Mediator (coactivator)
Mediator is a multiprotein complex that functions as a transcriptional coactivator in all eukaryotes. It was discovered in 1990 in the lab of Roger D. Kornberg, recipient of the 2006 Nobel Prize in Chemistry.[1][2] Mediator[lower-alpha 1] complexes interact with transcription factors and RNA polymerase II. The main function of mediator complexes is to transmit signals from the transcription factors to the polymerase.[3]
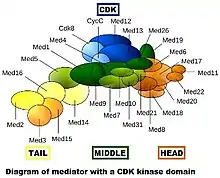
Mediator complexes are variable at the evolutionary, compositional and conformational levels.[3] The first image shows only one "snapshot" of what a particular mediator complex might be composed of,[lower-alpha 2] but it certainly does not accurately depict the conformation of the complex in vivo. During evolution, mediator has become more complex. The yeast Saccharomyces cerevisiae (a simple eukaryote) is thought to have up to 21 subunits in the core mediator (exclusive of the CDK module), while mammals have up to 26.
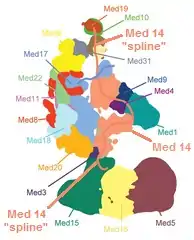
Individual subunits can be absent or replaced by other subunits under different conditions. Also, there are many intrinsically disordered regions in mediator proteins, which may contribute to the conformational flexibility seen both with and without other bound proteins or protein complexes. A more realistic model of a mediator complex without the CDK module is shown in the second figure.[4]
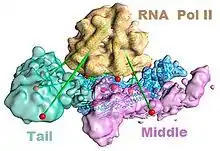
The mediator complex is required for the successful transcription by RNA polymerase II. Mediator has been shown to make contacts with the polymerase in the transcription preinitiation complex.[3] A recent model showing the association of the polymerase with mediator in the absence of DNA is shown in the figure to the left.[4] In addition to RNA polymerase II, mediator must also associate with transcription factors and DNA. A model of such interactions is shown in the figure to the right.[5] Note that the different morphologies of mediator do not necessarily mean that one of the models is correct; rather those differences may reflect the flexibility of mediator as it interacts with other molecules.[lower-alpha 3] For example, after binding the enhancer and core promoter, the mediator complex undergoes a compositional change in which the kinase module dissociates from the complex to allow association with RNA polymerase II and transcriptional activation.[6]
The Mediator complex is located within the cell nucleus. It is required for the successful transcription of nearly all class II gene promoters in yeast.[7] It works in the same manner in mammals. The mediator functions as a coactivator and binds to the C-terminal domain of RNA polymerase II holoenzyme, acting as a bridge between this enzyme and transcription factors.[8]
Structure
The yeast mediator complex is approximately as massive as a small subunit of a eukaryotic ribosome. The yeast mediator is composed of 25 subunits, while the mammalian mediator complexes are slightly larger.[3] Mediator can be divided into 4 main parts: The head, middle, tail, and the transiently associated CDK8 kinase module.[10]
Mediator subunits have many intrinsically disordered regions called "splines", which may be important to allow the structural changes of the mediator that change the function of the complex.[3][lower-alpha 4] The figure shows how the splines of the Med 14 subunit connect a large portion of the complex together while still allowing flexibility.[4][lower-alpha 5]
Mediator complexes that lack a subunit have been found or produced. These smaller mediators can still function normally in some activity, but lack other capabilities.[3] This indicates a somewhat independent function of some of the subunits while being part of the larger complex.
