Thyroid hormones
Thyroid hormones are two hormones produced and released by the thyroid gland, namely triiodothyronine (T3) and thyroxine (T4). They are tyrosine-based hormones that are primarily responsible for regulation of metabolism. T3 and T4 are partially composed of iodine. A deficiency of iodine leads to decreased production of T3 and T4, enlarges the thyroid tissue and will cause the disease known as simple goitre.
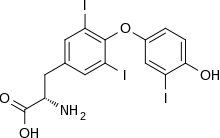
The major form of thyroid hormone in the blood is thyroxine (T4), which has a longer half-life than T3.[2] In humans, the ratio of T4 to T3 released into the blood is approximately 14:1.[3] T4 is converted to the active T3 (three to four times more potent than T4) within cells by deiodinases (5′-deiodinase). These are further processed by decarboxylation and deiodination to produce iodothyronamine (T1a) and thyronamine (T0a). All three isoforms of the deiodinases are selenium-containing enzymes, thus dietary selenium is essential for T3 production.
American chemist Edward Calvin Kendall was responsible for the isolation of thyroxine in 1915.[4] In 2018, levothyroxine, a manufactured form of thyroxine, was the second most commonly prescribed medication in the United States, with more than 105 million prescriptions.[5][6] Levothyroxine is on the World Health Organization's List of Essential Medicines.[7]
Function
The thyroid hormones act on nearly every cell in the body. It acts to increase the basal metabolic rate, affect protein synthesis, help regulate long bone growth (synergy with growth hormone) and neural maturation, and increase the body's sensitivity to catecholamines (such as adrenaline) by permissiveness. The thyroid hormones are essential to proper development and differentiation of all cells of the human body. These hormones also regulate protein, fat, and carbohydrate metabolism, affecting how human cells use energetic compounds. They also stimulate vitamin metabolism. Numerous physiological and pathological stimuli influence thyroid hormone synthesis.
Thyroid hormone leads to heat generation in humans. However, the thyronamines function via some unknown mechanism to inhibit neuronal activity; this plays an important role in the hibernation cycles of mammals and the moulting behaviour of birds. One effect of administering the thyronamines is a severe drop in body temperature.
Medical use
Both T3 and T4 are used to treat thyroid hormone deficiency (hypothyroidism). They are both absorbed well by the stomach, so can be given orally. Levothyroxine is the pharmaceutical name of the manufactured version of T4, which is metabolised more slowly than T3 and hence usually only needs once-daily administration. Natural desiccated thyroid hormones are derived from pig thyroid glands, and are a "natural" hypothyroid treatment containing 20% T3 and traces of T2, T1 and calcitonin. Also available are synthetic combinations of T3/T4 in different ratios (such as liotrix) and pure-T3 medications (INN: liothyronine). Levothyroxine Sodium is usually the first course of treatment tried. Some patients feel they do better on desiccated thyroid hormones; however, this is based on anecdotal evidence and clinical trials have not shown any benefit over the biosynthetic forms.[8] Thyroid tablets are reported to have different effects, which can be attributed to the difference in torsional angles surrounding the reactive site of the molecule.[9]
Thyronamines have no medical usages yet, though their use has been proposed for controlled induction of hypothermia, which causes the brain to enter a protective cycle, useful in preventing damage during ischemic shock.
Synthetic thyroxine was first successfully produced by Charles Robert Harington and George Barger in 1926.
Formulations
Most people are treated with levothyroxine, or a similar synthetic thyroid hormone.[10][11][12] Different polymorphs of the compound have different solubilities and potencies.[13] Additionally, natural thyroid hormone supplements from the dried thyroids of animals are still available.[12][14][15] Levothyroxine contains T4 only and is therefore largely ineffective for patients unable to convert T4 to T3.[16] These patients may choose to take natural thyroid hormone, as it contains a mixture of T4 and T3,[12][17][18][19][20] or alternatively supplement with a synthetic T3 treatment.[21] In these cases, synthetic liothyronine is preferred due to the potential differences between the natural thyroid products. Some studies show that the mixed therapy is beneficial to all patients, but the addition of lyothyronine contains additional side effects and the medication should be evaluated on an individual basis.[22] Some natural thyroid hormone brands are FDA approved, but some are not.[23][24][25] Thyroid hormones are generally well tolerated.[11] Thyroid hormones are usually not dangerous for pregnant women or nursing mothers, but should be given under a doctor's supervision. In fact, if a woman who is hypothyroid is left untreated, her baby is at a higher risk for birth defects. When pregnant, a woman with a low-functioning thyroid will also need to increase her dosage of thyroid hormone.[11] One exception is that thyroid hormones may aggravate heart conditions, especially in older patients; therefore, doctors may start these patients on a lower dose and work up to a larger one to avoid risk of heart attack.[12]
Thyroid Metabolism
Central
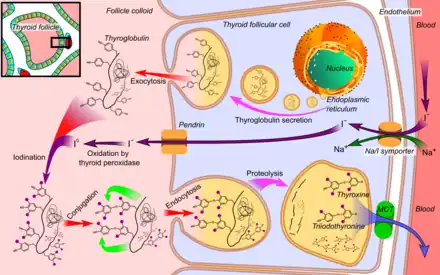
- Thyroglobulin is synthesized in the rough endoplasmic reticulum and follows the secretory pathway to enter the colloid in the lumen of the thyroid follicle by exocytosis.
