Beryllium fluoride
Beryllium fluoride is the inorganic compound with the formula BeF2. This white solid is the principal precursor for the manufacture of beryllium metal. Its structure resembles that of quartz, but BeF2 is highly soluble in water.
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Beryllium fluoride | |
| Other names
Beryllium difluoride Difluoroberyllane | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.029.198 |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| BeF2 | |
| Molar mass | 47.01 g/mol hygroscopic |
| Appearance | colorless, glassy lumps |
| Density | 1.986 g/cm3 |
| Melting point | 554 °C (1,029 °F; 827 K)[1] |
| Boiling point | 1,169 °C (2,136 °F; 1,442 K)[2] |
| very soluble | |
| Solubility | sparingly soluble in alcohol |
| Structure | |
| Trigonal, α-quartz | |
| P3121 (No. 152), Pearson symbol hP9[3] | |
a = 473.29 pm, c = 517.88 pm | |
| Linear | |
| Thermochemistry | |
Heat capacity (C) |
1.102 J/K or 59 J/mol K |
Std molar entropy (S⦵298) |
45 J/mol K |
Std enthalpy of formation (ΔfH⦵298) |
-1028.2 kJ/g or -1010 kJ/mol |
Gibbs free energy (ΔfG⦵) |
-941 kJ/mol |
| Hazards[4] | |
| GHS labelling: | |
    | |
| Danger | |
| H301, H305, H311, H314, H315, H319, H330, H335, H372, H411 | |
| P201, P202, P260, P264, P270, P271, P273, P280, P281, P284, P301+P310, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P310, P312, P314, P320, P321, P322, P330, P361, P363, P391, P403+P233, P405, P501 | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
90 mg/kg (oral, rat) 100 mg/kg (oral, mouse)[5] |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
TWA 0.002 mg/m3 C 0.005 mg/m3 (30 minutes), with a maximum peak of 0.025 mg/m3 (as Be)[6] |
REL (Recommended) |
Ca C 0.0005 mg/m3 (as Be)[6] |
IDLH (Immediate danger) |
Ca [4 mg/m3 (as Be)][6] |
| Safety data sheet (SDS) | InChem MSDS |
| Related compounds | |
Other anions |
Beryllium chloride Beryllium bromide Beryllium iodide |
Other cations |
Magnesium fluoride Calcium fluoride Strontium fluoride Barium fluoride Radium fluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Properties
Beryllium fluoride has distinctive optical properties. In the form of fluoroberyllate glass it has the lowest refractive index for a solid at room temperature of 1.275. Its dispersive power is the lowest for a solid at 0.0093, and the nonlinear coefficient is also the lowest at 2 × 10−14.
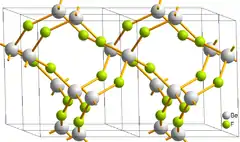
Structure and bonding

The structure of solid BeF2 resembles that of cristobalite. Be2+ centers are four coordinate and tetrahedral and the fluoride centers are two-coordinate.[7] The Be-F bond lengths are about 1.54 Å.[8] Analogous to SiO2, BeF2 can also adopt a number of related structures. An analogy also exists between BeF2 and AlF3: both adopt extended structures at mild temperature.
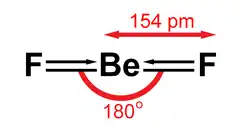
Gas and liquid BeF2
Gaseous beryllium fluoride adopts a linear structure, with a Be-F distance of 143 pm.[9] BeF2 reaches a vapor pressure of 10 Pa at 686 °C, 100 Pa at 767 °C, 1 kPa at 869 °C, 10 kPa at 999 °C, and 100 kPa at 1172 °C.[10] Molecular BeF2 in the gaseous state is isoelectronic to carbon dioxide.