Another example of structural variability is seen in vertebrates, in which 3 paralogues of subunits of the cyclin-dependent kinase module have evolved by 3 independent gene duplication events followed by sequence divergence.[3]

There is a report that mediator forms stable associations with a particular type of non-coding RNA, ncRNA-a.[11][lower-alpha 6] These stable associations have also been shown to regulate gene expression in vivo, and are prevented by mutations in MED12 that produce the human disease FG syndrome.[11] Thus, the structure of a mediator complex can be augmented by RNA as well as proteinaceous transcription factors.[3]
Function
Mediator was originally discovered because it was important for RNA polymerase II function, but it has many more functions than just interactions at the transcription start site.[3]
RNA polymerase II-Mediator core initiation complex
Mediator is a crucial component for transcription initiation. Mediator interacts with the pre-initiation complex, composed of RNA Polymerase II and general transcription factors TFIIB, TFIID, TFIIE, TFIIF, and TFIIH to stabilize and initiate transcription.[12] Studies of Mediator-RNA Pol II contacts in budding yeast have emphasized the importance of TFIIB-Mediator contacts in the formation of the complex. Interactions of Mediator with TFIID in the initiation complex has been shown.[10]
The Structure of a core Mediator (cMed) that's associated with a core pre-initiation complex was elucidated.[12]
RNA synthesis
The preinitiation complex, which contains mediator, transcription factors, a nucleosome[13][14][lower-alpha 7] and RNA polymerase II, is important to position the polymerase for the start of transcription. Before RNA synthesis can occur, the polymerase must dissociate from mediator. This appears to be accomplished by phosphorylation of part of the polymerase by a kinase. Importantly, mediator and transcription factors do not dissociate from the DNA at the time polymerase begins transcription. Rather, the complex remains at the promoter to recruit another RNA polymerase to begin another round of transcription.[3][lower-alpha 8]
There is some evidence to suggest that mediator in a yeast is involved in regulating RNA polymerase III (Pol III) transcripts of tRNAs[15] In support of that evidence, an independent report showed specific association of mediator with Pol III in Saccharomyces cerevisiae.[16] Those authors also reported specific associations with RNA polymerase I and proteins involved in transcription elongation and RNA processing, supporting other evidence of mediator's involvement in elongation and processing.[16]
Chromatin organization
Mediator is involved in "looping" of chromatin, which brings distant regions of a chromosome into closer physical proximity.[3] The ncRNA-a mentioned above[11] is involved in such looping.[lower-alpha 9] Enhancer RNAs (eRNAs) can function similarly.[3]
In addition to the looping of euchromatin, mediator appears to be involved in formation or maintenance of heterochromatin at centromeres and telomeres.[3]
Signal transduction
TGFβ signaling at the cell membrane results in 2 different intracellular pathways. One of them depends on MED15,[lower-alpha 10] while the other is independent of MED15.[17] In both human cells and Caenorhabditis elegans MED15 is involved in lipid homeostasis through the pathway involving SREBPs[18] In the model plant Arabidopsis thaliana the ortholog of MED15 is required for signaling by the plant hormone Salicylic acid,[19] while MED25 is required for the transcriptional activation of jasmonate and shade signalling responses.[20][21][22] Two components of the CDK module (MED12 and MED13) are involved in the Wnt signaling pathway[3] MED23 is involved in RAS/MAPK/ERK pathway[3] This abbreviated review shows the versatility of individual mediator subunits, and leads to the idea that mediator is an end-point of signaling pathways.[3]
Human disease
Involvement of mediator in various human diseases has been reviewed.[23][24][25][26][27][28][29][30][31][32][33] Since inhibiting one interaction of a disease-causing signaling pathway with a subunit of mediator may not inhibit general transcription needed for normal function, mediator subunits are attractive candidates for therapeutic drugs.[3]
Interactions

A method employing very gentle cell lysis in yeast followed by co-immunoprecipitation with an antibody to a mediator subunit (Med 17) has confirmed almost all previously reported or predicted interactions and revealed many previously unsuspected specific interactions of various proteins with mediator.[16]
MED 1
A discussion of all mediator subunits is beyond the scope of this article, but details of one of the subunits is illustrative of the types of information that may be gathered for other subunits.
