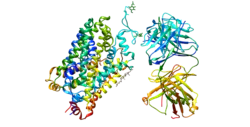
Serotonin transporter
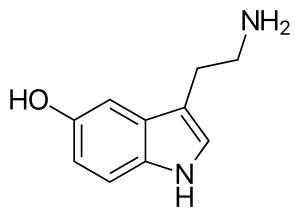
The serotonin transporter (SERT or 5-HTT) also known as the sodium-dependent serotonin transporter and solute carrier family 6 member 4 is a protein that in humans is encoded by the SLC6A4 gene.[5] SERT is a type of monoamine transporter protein that transports the neurotransmitter serotonin from the synaptic cleft back to the presynaptic neuron, in a process known as serotonin reuptake.[6]
This transport of serotonin by the SERT protein terminates the action of serotonin and recycles it in a sodium-dependent manner. Many antidepressant medications of the SSRI and tricyclic antidepressant classes work by binding to SERT and thus reducing serotonin reuptake.[7] It is a member of the sodium:neurotransmitter symporter family. A repeat length polymorphism in the promoter of this gene has been shown to affect the rate of serotonin uptake and may play a role in sudden infant death syndrome, aggressive behavior in Alzheimer disease patients, post-traumatic stress disorder and depression-susceptibility in people experiencing emotional trauma.[8]
Mechanism of action
Serotonin-reuptake transporters are dependent on both the concentration of potassium ion in the cytoplasm and the concentrations of sodium and chloride ions in the extracellular fluid. In order to function properly the serotonin transporter requires the membrane potential created by the sodium-potassium adenosine triphosphatase.
The serotonin transporter first binds a sodium ion, followed by the serotonin, and then a chloride ion; it is then allowed, thanks to the membrane potential, to flip inside the cell freeing all the elements previously bound. Right after the release of the serotonin in the cytoplasm a potassium ion binds to the transporter which is now able to flip back out returning to its active state.[9]
Function
The serotonin transporter removes serotonin from the synaptic cleft back into the synaptic boutons. Thus, it terminates the effects of serotonin and simultaneously enables its reuse by the presynaptic neuron.[7]
Neurons communicate by using chemical messengers like serotonin between cells. The transporter protein, by recycling serotonin, regulates its concentration in a gap, or synapse, and thus its effects on a receiving neuron's receptors.
Medical studies have shown that changes in serotonin transporter metabolism appear to be associated with many different phenomena, including alcoholism, clinical depression, obsessive–compulsive disorder (OCD), romantic love,[10] hypertension and generalized social phobia.[11]
The serotonin transporter is also present in platelets; there, serotonin functions as a vasoconstrictive substance. It also serves as a signalling molecule to induce platelet aggregation.
Pharmacology
In 1995 and 1996, scientists in Europe had identified the polymorphism 5-HTTLPR, a serotonin-transporter in the gene SLC6A4.[12][13] In December 1996, a group of researchers led by D.A. Collier of the Institute of Psychiatry, Psychology and Neuroscience, published their findings in Molecular Psychiatry, that, "5-HTTLPR-dependent variation in functional 5-HTT expression is a potential genetic susceptibility factor for affective disorders."[14]
SERT spans the plasma membrane 12 times. It belongs to the NE, DA, SERT monoamine transporter family. Transporters are important sites for agents that treat psychiatric disorders. Drugs that reduce the binding of serotonin to transporters (serotonin reuptake inhibitors, or SRIs) are used to treat mental disorders. The selective serotonin reuptake inhibitor (SSRI) fluoxetine and the tricyclic antidepressant (TCA) clomipramine are examples of serotonin reuptake inhibitors.
