Ziresovir
Ziresovir (RO-0529, AK0529) is an antiviral drug which was developed as a treatment for respiratory syncytial virus. It acts as a fusion inhibitor, and has shown good results in Phase II and III clinical trials.[1][2][3]
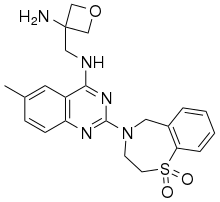 | |
| Clinical data | |
|---|---|
| Trade names | Ziresovir |
| Legal status | |
| Legal status | |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C22H25N5O3S |
| Molar mass | 439.53 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
See also
References
- Zheng X, Gao L, Wang L, Liang C, Wang B, Liu Y, et al. (July 2019). "Discovery of Ziresovir as a Potent, Selective, and Orally Bioavailable Respiratory Syncytial Virus Fusion Protein Inhibitor". Journal of Medicinal Chemistry. 62 (13): 6003–6014. doi:10.1021/acs.jmedchem.9b00654. PMID 31194544.
- Beigel JH, Nam HH, Adams PL, Krafft A, Ince WL, El-Kamary SS, Sims AC (July 2019). "Advances in respiratory virus therapeutics - A meeting report from the 6th isirv Antiviral Group conference". Antiviral Research. 167: 45–67. doi:10.1016/j.antiviral.2019.04.006. PMC 7132446. PMID 30974127.
- Taylor, Nick Paul (7 April 2022). "ArkBio's ex-Roche antiviral hits primary goal in RSV phase 3, sparking race to regulator and wait for more data". Fierce Biotech. Retrieved 17 July 2022.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.