- Meanwhile, a sodium-iodide (Na/I) symporter pumps iodide (I−) actively into the cell, which previously has crossed the endothelium by largely unknown mechanisms.
- This iodide enters the follicular lumen from the cytoplasm by the transporter pendrin, in a purportedly passive manner.
- In the colloid, iodide (I−) is oxidized to iodine (I0) by an enzyme called thyroid peroxidase.
- Iodine (I0) is very reactive and iodinates the thyroglobulin at tyrosyl residues in its protein chain (in total containing approximately 120 tyrosyl residues).
- In conjugation, adjacent tyrosyl residues are paired together.
- Thyroglobulin re-enters the follicular cell by endocytosis.
- Proteolysis by various proteases liberates thyroxine and triiodothyronine molecules
- Efflux of thyroxine and triiodothyronine from follicular cells, which appears to be largely through monocarboxylate transporter (MCT) 8 and 10,[27][28] and entry into the blood.
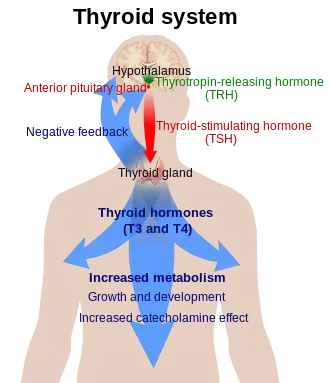
Thyroid hormones (T4 and T3) are produced by the follicular cells of the thyroid gland and are regulated by TSH made by the thyrotropes of the anterior pituitary gland. The effects of T4 in vivo are mediated via T3 (T4 is converted to T3 in target tissues). T3 is three to five times as active than T4.
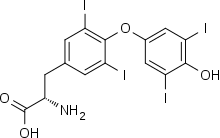
Thyroxine (3,5,3′,5′-tetraiodothyronine) is produced by follicular cells of the thyroid gland. It is produced as the precursor thyroglobulin (this is not the same as thyroxine-binding globulin (TBG)), which is cleaved by enzymes to produce active T4.
The steps in this process are as follows:[26]
- The Na+/I− symporter transports two sodium ions across the basement membrane of the follicular cells along with an iodide ion. This is a secondary active transporter that utilises the concentration gradient of Na+ to move I− against its concentration gradient.
- I− is moved across the apical membrane into the colloid of the follicle by pendrin .
- Thyroperoxidase oxidizes two I− to form I2. Iodide is non-reactive, and only the more reactive iodine is required for the next step.
- The thyroperoxidase iodinates the tyrosyl residues of the thyroglobulin within the colloid. The thyroglobulin was synthesised in the ER of the follicular cell and secreted into the colloid.
- Iodinated Thyroglobulin binds megalin for endocytosis back into cell.
- Thyroid-stimulating hormone (TSH) released from the anterior pituitary (also known as the adenohypophysis) binds the TSH receptor (a Gs protein-coupled receptor) on the basolateral membrane of the cell and stimulates the endocytosis of the colloid.
- The endocytosed vesicles fuse with the lysosomes of the follicular cell. The lysosomal enzymes cleave the T4 from the iodinated thyroglobulin.