Following the elucidation of structures of the homologous bacterial transporter, LeuT, co-crystallized with tricyclic antidepressants in the vestibule leading from the extracellular space to the central substrate site it was inferred that this binding site did also represent the binding site relevant for antidepressant binding in SERT.[15] However, studies on SERT showed that tricyclic antidepressants and selective serotonin reuptake inhibitors bind to the central binding site overlapping the substrate binding site.[16][17][18] The Drosophila dopamine transporter, which displays a pharmacology similar to SERT, was crystallized with tricyclic antidepressants and confirmed the earlier finding that the substrate binding site is also the antidepressant binding site.[19]
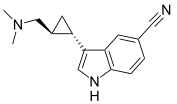
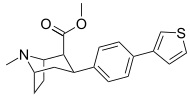
Ligands
- DASB
- compound 4b: Ki = 17 pM; 710-fold and 11,100-fold selective over DAT and NET[20]
- compound (+)-12a: Ki = 180 pM at hSERT; >1000-fold selective over hDAT, hNET, 5-HT1A, and 5-HT6.[21] Isosteres[22]
- 3-cis-(3-Aminocyclopentyl)indole 8a: Ki = 220 pM[23]
- allosteric modulator: 3′-Methoxy-8-methyl-spiro{8-azabicyclo[3.2.1]octane-3,5′(4′H)-isoxazole} (compound 7a)[24]
- allosteric modulator: p-Trifluoromethyl-methcathinone[25]
Genetics

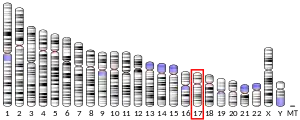
The gene that encodes the serotonin transporter is called solute carrier family 6 (neurotransmitter transporter, serotonin), member 4 (SLC6A4, see Solute carrier family). In humans the gene is found on chromosome 17 on location 17q11.1–q12.[27]
Mutations associated with the gene may result in changes in serotonin transporter function, and experiments with mice have identified more than 50 different phenotypic changes as a result of genetic variation. These phenotypic changes may, e.g., be increased anxiety and gut dysfunction.[28] Some of the human genetic variations associated with the gene are:[28]
- Length variation in the serotonin-transporter-gene-linked polymorphic region (5-HTTLPR)
- rs25531 — a single nucleotide polymorphism (SNP) in the 5-HTTLPR
- rs25532 — another SNP in the 5-HTTLPR
- STin2 — a variable number of tandem repeats (VNTR) in the functional intron 2
- G56A on the second exon
- I425V on the ninth exon
Length variation in 5-HTTLPR
According to a 1996 article in The Journal of Neurochemistry, the promoter region of the SLC6A4 gene contains a polymorphism with "short" and "long" repeats in a region: 5-HTT-linked polymorphic region (5-HTTLPR or SERTPR).[29] The short variation has 14 repeats of a sequence while the long variation has 16 repeats.[27] A second 1996 article stated that the short variation leads to less transcription for SLC6A4, and it has been found that it can partly account for anxiety-related personality traits.[30] This polymorphism has been extensively investigated in over 300 scientific studies (as of 2006).[31] The 5-HTTLPR polymorphism may be subdivided further: One study published in 2000 found 14 allelic variants (14-A, 14-B, 14-C, 14-D, 15, 16-A, 16-B, 16-C, 16-D, 16-E, 16-F, 19, 20 and 22) in a group of around 200 Japanese and Caucasian people.[27]
In addition to altering the expression of SERT protein and concentrations of extracellular serotonin in the brain, the 5-HTTLPR variation is associated with changes in brain structure. One 2005 study found less grey matter in perigenual anterior cingulate cortex and amygdala for short allele carriers of the 5-HTTLPR polymorphism compared to subjects with the long/long genotype.[32]
In contrast, a 2008 meta-analysis found no significant overall association between the 5-HTTLPR polymorphism and autism.[33] A hypothesized gene–environment interaction between the short/short allele of the 5-HTTLPR and life stress as predictor for major depression has suffered a similar fate: after an influential[34] initial report in 2003[35] there were mixed results in replication in 2008,[36] and a 2009 meta-analysis was negative.[37] See 5-HTTLPR for more information.
