society. 3 October 2006. Retrieved 30 March 2006.
- Gunter MJ, Hoover DR, Yu H, Wassertheil-Smoller S, Rohan TE, Manson JE, et al. (January 2009). "Insulin, insulin-like growth factor-I, and risk of breast cancer in postmenopausal women". Journal of the National Cancer Institute. 101 (1): 48–60. doi:10.1093/jnci/djn415. PMC 2639294. PMID 19116382.
- "Title". MedScape.
- McTiernan A, Thomas DB (September 1986). "Evidence for a protective effect of lactation on risk of breast cancer in young women. Results from a case-control study". American Journal of Epidemiology. 124 (3): 353–8. doi:10.1093/oxfordjournals.aje.a114405. PMID 3740036.
- Byers T, Graham S, Rzepka T, Marshall J (May 1985). "Lactation and breast cancer. Evidence for a negative association in premenopausal women". American Journal of Epidemiology. 121 (5): 664–74. doi:10.1093/aje/121.5.664. PMID 4014158.
- Collaborative Group on Hormonal Factors in Breast Cancer. (October 1997). "Breast cancer and hormone replacement therapy: collaborative reanalysis of data from 51 epidemiological studies of 52,705 women with breast cancer and 108,411 women without breast cancer. Collaborative Group on Hormonal Factors in Breast Cancer". Lancet. 350 (9084): 1047–59. doi:10.1016/S0140-6736(97)08233-0. PMID 10213546. S2CID 54389746.
- Newcomb PA, Storer BE, Longnecker MP, Mittendorf R, Greenberg ER, Clapp RW, et al. (January 1994). "Lactation and a reduced risk of premenopausal breast cancer". The New England Journal of Medicine. 330 (2): 81–7. doi:10.1056/NEJM199401133300201. PMID 8259187.
- Mitchell RS, Kumar V, Abbas AK, Fausto N, eds. (2007). "Chapter 19". Robbins Basic Pathology (8th ed.). Philadelphia: Saunders. ISBN 978-1-4160-2973-1.
- Collaborative Group on Hormonal Factors in Breast Cancer (June 1996). "Breast cancer and hormonal contraceptives: collaborative reanalysis of individual data on 53 297 women with breast cancer and 100 239 women without breast cancer from 54 epidemiological studies". Lancet. 347 (9017): 1713–27. doi:10.1016/S0140-6736(96)90806-5. PMID 8656904. S2CID 36136756.
- Collaborative Group on Hormonal Factors in Breast Cancer (September 1996). "Breast cancer and hormonal contraceptives: further results. Collaborative Group on Hormonal Factors in Breast Cancer". Contraception. 54 (3 Suppl): 1S–106S. doi:10.1016/s0010-7824(15)30002-0. PMID 8899264.
- National Cancer Institute (2006-08-03). "Hormone Therapy". Genetics of Breast and Ovarian Cancer. Retrieved 2006-08-12.
- World Health Organization International Agency for Research on Cancer (1999). "Hormonal Contraception and Post-menopausal Hormonal Therapy" (PDF). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. 72. Retrieved 12 March 2011.
- Stahlberg C, Pederson AT, Lynge E, Ottesen B (July 2003). "Hormone replacement therapy and risk of breast cancer: the role of progestins". Acta Obstetricia et Gynecologica Scandinavica. 82 (7): 335–44. doi:10.1034/j.1600-0412.2003.00551.x. PMID 12790856. S2CID 33887019.
- Heiss G, Wallace R, Anderson GL, Aragaki A, Beresford SA, Brzyski R, et al. (March 2008). "Health risks and benefits 3 years after stopping randomized treatment with estrogen and progestin". JAMA. 299 (9): 1036–45. doi:10.1001/jama.299.9.1036. PMID 18319414.
- "Guide to Clinical Preventive Services, Third Edition: Periodic Updates, 2000–2003". Agency for Healthcare Research and Quality. US Preventive Services Task Force. Retrieved 2007-10-07.
- U.S. Preventive Services Task Force (July 2002). "Chemoprevention of breast cancer: recommendations and rationale". Annals of Internal Medicine. 137 (1): 56–8. doi:10.7326/0003-4819-137-1-200207020-00016. PMID 12093249. S2CID 10445937.