- The thyroid hormones cross the follicular cell membrane towards the blood vessels by an unknown mechanism.[26] Text books have stated that diffusion is the main means of transport,[29] but recent studies indicate that monocarboxylate transporter (MCT) 8 and 10 play major roles in the efflux of the thyroid hormones from the thyroid cells.[27][28]
Thyroglobulin (Tg) is a 660 kDa, dimeric protein produced by the follicular cells of the thyroid and used entirely within the thyroid gland.[30] Thyroxine is produced by attaching iodine atoms to the ring structures of this protein's tyrosine residues; thyroxine (T4) contains four iodine atoms, while triiodothyronine (T3), otherwise identical to T4, has one less iodine atom per molecule. The thyroglobulin protein accounts for approximately half of the protein content of the thyroid gland. Each thyroglobulin molecule contains approximately 100–120 tyrosine residues, a small number of which (<20) are subject to iodination catalysed by thyroperoxidase.[31] The same enzyme then catalyses "coupling" of one modified tyrosine with another, via a free-radical-mediated reaction, and when these iodinated bicyclic molecules are released by hydrolysis of the protein, T3 and T4 are the result. Therefore, each thyroglobulin protein molecule ultimately yields very small amounts of thyroid hormone (experimentally observed to be on the order of 5–6 molecules of either T4 or T3 per original molecule of thyroglobulin).[31]
More specifically, the monatomic anionic form of iodine, iodide (I—), is actively absorbed from the bloodstream by a process called iodide trapping.[32] In this process, sodium is cotransported with iodide from the basolateral side of the membrane into the cell, and then concentrated in the thyroid follicles to about thirty times its concentration in the blood. Then, in the first reaction catalysed by the enzyme thyroperoxidase, tyrosine residues in the protein thyroglobulin are iodinated on their phenol rings, at one or both of the positions ortho to the phenolic hydroxyl group, yielding monoiodotyrosine (MIT) and diiodotyrosine (DIT), respectively. This introduces 1–2 atoms of the element iodine, covalently bound, per tyrosine residue. The further coupling together of two fully iodinated tyrosine residues, also catalysed by thyroperoxidase, yields the peptidic (still peptide-bound) precursor of thyroxine, and coupling one molecule of MIT and one molecule of DIT yields the comparable precursor of triiodothyronine:
- peptidic MIT + peptidic DIT → peptidic triiodothyronine (eventually released as triiodothyronine, T3)
- 2 peptidic DITs → peptidic thyroxine (eventually released as thyroxine, T4)
(Coupling of DIT to MIT in the opposite order yields a substance, r-T3, which is biologically inactive.) Hydrolysis (cleavage to individual amino acids) of the modified protein by proteases then liberates T3 and T4, as well as the non-coupled tyrosine derivatives MIT and DIT. The hormones T4 and T3 are the biologically active agents central to metabolic regulation.
Peripheral
Thyroxine is believed to be a prohormone and a reservoir for the most active and main thyroid hormone T3.[33] T4 is converted as required in the tissues by iodothyronine deiodinase.[34] Deficiency of deiodinase can mimic hypothyroidism due to iodine deficiency.[35] T3 is more active than T4,[36] though it is present in less quantity than T4.
Initiation of production in fetuses
Thyrotropin-releasing hormone (TRH) is released from hypothalamus by 6 – 8 weeks, and thyroid-stimulating hormone (TSH) secretion from fetal pituitary is evident by 12 weeks of gestation, and fetal production of thyroxine (T4) reaches a clinically significant level at 18–20 weeks.[37] Fetal triiodothyronine (T3) remains low (less than 15 ng/dL) until 30 weeks of gestation, and increases to 50 ng/dL at term.[37] Fetal self-sufficiency of thyroid hormones protects the fetus against e.g. brain development abnormalities caused by maternal hypothyroidism.[38]
Iodine deficiency
If there is a deficiency of dietary iodine, the thyroid will not be able to make thyroid hormones. The lack of thyroid hormones will lead to decreased negative feedback on the pituitary, leading to increased production of thyroid-stimulating hormone, which causes the thyroid to enlarge (the resulting medical condition is called endemic colloid goitre; see goitre).[39] This has the effect of increasing the thyroid's ability to trap more iodide, compensating for the iodine deficiency and allowing it to produce adequate amounts of thyroid hormone.
Circulation and transport
Plasma transport
Most of the thyroid hormone circulating in the blood is bound to transport proteins, and only a very small fraction is unbound and biologically active. Therefore, measuring concentrations of free thyroid hormones is important for diagnosis, while measuring total levels can be misleading.