rs25532
rs25532 is a SNP (C>T) close to the site of 5-HTTLPR. It has been examined in connection with obsessive compulsive disorder (OCD).[38]
I425V
I425V is a rare mutation on the ninth exon. In 2003, researchers from Japan and the US reported that they had found this genetic variation in unrelated families with OCD, and have found that it leads to faulty transporter function and regulation. A second variant in the same gene of some patients with this mutation suggests a genetic "double hit", resulting in greater biochemical effects and more severe symptoms.[39][40][41]
VNTR in STin2
Another noncoding polymorphism is a VNTR in the second intron (STin2). In a 2005 study, it was found with three alleles: 9, 10 and 12 repeats. A meta-analysis has found that the 12 repeat allele of the STin2 VNTR polymorphism had some minor (with odds ratio 1.24), but statistically significant, association with schizophrenia.[42] A 2008 meta-analysis found no significant overall association between the STin2 VNTR polymorphism and autism.[33] Furthermore, a 2003 meta-analysis of affective disorders, major depressive disorder and bipolar disorder, found a minor association to the intron 2 VNTR polymorphism, but the results of the meta-analysis were dependent upon a large effect from one individual study.[43]
The polymorphism has also been related to personality traits with a 2008 Russian study finding individuals with the STin2.10 allele having lower neuroticism scores as measured with the Eysenck Personality Inventory.[44]
Neuroimaging
The distribution of the serotonin transporter in the brain may be imaged with positron emission tomography using radioligands called DASB and DAPP; the first such studies on the human brain were reported in 2000.[45] DASB and DAPP are not the only radioligands for the serotonin transporter. There are numerous others, with the most popular probably being the β-CIT radioligand with an iodine-123 isotope that is used for brain scanning with single-photon emission computed tomography (SPECT) according to a 1993 article in the Journal of Neural Transmission.[46] The radioligands were used in 2006 to examine whether variables such as age, gender or genotype are associated with differential serotonin transporter binding.[47] Healthy subjects that have a high score of neuroticism—a personality trait in the Revised NEO Personality Inventory—were found to have more serotonin transporter binding in the thalamus in 2007.[48]
Neuroimaging and genetics
Studies on the serotonin transporter have combined neuroimaging and genetics methods, e.g., a voxel-based morphometry study found less grey matter in perigenual anterior cingulate cortex and amygdala for short allele carriers of the 5-HTTLPR polymorphism compared to subjects with the long/long genotype.[32]
References
- GRCh38: Ensembl release 89: ENSG00000108576 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000020838 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "SLC6A4 Gene (Protein Coding)".
- "SLC6A4 - Sodium-dependent serotonin transporter - Homo sapiens (Human) - SLC6A4 gene & protein".
- Squire, Larry; et al., eds. (2008). Fundamental neuroscience (3rd ed.). Amsterdam: Elsevier / Academic Press. p. 143. ISBN 978-0-12-374019-9.
- "Entrez Gene: SLC6A4 solute carrier family 6 (neurotransmitter transporter, serotonin), member 4".
- "Mechanism of Action of the Serotonin Transporter". web.williams.edu. Retrieved 28 December 2016.
- Marazziti D, Akiskal HS, Rossi A, Cassano GB (May 1999). "Alteration of the platelet serotonin transporter in romantic love". Psychological Medicine. 29 (3): 741–5. doi:10.1017/S0033291798007946. PMID 10405096. S2CID 12630172.
- van der Wee NJ, van Veen JF, Stevens H, van Vliet IM, van Rijk PP, Westenberg HG (May 2008). "Increased serotonin and dopamine transporter binding in psychotropic medication-naive patients with generalized social anxiety disorder shown by 123I-beta-(4-iodophenyl)-tropane SPECT". Journal of Nuclear Medicine. 49 (5): 757–63. doi:10.2967/jnumed.107.045518. PMID 18413401.
- Heils A, Teufel A, Petri S, Seemann M, Bengel D, Balling U, Riederer P, Lesch KP (1995). "Functional promoter and polyadenylation site mapping of the human serotonin (5-HT) transporter gene". Journal of Neural Transmission. General Section. 102 (3): 247–54. doi:10.1007/BF01281159. PMID 8788073. S2CID 8474414.
- Heils A, Teufel A, Petri S, Stöber G, Riederer P, Bengel D, Lesch KP (June 1996). "Allelic variation of human serotonin transporter gene expression". Journal of Neurochemistry. 66 (6): 2621–4. doi:10.1046/j.1471-4159.1996.66062621.x. PMID 8632190. S2CID 42037860.
- Collier DA, Stöber G, Li T, Heils A, Catalano M, Di Bella D, Arranz MJ, Murray RM, Vallada HP, Bengel D, Müller CR, Roberts GW, Smeraldi E, Kirov G, Sham P, Lesch KP (December 1996). "A novel functional polymorphism within the promoter of the serotonin transporter gene: possible role in susceptibility to affective disorders". Molecular Psychiatry. 1 (6): 453–60. PMID 9154246.
- Zhou Z, Zhen J, Karpowich NK, Goetz RM, Law CJ, Reith ME, Wang DN (September 2007). "LeuT-desipramine structure reveals how antidepressants block neurotransmitter reuptake". Science. 317 (5843): 1390–3. Bibcode:2007Sci...317.1390Z. doi:10.1126/science.1147614. PMC 3711652. PMID 17690258.