- Kinsinger LS, Harris R, Woolf SH, Sox HC, Lohr KN (July 2002). "Chemoprevention of breast cancer: a summary of the evidence for the U.S. Preventive Services Task Force". Annals of Internal Medicine. 137 (1): 59–69. doi:10.7326/0003-4819-137-1-200207020-00017. PMID 12093250. S2CID 8608388.
- Cummings SR, Eckert S, Krueger KA, Grady D, Powles TJ, Cauley JA, et al. (June 1999). "The effect of raloxifene on risk of breast cancer in postmenopausal women: results from the MORE randomized trial. Multiple Outcomes of Raloxifene Evaluation". JAMA. 281 (23): 2189–97. doi:10.1001/jama.281.23.2189. PMID 10376571.
- Fisher B, Costantino JP, Wickerham DL, Cecchini RS, Cronin WM, Robidoux A, et al. (November 2005). "Tamoxifen for the prevention of breast cancer: current status of the National Surgical Adjuvant Breast and Bowel Project P-1 study". Journal of the National Cancer Institute. 97 (22): 1652–62. doi:10.1093/jnci/dji372. PMID 16288118.
- Fisher B, Costantino JP, Wickerham DL, Redmond CK, Kavanah M, Cronin WM, et al. (September 1998). "Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study". Journal of the National Cancer Institute. 90 (18): 1371–88. doi:10.1093/jnci/90.18.1371. PMID 9747868.
- Veronesi U, Maisonneuve P, Rotmensz N, Bonanni B, Boyle P, Viale G, et al. (May 2007). "Tamoxifen for the prevention of breast cancer: late results of the Italian Randomized Tamoxifen Prevention Trial among women with hysterectomy". Journal of the National Cancer Institute. 99 (9): 727–37. doi:10.1093/jnci/djk154. PMID 17470740.
- Cuzick J, Forbes JF, Sestak I, Cawthorn S, Hamed H, Holli K, Howell A (February 2007). "Long-term results of tamoxifen prophylaxis for breast cancer--96-month follow-up of the randomized IBIS-I trial". Journal of the National Cancer Institute. 99 (4): 272–82. doi:10.1093/jnci/djk049. PMID 17312304.
- Vogel VG, Costantino JP, Wickerham DL, Cronin WM, Cecchini RS, Atkins JN, et al. (June 2006). "Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 trial". JAMA. 295 (23): 2727–41. doi:10.1001/jama.295.23.joc60074. PMID 16754727.
- Barrett-Connor E, Mosca L, Collins P, Geiger MJ, Grady D, Kornitzer M, et al. (July 2006). "Effects of raloxifene on cardiovascular events and breast cancer in postmenopausal women". The New England Journal of Medicine. 355 (2): 125–37. doi:10.1056/NEJMoa062462. hdl:2437/113085. PMID 16837676.
- AFP.google.com, US approves Lilly's Evista for breast cancer prevention Archived 2011-05-20 at the Wayback Machine
- Palmer JR, Wise LA, Hatch EE, Troisi R, Titus-Ernstoff L, Strohsnitter W, et al. (August 2006). "Prenatal diethylstilbestrol exposure and risk of breast cancer". Cancer Epidemiology, Biomarkers & Prevention. 15 (8): 1509–14. doi:10.1158/1055-9965.EPI-06-0109. PMID 16896041. S2CID 7225182.
- Brody JG, Moysich KB, Humblet O, Attfield KR, Beehler GP, Rudel RA (June 2007). "Environmental pollutants and breast cancer: epidemiologic studies". Cancer. 109 (12 Suppl): 2667–711. doi:10.1002/cncr.22655. PMID 17503436. S2CID 2620028.
- Vogel SA (November 2009). "The politics of plastics: the making and unmaking of bisphenol a "safety"". American Journal of Public Health. 99 Suppl 3 (S3): S559-66. doi:10.2105/AJPH.2008.159228. PMC 2774166. PMID 19890158.
- Soto AM, Sonnenschein C (July 2010). "Environmental causes of cancer: endocrine disruptors as carcinogens". Nature Reviews. Endocrinology. 6 (7): 363–70. doi:10.1038/nrendo.2010.87. PMC 3933258. PMID 20498677.
- Vogel SA (November 2009). "The politics of plastics: the making and unmaking of bisphenol a "safety"". American Journal of Public Health. 99 Suppl 3 (3): S559-66. doi:10.2105/AJPH.2008.159228. PMC 2774166. PMID 19890158.