'Molecules' of liquid beryllium fluoride have a fluctuating tetrahedral structure. Additionally, the density of liquid BeF2 decreases near its freezing point, as Be2+ and F− ions begin to coordinate more strongly with one another, leading to the expansion of voids between formula units.[11]
Production
The processing of beryllium ores generates impure Be(OH)2. This material reacts with ammonium bifluoride to give ammonium tetrafluoroberyllate:
- Be(OH)2 + 2 (NH4)HF2 → (NH4)2BeF4 + 2 H2O
Tetrafluoroberyllate is a robust ion, which allows its purification by precipitation of various impurities as their hydroxides. Heating purified (NH4)2BeF4 gives the desired product:
- (NH4)2BeF4 → 2 NH3 + 2 HF + BeF2
In general the reactivity of BeF2 ions with fluoride are quite analogous to the reactions of SiO2 with oxides.[12]
Applications
Reduction of BeF2 at 1300 °C with magnesium in a graphite crucible provides the most practical route to metallic beryllium:[9]
- BeF2 + Mg → Be + MgF2
The chloride is not a useful precursor because of its volatility.
Niche uses
Beryllium fluoride is used in biochemistry, particularly protein crystallography as a mimic of phosphate. Thus, ADP and beryllium fluoride together tend to bind to ATP sites and inhibit protein action, making it possible to crystallise proteins in the bound state.[13][14]
Beryllium fluoride forms a basic constituent of the preferred fluoride salt mixture used in liquid-fluoride nuclear reactors. Typically beryllium fluoride is mixed with lithium fluoride to form a base solvent (FLiBe), into which fluorides of uranium and thorium are introduced. Beryllium fluoride is exceptionally chemically stable, and LiF/BeF2 mixtures (FLiBe) have low melting points (360–459 °C) and the best neutronic properties of fluoride salt combinations appropriate for reactor use. MSRE used two different mixtures in the two cooling circuits.
Safety
Beryllium compounds are highly toxic. The increased toxicity of beryllium in the presence of fluoride has been noted as early as 1949.[15] The LD50 in mice is about 100 mg/kg by ingestion and 1.8 mg/kg by intravenous injection.
References
- "Beryllium Fluoride". American Elements. Retrieved 10 July 2023.
- Lide, David R., ed. (2006). CRC Handbook of Chemistry and Physics (87th ed.). Boca Raton, FL: CRC Press. ISBN 0-8493-0487-3.
- Wright, Albert F.; Fitch, Andrew N.; Wright, Adrian C. (1988). "The preparation and structure of the α- and β-quartz polymorphs of beryllium fluoride". Journal of Solid State Chemistry. 73 (2): 298. Bibcode:1988JSSCh..73..298W. doi:10.1016/0022-4596(88)90113-2.
- "Beryllium Difluoride". PubChem. National Institute of Health. Retrieved October 13, 2017.
- "Beryllium compounds (as Be)". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- NIOSH Pocket Guide to Chemical Hazards. "#0054". National Institute for Occupational Safety and Health (NIOSH).
- Wells A.F. (1984) Structural Inorganic Chemistry 5th edition Oxford Science Publications ISBN 0-19-855370-6
- Pallavi Ghalsasi, Prasanna S. Ghalsasi, "Single Crystal X-Ray Structure of BeF2: α-Quartz" Inorg. Chem., 2011, 50 (1), pp 86–89. doi:10.1021/ic101248g
- Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
- Vapor pressure, physics.nyu.edu, p. 6-63, from Ohe, S. (1976) Computer Aided Data Book of Vapor Pressure, Data Book Publishing Co., Tokyo.
- Agarwal, M.; Chakravarty C (2007). "Waterlike Structural and Excess Entropy Anomalies in Liquid Beryllium Fluoride". J. Phys. Chem. B. 111 (46): 13294–300. doi:10.1021/jp0753272. PMID 17963376.
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- Reiko Kagawa; Martin G. Montgomery; Kerstin Braig; Andrew G. W. Leslie; John E. Walker (2004). "The structure of bovine F1-ATPase inhibited by ADP and beryllium fluoride". The EMBO Journal. 23 (5): 2734–2744. doi:10.1038/sj.emboj.7600293. PMC 514953. PMID 15229653.
- Bigay J.; Deterre P.; Pfister C.; Chabre M. (1987). "Fluoride complexes of aluminium or beryllium act on G-proteins as reversibly bound analogues of the gamma phosphate of GTP". The EMBO Journal. 6 (10): 2907–2913. doi:10.1002/j.1460-2075.1987.tb02594.x. PMC 553725. PMID 2826123.
- Fluoride in Drinking Water: A Scientific Review of EPA's Standards. The National Academies Press. 2006. pp. 51–52. doi:10.17226/11571. ISBN 978-0-309-10128-8.