Thyroid hormone in the blood is usually distributed as follows:
| Type | Percent |
|---|---|
| bound to thyroxine-binding globulin (TBG) | 70% |
| bound to transthyretin or "thyroxine-binding prealbumin" (TTR or TBPA) | 10–15% |
| albumin | 15–20% |
| unbound T4 (fT4) | 0.03% |
| unbound T3 (fT3) | 0.3% |
Despite being lipophilic, T3 and T4 cross the cell membrane via carrier-mediated transport, which is ATP-dependent.[40]
T1a and T0a are positively charged and do not cross the membrane; they are believed to function via the trace amine-associated receptor TAAR1 (TAR1, TA1), a G-protein-coupled receptor located in the cytoplasm.
Another critical diagnostic tool is measurement of the amount of thyroid-stimulating hormone (TSH) that is present.
Membrane transport
Contrary to common belief, thyroid hormones cannot traverse cell membranes in a passive manner like other lipophilic substances. The iodine in o-position makes the phenolic OH-group more acidic, resulting in a negative charge at physiological pH. However, at least 10 different active, energy-dependent and genetically regulated iodothyronine transporters have been identified in humans. They guarantee that intracellular levels of thyroid hormones are higher than in blood plasma or interstitial fluids.[41]
Intracellular transport
Little is known about intracellular kinetics of thyroid hormones. However, recently it could be demonstrated that the crystallin CRYM binds 3,5,3′-triiodothyronine in vivo.[42]
Mechanism of action
The thyroid hormones function via a well-studied set of nuclear receptors, termed the thyroid hormone receptors. These receptors, together with corepressor molecules, bind DNA regions called thyroid hormone response elements (TREs) near genes. This receptor-corepressor-DNA complex can block gene transcription. Triiodothyronine (T3), which is the active form of thyroxine (T4), goes on to bind to receptors. The deiodinase catalyzed reaction removes an iodine atom from the 5′ position of the outer aromatic ring of thyroxine's (T4) structure.[43] When triiodothyronine (T3) binds a receptor, it induces a conformational change in the receptor, displacing the corepressor from the complex. This leads to recruitment of coactivator proteins and RNA polymerase, activating transcription of the gene.[44] Although this general functional model has considerable experimental support, there remain many open questions.[45]
More recently genetic evidence has been obtained for a second mechanism of thyroid hormone action involving one of the same nuclear receptors, TRβ, acting rapidly in the cytoplasm through the PI3K.[46][47] This mechanism is conserved in all mammals but not fish or amphibians, and regulates brain development[46] and adult metabolism.[47] The mechanism itself parallels the actions of the nuclear receptor in the nucleus: in the absence of hormone, TRβ binds to PI3K and inhibits its activity, but when hormone binds the complex dissociates, PI3K activity increases, and the hormone bound receptor diffuses into the nucleus.[46]
Thyroxine, iodine and apoptosis
Thyroxine and iodine stimulate the spectacular apoptosis of the cells of the larval gills, tail and fins in amphibian metamorphosis, and stimulate the evolution of their nervous system transforming the aquatic, vegetarian tadpole into the terrestrial, carnivorous frog. In fact, amphibian frog Xenopus laevis serves as an ideal model system for the study of the mechanisms of apoptosis.[48][49][50][51]
Effects of triiodothyronine
Effects of triiodothyronine (T3) which is the metabolically active form:
- Increases cardiac output
- Increases heart rate
- Increases ventilation rate
- Increases basal metabolic rate
- Potentiates the effects of catecholamines (i.e. increases sympathetic activity)
- Potentiates brain development
- Thickens endometrium in females
- Increases catabolism of proteins and carbohydrates[52]
Measurement
Further information: Thyroid function tests
Triiodothyronine (T3) and thyroxine (T4) can be measured as free T3 and free T4, which are indicators of their activities in the body.[53] They can also be measured as total T3 and total T4, which depend on the amount that is bound to thyroxine-binding globulin (TBG).[53] A related parameter is the free thyroxine index, which is total T4 multiplied by thyroid hormone uptake, which, in turn, is a measure of the unbound TBG.[54] Additionally, thyroid disorders can be detected prenatally using advanced imaging techniques and testing fetal hormone levels.[55]
Related diseases
Both excess and deficiency of thyroxine can cause disorders.
- Hyperthyroidism (an example is Graves' disease) is the clinical syndrome caused by an excess of circulating free thyroxine, free triiodothyronine, or both. It is a common disorder that affects approximately 2% of women and 0.2% of men. Thyrotoxicosis is often used interchangeably with hyperthyroidism, but there are subtle differences. Although thyrotoxicosis also refers to an increase in circulating thyroid hormones, it can be caused by the intake of thyroxine tablets or by an over-active thyroid, whereas hyperthyroidism refers solely to an over-active thyroid.