- Jenkins S, Raghuraman N, Eltoum I, Carpenter M, Russo J, Lamartiniere CA (June 2009). "Oral exposure to bisphenol a increases dimethylbenzanthracene-induced mammary cancer in rats". Environmental Health Perspectives. 117 (6): 910–5. doi:10.1289/ehp.11751. PMC 2702405. PMID 19590682.
- Fernandez SV, Russo J (January 2010). "Estrogen and xenoestrogens in breast cancer". Toxicologic Pathology. 38 (1): 110–22. doi:10.1177/0192623309354108. PMC 2907875. PMID 19933552.
- "Living on Earth: War of the Sciences".
- FDA Weighs Safety Of Bisphenol A
- State of the Evidence, The Connection Between Breast Cancer and the Environment, 2008
- Sonawane BR (September 1995). "Chemical contaminants in human milk: an overview". Environmental Health Perspectives. 103 Suppl 6 (S6): 197–205. doi:10.1289/ehp.95103s6197. JSTOR 3432374. PMC 1518901. PMID 8549474.
- Eskenazi B, Chevrier J, Rosas LG, Anderson HA, Bornman MS, Bouwman H, et al. (September 2009). "The Pine River statement: human health consequences of DDT use". Environmental Health Perspectives. 117 (9): 1359–67. doi:10.1289/ehp.11748. PMC 2737010. PMID 19750098.
- Clapp RW, Jacobs MM, Loechler EL (2008). "Environmental and occupational causes of cancer: new evidence 2005-2007". Reviews on Environmental Health. 23 (1): 1–37. doi:10.1515/REVEH.2008.23.1.1. PMC 2791455. PMID 18557596.
- ACS (2005). "Breast Cancer Facts & Figures 2005–2006" (PDF). Archived from the original (PDF) on 2007-06-13. Retrieved 2007-04-26.
- California Environmental Protection Agency: Air Resources Board, "Proposed Identification of Environmental Tobacco Smoke as a Toxic Air Contaminant" (June 24, 2005); on January 26, 2006, the Air Resources Board, following a lengthy review and public outreach process, determined ETS to be a Toxic Air Contaminant (TAC).
- U.S. Department of Health and Human Services. "The Health Consequences of Involuntary Exposure to Tobacco Smoke: A Report of the Surgeon General", 2006
- J. Russo, I. Russo. "Molecular Basis of Breast Cancer: Prevention and Treatment," Springer, 2003
- Cohen M, Lippman M, Chabner B (October 1978). "Role of pineal gland in aetiology and treatment of breast cancer". Lancet. 2 (8094): 814–6. doi:10.1016/S0140-6736(78)92591-6. PMID 81365. S2CID 10052553.
- Blask DE, Brainard GC, Dauchy RT, Hanifin JP, Davidson LK, Krause JA, et al. (December 2005). "Melatonin-depleted blood from premenopausal women exposed to light at night stimulates growth of human breast cancer xenografts in nude rats". Cancer Research. 65 (23): 11174–84. doi:10.1158/0008-5472.CAN-05-1945. PMID 16322268.
- Gunter MJ, Hoover DR, Yu H, Wassertheil-Smoller S, Rohan TE, Manson JE, et al. (2009). "Insulin, insulin-like growth factor-I and risk of breast cancer in postmenopausal women". J Natl Cancer Inst. 101 (1): 48–60. doi:10.1093/jnci/djn415. PMC 2639294. PMID 19116382. Lay summary – Science Daily.
{{cite journal}}: Cite uses deprecated parameter|lay-url=(help) - "Press release No. 180". International Agency for Research on Cancer. 2007-12-05. Archived from the original on 2008-04-11.
- Schernhammer ES, Schulmeister K (March 2004). "Melatonin and cancer risk: does light at night compromise physiologic cancer protection by lowering serum melatonin levels?". British Journal of Cancer. 90 (5): 941–3. doi:10.1038/sj.bjc.6601626. PMC 2409637. PMID 14997186.
- Hansen J (January 2001). "Increased breast cancer risk among women who work predominantly at night". Epidemiology. 12 (1): 74–7. doi:10.1097/00001648-200101000-00013. PMID 11138824. S2CID 34390800.
- Hansen J (October 2001). "Light at night, shiftwork, and breast cancer risk". Journal of the National Cancer Institute. 93 (20): 1513–5. doi:10.1093/jnci/93.20.1513. PMID 11604468.