- Sinning S, Musgaard M, Jensen M, Severinsen K, Celik L, Koldsø H, Meyer T, Bols M, Jensen HH, Schiøtt B, Wiborg O (March 2010). "Binding and orientation of tricyclic antidepressants within the central substrate site of the human serotonin transporter". The Journal of Biological Chemistry. 285 (11): 8363–74. doi:10.1074/jbc.M109.045401. PMC 2832986. PMID 19948720.
- Andersen J, Taboureau O, Hansen KB, Olsen L, Egebjerg J, Strømgaard K, Kristensen AS (April 2009). "Location of the antidepressant binding site in the serotonin transporter: importance of Ser-438 in recognition of citalopram and tricyclic antidepressants". The Journal of Biological Chemistry. 284 (15): 10276–84. doi:10.1074/jbc.M806907200. PMC 2665081. PMID 19213730.
- Koldsø H, Severinsen K, Tran TT, Celik L, Jensen HH, Wiborg O, Schiøtt B, Sinning S (February 2010). "The two enantiomers of citalopram bind to the human serotonin transporter in reversed orientations". Journal of the American Chemical Society. 132 (4): 1311–22. doi:10.1021/ja906923j. PMID 20055463.
- Penmatsa A, Wang KH, Gouaux E (November 2013). "X-ray structure of dopamine transporter elucidates antidepressant mechanism". Nature. 503 (7474): 85–90. Bibcode:2013Natur.503...85P. doi:10.1038/nature12533. PMC 3904663. PMID 24037379.
- Tamagnan G, Alagille D, Fu X, Kula NS, Baldessarini RJ, Innis RB, Baldwin RM (February 2005). "Synthesis and monoamine transporter affinity of new 2beta-carbomethoxy-3beta-[4-(substituted thiophenyl)]phenyltropanes: discovery of a selective SERT antagonist with picomolar potency". Bioorganic & Medicinal Chemistry Letters. 15 (4): 1131–3. doi:10.1016/j.bmcl.2004.12.014. PMID 15686927.
- Mattson RJ, Catt JD, Denhart DJ, Deskus JA, Ditta JL, Higgins MA, Marcin LR, Sloan CP, Beno BR, Gao Q, Cunningham MA, Mattson GK, Molski TF, Taber MT, Lodge NJ (September 2005). "Conformationally restricted homotryptamines. 2. Indole cyclopropylmethylamines as selective serotonin reuptake inhibitors". Journal of Medicinal Chemistry. 48 (19): 6023–34. doi:10.1021/jm0503291. PMID 16162005.
- Dalton King H, Denhart DJ, Deskus JA, Ditta JL, Epperson JR, Higgins MA, Kung JE, Marcin LR, Sloan CP, Mattson GK, Molski TF, Krause RG, Bertekap RL, Lodge NJ, Mattson RJ, Macor JE (October 2007). "Conformationally restricted homotryptamines. Part 4: Heterocyclic and naphthyl analogs of a potent selective serotonin reuptake inhibitor". Bioorganic & Medicinal Chemistry Letters. 17 (20): 5647–51. doi:10.1016/j.bmcl.2007.07.083. PMID 17766113.
- King HD, Meng Z, Deskus JA, Sloan CP, Gao Q, Beno BR, Kozlowski ES, Lapaglia MA, Mattson GK, Molski TF, Taber MT, Lodge NJ, Mattson RJ, Macor JE (November 2010). "Conformationally restricted homotryptamines. Part 7: 3-cis-(3-aminocyclopentyl)indoles as potent selective serotonin reuptake inhibitors". Journal of Medicinal Chemistry. 53 (21): 7564–72. doi:10.1021/jm100515z. PMID 20949929.
- Dallanoce C, Canovi M, Matera C, Mennini T, De Amici M, Gobbi M, De Micheli C (November 2012). "A novel spirocyclic tropanyl-Δ²-isoxazoline derivative enhances citalopram and paroxetine binding to serotonin transporters as well as serotonin uptake". Bioorganic & Medicinal Chemistry. 20 (21): 6344–55. doi:10.1016/j.bmc.2012.09.004. PMID 23022052.