- Hypothyroidism (an example is Hashimoto's thyroiditis) is the case where there is a deficiency of thyroxine, triiodothyronine, or both.
- Clinical depression can sometimes be caused by hypothyroidism.[56] Some research[57] has shown that T3 is found in the junctions of synapses, and regulates the amounts and activity of serotonin, norepinephrine, and γ-aminobutyric acid (GABA) in the brain.
- Hair loss can sometimes be attributed to a malfunction of T3 and T4. Normal hair growth cycle may be affected disrupting the hair growth.
- Both thyroid excess and deficiency can cause cardiovascular disorders or make preexisting conditions worse.[58] The link between excess and deficiency of thyroid hormone on conditions like arrhythmias, heart failure, and atherosclerotic vascular diseases, have been established for nearly 200 years.[59][58]
Preterm births can suffer neurodevelopmental disorders due to lack of maternal thyroid hormones, at a time when their own thyroid is unable to meet their postnatal needs.[60] Also in normal pregnancies, adequate levels of maternal thyroid hormone are vital in order to ensure thyroid hormone availability for the foetus and its developing brain.[61] Congenital hypothyroidism occurs in every 1 in 1600–3400 newborns with most being born asymptomatic and developing related symptoms weeks after birth.[62]
Anti-thyroid drugs
Iodine uptake against a concentration gradient is mediated by a sodium–iodine symporter and is linked to a sodium-potassium ATPase. Perchlorate and thiocyanate are drugs that can compete with iodine at this point. Compounds such as goitrin, carbimazole, methimazole, propylthiouracil can reduce thyroid hormone production by interfering with iodine oxidation.[63]
See also
- Goitre
- Graves–Basedow disease
- Hashimoto's thyroiditis
- Hormone
- Polar T3 syndrome
- Thyroid gland
- Thyroid-stimulating hormone
- Thyronamines, metabolites of the thyroid hormones that act at the trace amine-associated receptor TAAR1 (TAR1)
References
- ↑ References used in image are found in image article in Commons:Commons:File:Thyroid system.png#References.
- ↑ Irizarry L (23 April 2014). "Thyroid Hormone Toxicity". Medscape. WedMD LLC. Retrieved 2 May 2014.
- ↑ Pilo A, Iervasi G, Vitek F, Ferdeghini M, Cazzuola F, Bianchi R (April 1990). "Thyroidal and peripheral production of 3,5,3′-triiodothyronine in humans by multicompartmental analysis". The American Journal of Physiology. 258 (4 Pt 1): E715–E726. doi:10.1152/ajpendo.1990.258.4.E715. PMID 2333963.
- ↑ "1926 Edward C Kendall". American Society for Biochemistry and Molecular Biology. Archived from the original on 2012-03-19. Retrieved 2011-07-04.
- ↑ "The Top 300 of 2021". ClinCalc. Retrieved 18 February 2021.
- ↑ "Levothyroxine - Drug Usage Statistics". ClinCalc. Retrieved 18 February 2021.
- ↑ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ Grozinsky-Glasberg S, Fraser A, Nahshoni E, Weizman A, Leibovici L (July 2006). "Thyroxine-triiodothyronine combination therapy versus thyroxine monotherapy for clinical hypothyroidism: meta-analysis of randomized controlled trials". The Journal of Clinical Endocrinology and Metabolism. 91 (7): 2592–2599. doi:10.1210/jc.2006-0448. PMID 16670166.
- ↑ Schweizer U, Steegborn C (October 2015). "Thyroid hormones--From Crystal Packing to Activity to Reactivity". Angewandte Chemie. 54 (44): 12856–12858. doi:10.1002/anie.201506919. PMID 26358899.
- ↑ Robert Lloyd Segal, MD Endocrinologist
- 1 2 3 "preferred thyroid hormone --Levothyroxine Sodium (Synthroid, Levoxyl, Levothroid, Unithroid)". MedicineNet.com. Retrieved 27 March 2009.
- 1 2 3 4 "Hypothyroidism Causes, Symptoms, Diagnosis, Treatment Information Produced by Medical Doctors". MedicineNet.com. Retrieved 27 March 2009.
- ↑ Mondal S, Mugesh G (September 2015). "Structure Elucidation and Characterization of Different Thyroxine Polymorphs". Angewandte Chemie. 54 (37): 10833–10837. doi:10.1002/anie.201505281. PMID 26213168.