- Schernhammer ES, Laden F, Speizer FE, Willett WC, Hunter DJ, Kawachi I, Colditz GA (October 2001). "Rotating night shifts and risk of breast cancer in women participating in the nurses' health study". Journal of the National Cancer Institute. 93 (20): 1563–8. doi:10.1093/jnci/93.20.1563. PMID 11604480.
- Navara KJ, Nelson RJ (October 2007). "The dark side of light at night: physiological, epidemiological, and ecological consequences" (PDF). Journal of Pineal Research. 43 (3): 215–24. doi:10.1111/j.1600-079X.2007.00473.x. PMID 17803517. S2CID 11860550. Archived from the original (PDF) on 2011-12-14.
- Vainshtein J (July 2008). "Disparities in breast cancer incidence across racial/ethnic strata and socioeconomic status: a systematic review". Journal of the National Medical Association. 100 (7): 833–9. doi:10.1016/S0027-9684(15)31378-X. PMID 18672561.
- Wisconsin Cancer Incidence and Mortality, 2000–2004 Archived 2008-05-30 at the Wayback Machine Wisconsin Department of Health and Family Services
- Tammemagi CM (February 2007). "Racial/ethnic disparities in breast and gynecologic cancer treatment and outcomes". Current Opinion in Obstetrics & Gynecology. 19 (1): 31–6. doi:10.1097/GCO.0b013e3280117cf8. PMID 17218849. S2CID 1445353.
- Hirschman J, Whitman S, Ansell D (April 2007). "The black:white disparity in breast cancer mortality: the example of Chicago". Cancer Causes & Control. 18 (3): 323–33. doi:10.1007/s10552-006-0102-y. PMID 17285262. S2CID 3349878.
- Breast cancer rates differ in races by Amanda Villa Wednesday, October 24, 2007. Badger Herald
- Benjamin M, Reddy S, Brawley OW (March 2003). "Myeloma and race: a review of the literature". Cancer and Metastasis Reviews. 22 (1): 87–93. doi:10.1023/A:1022268103136. PMID 12716040. S2CID 27614206.
- Zuckerman D (2009). "The Ethics of Inclusion and Exclusion in Clinical Trials: Race, Sex, and Age". In Ravitsky V, Fiester A, Caplan A (eds.). The Penn Center Guide to bioethics. Springer. ISBN 978-0-8261-1522-5.
- Demicheli R, Retsky MW, Hrushesky WJ, Baum M, Gukas ID, Jatoi I (November 2007). "Racial disparities in breast cancer outcome: insights into host-tumor interactions". Cancer. 110 (9): 1880–8. doi:10.1002/cncr.22998. PMID 17876835. S2CID 46360199.
- Hausauer AK, Keegan TH, Chang ET, Clarke CA (2007). "Recent breast cancer trends among Asian/Pacific Islander, Hispanic, and African-American women in the US: changes by tumor subtype". Breast Cancer Research. 9 (6): R90. doi:10.1186/bcr1839. PMC 2246193. PMID 18162138.
- Kumar N, Titus-Ernstoff L, Newcomb PA, Trentham-Dietz A, Anic G, Egan KM (January 2009). "Tea consumption and risk of breast cancer". Cancer Epidemiology, Biomarkers & Prevention. 18 (1): 341–5. doi:10.1158/1055-9965.EPI-08-0819. PMC 3156033. PMID 19124518.
- "Breast cancer: weak tea study". January 22, 2009. Retrieved 2009-01-22.
- Ganmaa D, Willett WC, Li TY, Feskanich D, van Dam RM, Lopez-Garcia E, et al. (May 2008). "Coffee, tea, caffeine and risk of breast cancer: a 22-year follow-up". International Journal of Cancer. 122 (9): 2071–6. doi:10.1002/ijc.23336. PMC 4186696. PMID 18183588.
- Wu AH, Yu MC, Tseng CC, Hankin J, Pike MC (September 2003). "Green tea and risk of breast cancer in Asian Americans". International Journal of Cancer. 106 (4): 574–9. doi:10.1002/ijc.11259. PMID 12845655. S2CID 2821903.
- Gierach GL, Lissowska J, Garcia-Closas M, Yang XR, Figueroa JD, Anzick S, et al. (April 19, 2010). "Relationship of mammographic density with breast cancer subtypes".
{{cite journal}}: Cite journal requires|journal=(help)} - "Breast Density Change Linked to Cancer Development in WHI Hormone Replacement Study". Archived from the original on 2010-06-09. Retrieved 2010-04-23.
