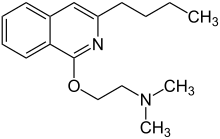
Quinisocaine
Quinisocaine (INN) or dimethisoquin (BAN and USAN) is a topical anesthetic used as an antipruritic.[1]
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.001.546 |
| Chemical and physical data | |
| Formula | C17H24N2O |
| Molar mass | 272.392 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
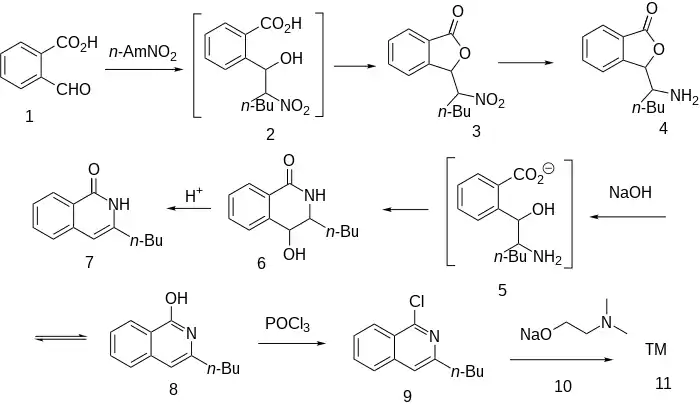
Synthesis
The Henry reaction between phthalaldehydic acid (2-Formylbenzoic acid) [119-67-5] (1) and 1-nitropentane [628-05-7] occurs by a mechanism that involves a hydroxy acid (2). Expulsion of water then gives (3). Reduction of the nitro group via catalytic hydrogenation leads to the amine, CID:158569430 (4). Treatment of that amine with sodium hydroxide leads to ring opening of the lactone ring to the intermediary amino acid (5). This cyclises spontaneously to the lactam so that the product isolated from the reaction mixture is in fact the isoquinoline derivative, CID:154188092 (7). Dehydration by means of strong acid gives 3-Butylisocarbostyril [132-90-1] (8). Phosphorus oxychloride converts the oxygen function to the corresponding chloride via the enol forms 3-butyl-1-chloroisoquinoline [87-06-9] (9). Displacement of halogen with the sodium salt from 2-dimethylaminoethanol (10) affords dimethisoquin (11).
References
- Elks J (1990). "Dimethisoquin". The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Dordrecht: Springer. p. 430. ISBN 978-1-4757-2085-3.
- Eloy, F. et al, Chim. Ther., 1969, 4, 469.
- Wilson, James W.; Dawson, Norman D.; Brooks, Walter.; Ullyot, Glenn E. (1949). "Local Anesthetics. Aminoalkoxyisoquinoline Derivatives". Journal of the American Chemical Society 71 (3): 937–938. doi:10.1021/ja01171a047.
- Anon., GB 681358 (1952 to Smith Kline and French International Co).
- Ullyot, U.S. Patent 2,612,503 (1952 to SK & F).