- ↑ Cooper DS (May 1989). "Thyroid hormone treatment: new insights into an old therapy". JAMA. 261 (18): 2694–2695. doi:10.1001/jama.1989.03420180118042. PMID 2709547.
- ↑ Clyde PW, Harari AE, Mohamed Shakir KM (2004). "Synthetic Thyroxine vs Desiccated Thyroid -Reply (citing Cooper, DS, above)". JAMA: The Journal of the American Medical Association. 291 (12): 1445. doi:10.1001/jama.291.12.1445-b.
- ↑ Wiersinga WM (2001). "Thyroid hormone replacement therapy". Hormone Research. 56 (Suppl 1): 74–81. doi:10.1159/000048140. PMID 11786691. S2CID 46756918.
- ↑ "Consequences of Not Taking Thyroid Medications - Implications of Failing to Take Prescription Thyroid Drugs", Retrieved on 2009-03-27
- ↑ "Armour Thyroid", Retrieved on 2009-04-01
- ↑ "Nature-Throid", Retrieved on 2009-04-01
- ↑ "Armour Thyroid Shortages Worsening: What Can Thyroid Patients Do?", Retrieved on 2009-03-27
- ↑ Liothyronine
- ↑ Escobar-Morreale HF, Botella-Carretero JI, Morreale de Escobar G (January 2015). "Treatment of hypothyroidism with levothyroxine or a combination of levothyroxine plus L-triiodothyronine". Best Practice & Research. Clinical Endocrinology & Metabolism. 29 (1): 57–75. doi:10.1016/j.beem.2014.10.004. hdl:10261/124621. PMID 25617173.
- ↑ "Thyroid Information" Archived 2012-02-27 at the Wayback Machine, Retrieved on 2009-03-27
- ↑ Eliason BC, Doenier JA, Nuhlicek DN (March 1994). "Desiccated thyroid in a nutritional supplement". The Journal of Family Practice. 38 (3): 287–288. PMID 8126411.
- ↑ "Nature-Throid", Retrieved on 2009-04-01
- 1 2 3 Chapter 49, "Synthesis of Thyroid Hormones" in: Walter F. Boron; Emile L. Boulpaep (2012). Medical Physiology (2nd ed.). Elsevier/Saunders. ISBN 9781437717532.
- 1 2 Friesema EC, Jansen J, Jachtenberg JW, Visser WE, Kester MH, Visser TJ (June 2008). "Effective cellular uptake and efflux of thyroid hormone by human monocarboxylate transporter 10". Molecular Endocrinology. 22 (6): 1357–1369. doi:10.1210/me.2007-0112. PMC 5419535. PMID 18337592.
- 1 2 Brix K, Führer D, Biebermann H (August 2011). "Molecules important for thyroid hormone synthesis and action - known facts and future perspectives". Thyroid Research. 4 (Suppl. 1): S9. doi:10.1186/1756-6614-4-S1-S9. PMC 3155115. PMID 21835056.
- ↑ Human Anatomy & Physiology, Sixth Edition. Benjamin Cummings. May 2, 2003. ISBN 978-0805354621.
- ↑ Dabbs DJ (2019). Diagnostic Immunohistochemistry. Elsevier. pp. 345–389.
- 1 2 Boron, W.F. (2003). Medical Physiology: A Cellular And Molecular Approach. Elsevier/Saunders. ISBN 1416023283.
- ↑ Ahad F, Ganie SA (January 2010). "Iodine, Iodine metabolism and Iodine deficiency disorders revisited". Indian Journal of Endocrinology and Metabolism. 14 (1): 13–17. PMC 3063534. PMID 21448409.
- ↑ Kansagra SM, McCudden CR, Willis MS (June 2010). "The Challenges and Complexities of Thyroid Hormone Replacement". Laboratory Medicine. 41 (6): 338–348. doi:10.1309/LMB39TH2FZGNDGIM.
- ↑ St Germain DL, Galton VA, Hernandez A (March 2009). "Minireview: Defining the roles of the iodothyronine deiodinases: current concepts and challenges". Endocrinology. 150 (3): 1097–1107. doi:10.1210/en.2008-1588. PMC 2654746. PMID 19179439.
- ↑ Wass JA, Stewart PM, eds. (2011). Oxford Textbook of Endocrinology and Diabetes (2nd ed.). Oxford: Oxford University Press. p. 565. ISBN 978-0-19-923529-2.
- ↑ Wass JA, Stewart PM, eds. (2011). Oxford Textbook of Endocrinology and diabetes (2nd ed.). Oxford: Oxford University Press. p. 18. ISBN 978-0-19-923529-2.