- Masarwah A, Auvinen P, Sudah M, Rautiainen S, Sutela A, Pelkonen O, et al. (July 2015). "Very low mammographic breast density predicts poorer outcome in patients with invasive breast cancer". European Radiology. 25 (7): 1875–82. doi:10.1007/s00330-015-3626-2. PMID 25735512. S2CID 25084312.
- Dees C, Askari M, Garrett S, Gehrs K, Henley D, Ardies CM (April 1997). "Estrogenic and DNA-damaging activity of Red No. 3 in human breast cancer cells". Environmental Health Perspectives. Environmental Health Perspectives, Vol. 105. 105 Suppl 3 (Suppl 3): 625–32. doi:10.2307/3433381. JSTOR 3433381. PMC 1469907. PMID 9168006.
- Heng B, Glenn WK, Ye Y, Tran B, Delprado W, Lutze-Mann L, et al. (October 2009). "Human papilloma virus is associated with breast cancer". British Journal of Cancer. 101 (8): 1345–50. doi:10.1038/sj.bjc.6605282. PMC 2737128. PMID 19724278.
- Lawson JS, Günzburg WH, Whitaker NJ (June 2006). "Viruses and human breast cancer". Future Microbiology. 1 (1): 33–51. doi:10.2217/17460913.1.1.33. PMID 17661684.
- Joshi D, Buehring GC (August 2012). "Are viruses associated with human breast cancer? Scrutinizing the molecular evidence". Breast Cancer Research and Treatment. 135 (1): 1–15. doi:10.1007/s10549-011-1921-4. PMID 22274134. S2CID 1310301.
- Lawson JS, Tran D, Rawlinson WD (2001). "From Bittner to Barr: a viral, diet and hormone breast cancer aetiology hypothesis". Breast Cancer Research. 3 (2): 81–5. doi:10.1186/bcr275. PMC 138675. PMID 11250750.
- Taneja P, Frazier DP, Kendig RD, Maglic D, Sugiyama T, Kai F, et al. (July 2009). "MMTV mouse models and the diagnostic values of MMTV-like sequences in human breast cancer". Expert Review of Molecular Diagnostics. 9 (5): 423–40. doi:10.1586/erm.09.31. PMC 2759974. PMID 19580428.
- "WHO | Induced abortion does not increase breast cancer risk". who.int. Archived from the original on 2007-12-14. Retrieved 2007-12-24.
- "Bras and breast cancer risk". Cancer Research UK. Archived from the original on April 6, 2012. Retrieved April 6, 2013.
- "Breast cancer". A.D.A.M. Medical Encyclopedia, U.S. National Library of Medicine. November 17, 2012. Retrieved April 6, 2013.
- Gorski D (6 October 2014). "Breast cancer myths: No, antiperspirants do not cause breast cancer". Science-Based Medicine.
- Namer M, Luporsi E, Gligorov J, Lokiec F, Spielmann M (September 2008). "[The use of deodorants/antiperspirants does not constitute a risk factor for breast cancer]" [The use of deodorants/antiperspirants does not constitute a risk factor for breast cancer]. Bulletin du Cancer (Comprehensive literature review) (in French). 95 (9): 871–80. doi:10.1684/bdc.2008.0679 (inactive 31 July 2022). PMID 18829420.
{{cite journal}}: CS1 maint: DOI inactive as of July 2022 (link) - Potashnik G, Lerner-Geva L, Genkin L, Chetrit A, Lunenfeld E, Porath A (May 1999). "Fertility drugs and the risk of breast and ovarian cancers: results of a long-term follow-up study". Fertility and Sterility. 71 (5): 853–9. doi:10.1016/S0015-0282(99)00085-0. PMID 10231045.
- Yalom M (1997). A history of the breast. New York: Alfred A. Knopf. p. 234. ISBN 978-0-679-43459-7.
- Olson JS (2002). Bathsheba's breast: women, cancer & history. Baltimore: The Johns Hopkins University Press. pp. 32–33. ISBN 978-0-8018-6936-5.
External links
- Breast Cancer Myths and Facts
- Breast cancer at Curlie
- GeneReviews/NCBI/NIH/UW entry on BRCA1 and BRCA2 Hereditary Breast/Ovarian Cancer
- Breast cancer database at Silent Spring Institute
- Risk factors for breast cancer at Curlie