- Niello M, Cintulova D, Hellsberg E, Jäntsch K, Holy M, Ayatollahi LH, Cozzi NV, Freissmuth M, Sandtner W, Ecker GF, Mihovilovic MD, Sitte HH (2019). "para-Trifluoromethyl-methcathinone is an allosteric modulator of the serotonin transporter". Neuropharmacology. 161: 107615. doi:10.1016/j.neuropharm.2019.04.021. PMID 31028773.
- Dahlin A, Royall J, Hohmann JG, Wang J (May 2009). "Expression profiling of the solute carrier gene family in the mouse brain". The Journal of Pharmacology and Experimental Therapeutics. 329 (2): 558–70. doi:10.1124/jpet.108.149831. PMC 2672879. PMID 19179540.
- Nakamura M, Ueno S, Sano A, Tanabe H (January 2000). "The human serotonin transporter gene linked polymorphism (5-HTTLPR) shows ten novel allelic variants". Molecular Psychiatry. 5 (1): 32–8. doi:10.1038/sj.mp.4000698. PMID 10673766.
- Murphy DL, Lesch KP (February 2008). "Targeting the murine serotonin transporter: insights into human neurobiology". Nature Reviews. Neuroscience. 9 (2): 85–96. doi:10.1038/nrn2284. PMID 18209729. S2CID 7563088.
- Heils A, Teufel A, Petri S, Stöber G, Riederer P, Bengel D, Lesch KP (June 1996). "Allelic variation of human serotonin transporter gene expression". Journal of Neurochemistry. 66 (6): 2621–4. doi:10.1046/j.1471-4159.1996.66062621.x. PMID 8632190. S2CID 42037860.
- Lesch KP, Bengel D, Heils A, Sabol SZ, Greenberg BD, Petri S, Benjamin J, Müller CR, Hamer DH, Murphy DL (November 1996). "Association of anxiety-related traits with a polymorphism in the serotonin transporter gene regulatory region". Science. 274 (5292): 1527–31. Bibcode:1996Sci...274.1527L. doi:10.1126/science.274.5292.1527. PMID 8929413. S2CID 35503987.
- Wendland JR, Martin BJ, Kruse MR, Lesch KP, Murphy DL (March 2006). "Simultaneous genotyping of four functional loci of human SLC6A4, with a reappraisal of 5-HTTLPR and rs25531". Molecular Psychiatry. 11 (3): 224–6. doi:10.1038/sj.mp.4001789. PMID 16402131.
- Pezawas L, Meyer-Lindenberg A, Drabant EM, Verchinski BA, Munoz KE, Kolachana BS, Egan MF, Mattay VS, Hariri AR, Weinberger DR (June 2005). "5-HTTLPR polymorphism impacts human cingulate-amygdala interactions: a genetic susceptibility mechanism for depression". Nature Neuroscience. 8 (6): 828–34. doi:10.1038/nn1463. PMID 15880108. S2CID 1864631.
- Huang CH, Santangelo SL (September 2008). "Autism and serotonin transporter gene polymorphisms: a systematic review and meta-analysis". American Journal of Medical Genetics Part B. 147B (6): 903–13. doi:10.1002/ajmg.b.30720. PMID 18286633. S2CID 9491697.
- Nierenberg AA (September 2009). "The long tale of the short arm of the promoter region for the gene that encodes the serotonin uptake protein". CNS Spectrums. 14 (9): 462–3. doi:10.1017/s1092852900023506. PMID 19890228. S2CID 24236284.
- Caspi A, Sugden K, Moffitt TE, Taylor A, Craig IW, Harrington H, McClay J, Mill J, Martin J, Braithwaite A, Poulton R (July 2003). "Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene". Science. 301 (5631): 386–9. Bibcode:2003Sci...301..386C. doi:10.1126/science.1083968. PMID 12869766. S2CID 146500484.
- Uher R, McGuffin P (February 2008). "The moderation by the serotonin transporter gene of environmental adversity in the aetiology of mental illness: review and methodological analysis". Molecular Psychiatry. 13 (2): 131–46. doi:10.1038/sj.mp.4002067. PMID 17700575.
- Risch N, Herrell R, Lehner T, Liang KY, Eaves L, Hoh J, Griem A, Kovacs M, Ott J, Merikangas KR (June 2009). "Interaction between the serotonin transporter gene (5-HTTLPR), stressful life events, and risk of depression: a meta-analysis". JAMA. 301 (23): 2462–71. doi:10.1001/jama.2009.878. PMC 2938776. PMID 19531786.