- 1 2 Eugster EA, Pescovitz OH (2004). Pediatric endocrinology: mechanisms, manifestations and management. Hagerstwon, MD: Lippincott Williams & Wilkins. pp. 493 (Table 33-3). ISBN 978-0-7817-4059-3.
- ↑ Zoeller RT (April 2003). "Transplacental thyroxine and fetal brain development". The Journal of Clinical Investigation. 111 (7): 954–957. doi:10.1172/JCI18236. PMC 152596. PMID 12671044.
- ↑ McPherson, Richard A.; Pincus, Matthew R. (2017-04-05). Henry's clinical diagnosis and management by laboratory methods. McPherson, Richard A.,, Pincus, Matthew R. (23rd ed.). St. Louis, Mo. ISBN 9780323413152. OCLC 949280055.
- ↑ Hennemann G, Docter R, Friesema EC, de Jong M, Krenning EP, Visser TJ (August 2001). "Plasma membrane transport of thyroid hormones and its role in thyroid hormone metabolism and bioavailability". Endocrine Reviews. 22 (4): 451–476. doi:10.1210/edrv.22.4.0435. PMID 11493579.
- ↑ Dietrich JW, Brisseau K, Boehm BO (August 2008). "[Absorption, transport and bio-availability of iodothyronines]". Deutsche Medizinische Wochenschrift. 133 (31–32): 1644–1648. doi:10.1055/s-0028-1082780. PMID 18651367.
- ↑ Suzuki S, Suzuki N, Mori J, Oshima A, Usami S, Hashizume K (April 2007). "micro-Crystallin as an intracellular 3,5,3′-triiodothyronine holder in vivo". Molecular Endocrinology. 21 (4): 885–894. doi:10.1210/me.2006-0403. PMID 17264173.
- ↑ Mullur R, Liu YY, Brent GA (April 2014). "Thyroid hormone regulation of metabolism". Physiological Reviews. 94 (2): 355–382. doi:10.1152/physrev.00030.2013. PMC 4044302. PMID 24692351.
- ↑ Wu Y, Koenig RJ (August 2000). "Gene regulation by thyroid hormone". Trends in Endocrinology and Metabolism. 11 (6): 207–211. doi:10.1016/s1043-2760(00)00263-0. PMID 10878749. S2CID 44602986.
- ↑ Ayers S, Switnicki MP, Angajala A, Lammel J, Arumanayagam AS, Webb P (2014). "Genome-wide binding patterns of thyroid hormone receptor beta". PLOS ONE. 9 (2): e81186. Bibcode:2014PLoSO...981186A. doi:10.1371/journal.pone.0081186. PMC 3928038. PMID 24558356.
- 1 2 3 Martin NP, Marron Fernandez de Velasco E, Mizuno F, Scappini EL, Gloss B, Erxleben C, et al. (September 2014). "A rapid cytoplasmic mechanism for PI3-kinase regulation by the nuclear thyroid hormone receptor, TRβ, and genetic evidence for its role in the maturation of mouse hippocampal synapses in vivo". Endocrinology. 155 (9): 3713–3724. doi:10.1210/en.2013-2058. PMC 4138568. PMID 24932806.
- 1 2 Hönes GS, Rakov H, Logan J, Liao XH, Werbenko E, Pollard AS, et al. (December 2017). "Noncanonical thyroid hormone signaling mediates cardiometabolic effects in vivo". Proceedings of the National Academy of Sciences of the United States of America. 114 (52): E11323–E11332. doi:10.1073/pnas.1706801115. PMC 5748168. PMID 29229863.
- ↑ Jewhurst K, Levin M, McLaughlin KA (2014). "Optogenetic Control of Apoptosis in Targeted Tissues of Xenopus laevis Embryos". Journal of Cell Death. 7: 25–31. doi:10.4137/JCD.S18368. PMC 4213186. PMID 25374461.
- ↑ Venturi, Sebastiano (2011). "Evolutionary Significance of Iodine". Current Chemical Biology. 5 (3): 155–162. doi:10.2174/187231311796765012. ISSN 1872-3136.
- ↑ Venturi S, Venturi M (2014). "Iodine, PUFAs and Iodolipids in Health and Disease: An Evolutionary Perspective". Human Evolution. 29 (1–3): 185–205.
- ↑ Tamura K, Takayama S, Ishii T, Mawaribuchi S, Takamatsu N, Ito M (June 2015). "Apoptosis and differentiation of Xenopus tail-derived myoblasts by thyroid hormone". Journal of Molecular Endocrinology. 54 (3): 185–192. doi:10.1530/JME-14-0327. PMID 25791374.
- ↑ Gelfand RA, Hutchinson-Williams KA, Bonde AA, Castellino P, Sherwin RS (June 1987). "Catabolic effects of thyroid hormone excess: the contribution of adrenergic activity to hypermetabolism and protein breakdown". Metabolism. 36 (6): 562–569. doi:10.1016/0026-0495(87)90168-5. PMID 2884552.
- 1 2 Stockigt, Jim R (January 2002). "Case finding and screening strategies for thyroid dysfunction". Clinica Chimica Acta. 315 (1–2): 111–124. doi:10.1016/s0009-8981(01)00715-x. ISSN 0009-8981. PMID 11728414.
- ↑ Military Obstetrics & Gynecology > Thyroid Function Tests In turn citing: Operational Medicine 2001, Health Care in Military Settings, NAVMED P-5139, May 1, 2001, Bureau of Medicine and Surgery, Department of the Navy, 2300 E Street NW, Washington, D.C., 20372-5300
- ↑ Polak M, Luton D (March 2014). "Fetal thyroïdology". Best Practice & Research. Clinical Endocrinology & Metabolism. 28 (2): 161–73. doi:10.1016/j.beem.2013.04.013. PMID 24629859.
- ↑ Kirkegaard C, Faber J (January 1998). "The role of thyroid hormones in depression". European Journal of Endocrinology. 138 (1): 1–9. doi:10.1530/eje.0.1380001. PMID 9461307.
- ↑ Dratman MB, Gordon JT (December 1996). "Thyroid hormones as neurotransmitters". Thyroid. 6 (6): 639–647. doi:10.1089/thy.1996.6.639. PMID 9001201.
- 1 2 Cappola AR, Desai AS, Medici M, Cooper LS, Egan D, Sopko G, Fishman GI, Goldman S, Cooper DS, Mora S, Kudenchuk PJ, Hollenberg AN, McDonald CL, Ladenson PW. Thyroid and Cardiovascular Disease Research Agenda for Enhancing Knowledge, Prevention, and Treatment. Circulation. 2019 May 13:10.1161/CIRCULATIONAHA.118.036859. doi: 10.1161/CIRCULATIONAHA.118.036859. Epub ahead of print. PMID 31081673; PMCID: PMC6851449.
- ↑ Parry CH. Elements of Pathology and Therapeutics, Being the Outlines of a Work. Bath, England: R. Cruttwell, 1815.
- ↑ Berbel P, Navarro D, Ausó E, Varea E, Rodríguez AE, Ballesta JJ, et al. (June 2010). "Role of late maternal thyroid hormones in cerebral cortex development: an experimental model for human prematurity". Cerebral Cortex. 20 (6): 1462–1475. doi:10.1093/cercor/bhp212. PMC 2871377. PMID 19812240.
- ↑ Korevaar TI, Muetzel R, Medici M, Chaker L, Jaddoe VW, de Rijke YB, et al. (January 2016). "Association of maternal thyroid function during early pregnancy with offspring IQ and brain morphology in childhood: a population-based prospective cohort study". The Lancet. Diabetes & Endocrinology. 4 (1): 35–43. doi:10.1016/s2213-8587(15)00327-7. PMID 26497402.
- ↑ Szinnai G (March 2014). "Genetics of normal and abnormal thyroid development in humans". Best Practice & Research. Clinical Endocrinology & Metabolism. 28 (2): 133–150. doi:10.1016/j.beem.2013.08.005. PMID 24629857.
- ↑ Spiegel C, Bestetti GE, Rossi GL, Blum JW (September 1993). "Normal circulating triiodothyronine concentrations are maintained despite severe hypothyroidism in growing pigs fed rapeseed presscake meal". The Journal of Nutrition. 123 (9): 1554–1561. doi:10.1093/jn/123.9.1554. PMID 8360780.
External links
- Find TH response elements in DNA sequences.
- Triiodothyronine bound to proteins in the PDB
- Thyroxine bound to proteins in the PDB
- T4 at Lab Tests Online Archived 2013-11-01 at the Wayback Machine
Thyroid hormone treatment in thyroid disease
- Thyroid Hormone Treatment Brochure by the American Thyroid Association
- Elaborate article about the use of thyroid drugs Written by an MD
- Thyroid Disease Manager Collection of elaborate medical articles on thyroid disease, including information on thyroid hormones