- Wendland JR, Moya PR, Kruse MR, Ren-Patterson RF, Jensen CL, Timpano KR, Murphy DL (March 2008). "A novel, putative gain-of-function haplotype at SLC6A4 associates with obsessive-compulsive disorder". Human Molecular Genetics. 17 (5): 717–23. doi:10.1093/hmg/ddm343. PMID 18055562.
- Ozaki N, Goldman D, Kaye WH, Plotnicov K, Greenberg BD, Lappalainen J, Rudnick G, Murphy DL (November 2003). "Serotonin transporter missense mutation associated with a complex neuropsychiatric phenotype". Molecular Psychiatry. 8 (11): 933–6. doi:10.1038/sj.mp.4001365. PMID 14593431.
News article:
- "Gene Found for Obsessive-Compulsive Disorder". Mental Health E-News. Reuters. 27 October 2003. Archived from the original on 6 October 2006. Retrieved 25 January 2008.
- Delorme R, Betancur C, Wagner M, Krebs MO, Gorwood P, Pearl P, Nygren G, Durand CM, Buhtz F, Pickering P, Melke J, Ruhrmann S, Anckarsäter H, Chabane N, Kipman A, Reck C, Millet B, Roy I, Mouren-Simeoni MC, Maier W, Råstam M, Gillberg C, Leboyer M, Bourgeron T (December 2005). "Support for the association between the rare functional variant I425V of the serotonin transporter gene and susceptibility to obsessive compulsive disorder". Molecular Psychiatry. 10 (12): 1059–61. doi:10.1038/sj.mp.4001728. PMC 2547479. PMID 16088327.
- Stephen Wheless. ""The OCD Gene" Popular Press v. Scientific Literature: Is SERT Responsible for Obsessive-Compulsive Disorder?". Davidson College. Retrieved 12 June 2008.
- Fan JB, Sklar P (October 2005). "Meta-analysis reveals association between serotonin transporter gene STin2 VNTR polymorphism and schizophrenia". Molecular Psychiatry. 10 (10): 928–38, 891. doi:10.1038/sj.mp.4001690. PMID 15940296.
- Anguelova M, Benkelfat C, Turecki G (June 2003). "A systematic review of association studies investigating genes coding for serotonin receptors and the serotonin transporter: I. Affective disorders". Molecular Psychiatry. 8 (6): 574–91. doi:10.1038/sj.mp.4001328. PMID 12851635.
- Kazantseva AV, Gaysina DA, Faskhutdinova GG, Noskova T, Malykh SB, Khusnutdinova EK (August 2008). "Polymorphisms of the serotonin transporter gene (5-HTTLPR, A/G SNP in 5-HTTLPR, and STin2 VNTR) and their relation to personality traits in healthy individuals from Russia". Psychiatric Genetics. 18 (4): 167–76. doi:10.1097/YPG.0b013e328304deb8. PMID 18628678. S2CID 7423923.
- Houle S, Ginovart N, Hussey D, Meyer JH, Wilson AA (November 2000). "Imaging the serotonin transporter with positron emission tomography: initial human studies with [11C]DAPP and [11C]DASB". European Journal of Nuclear Medicine. 27 (11): 1719–22. doi:10.1007/s002590000365. PMID 11105830. S2CID 18932686.
- Brücke T, Kornhuber J, Angelberger P, Asenbaum S, Frassine H, Podreka I (1993). "SPECT imaging of dopamine and serotonin transporters with [123I]beta-CIT. Binding kinetics in the human brain". Journal of Neural Transmission. General Section. 94 (2): 137–46. doi:10.1007/BF01245007. PMID 8110440. S2CID 22034290.
- Brust P, Hesse S, Muller U, Szabo Z (February 2006). "Neuroimaging of the Serotonin Transporter — Possibilities and Pitfalls" (PDF). Current Psychiatry Reviews. 2 (1): 111–149. doi:10.2174/157340006775101508. Archived from the original (PDF) on 23 July 2011.
- Takano A, Arakawa R, Hayashi M, Takahashi H, Ito H, Suhara T (September 2007). "Relationship between neuroticism personality trait and serotonin transporter binding". Biological Psychiatry. 62 (6): 588–92. doi:10.1016/j.biopsych.2006.11.007. PMID 17336939. S2CID 41664835.