regulation by Micro RNAs
Micro RNAs are involved in regulating the expression of many proteins. Med1 is targeted by miR-1, which is important in gene regulation in cancers.[34] The tumor suppressor miR-137 also regulates MED1.[35]
Mouse embryonic development
Null mutants die at an early gestational age (embryonic day 11.5).[36][37] By investigating hypomorphic mutants (which can survive 2 days longer), it was found that placental defects were primarily lethal and that there were also defects in cardiac and hepatic development, but many other organs were normal[37]
Mouse cells and tissues
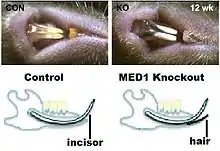
Conditional mutations can be produced in mice which affect only specific cells or tissues at specific times, so that the mouse can develop to adulthood and the adult phenotype can be studied. In one case, MED1 was found to participate in controlling the timing of events of meiosis in male mice.[38] Conditional mutants in keratinocytes show differences in skin wound healing.[39] A conditional mutant in mice was found to change dental epithelium into epidermal epithelium, which caused hair to grow associated with the incisors.[40]
Subunit composition
The Mediator complex is composed at least 31 subunits in all eukaryotes studied: MED1, MED4, MED6, MED7, MED8, MED9, MED10, MED11, MED12, MED13, MED13L, MED14, MED15, MED16, MED17, MED18, MED19, MED20, MED21, MED22, MED23, MED24, MED25, MED26, MED27, MED28, MED29, MED30, MED31, CCNC, and CDK8. There are three fungal-specific components, referred to as Med2, Med3 and Med5.[41]
The subunits form at least three structurally distinct submodules. The head and the middle modules interact directly with RNA polymerase II, whereas the elongated tail module interacts with gene-specific regulatory proteins. Mediator containing the CDK8 module is less active than Mediator lacking this module in supporting transcriptional activation.
- The head module contains: MED6, MED8, MED11, SRB4/MED17, SRB5/MED18, ROX3/MED19, SRB2/MED20 and SRB6/MED22.
- The middle module contains: MED1, MED4, NUT1/MED5, MED7, CSE2/MED9, NUT2/MED10, SRB7/MED21 and SOH1/MED31. CSE2/MED9 interacts directly with MED4.
- The tail module contains: MED2, PGD1/MED3, RGR1/MED14, GAL11/MED15 and SIN4/MED16.
- The CDK8 module contains: MED12, MED13, CCNC and CDK8. Individual preparations of the Mediator complex lacking one or more distinct subunits have been variously termed ARC, CRSP, DRIP, PC2, SMCC and TRAP.
In other species
Below is a cross-species comparison of mediator complex subunits.[41][42]
| Subunit No. | Human gene | C. elegans gene | D. melanogaster gene | S. cerevisiae gene | Sch. pombe gene |
|---|---|---|---|---|---|
| MED1 | MED1 | Sop3/mdt-1.1, 1.2 | MED1 | MED1 | med1 |
| Med2 [lower-alpha 11] | MED2 | ||||
| Med3 [lower-alpha 11] | PGD1 | ||||
| MED4 | MED4 | MED4 | MED4 | med4 | |
| Med5 [lower-alpha 11] | NUT1 | ||||
| MED6 | MED6 | MDT-6 | MED6 | MED6 | med6 |
| MED7 | MED7 | MDT-7/let-49 | MED7 | MED7 | med7 |
| MED8 | MED8 | MDT-8 | MED8 | MED8 | med8 |
| MED9 | MED9 | MED9 | CSE2 | ||
| MED10 | MED10 | MDT-10 | NUT2 | med10 | |
| MED11 | MED11 | MDT-11 | MED11 | MED11 | med11 |
| MED12 | MED12 | MDT-12/dpy-22 | MED12 | SRB8 | srb8 |
| MED12L | MED12L | ||||
| MED13 | MED13 | MDT-13/let-19 | MED13 | SSN2 | srb9 |
| MED14 | MED14 | MDT-14/rgr-1 | MED14 | RGR1 | med14 |
| MED15 | MED15 | mdt-15 | MED15 | GAL11 | YN91_SCHPO [lower-alpha 12] |
| MED16 | MED16 | MED16 | SIN4 | ||
| MED17 | MED17 | MDT-17 | MED17 | SRB4 | med17 |
| MED18 | MED18 | MDT-18 | MED18 | SRB5 | med18 |
| MED19 | MED19 | MDT-19 | MED19 | ROX3[41] | med19 |
| MED20 | MED20 | MDT-20 | MED20 | SRB2 | med20 |
| MED21 | MED21 | MDT-21 | MED21 | SRB7 | med21 |
| MED22 | MED22 | MDT-22 | MED22 | SRB6 | med22 |
| MED23 | MED23 | MDT-23/sur-2 | MED23 | ||
| MED24 | MED24 | MED24 | |||
| MED25 | MED25 | MED25 | |||
| MED26 | MED26 | MED26 | |||
| MED27 | MED27 | MED27 | med27 | ||
| MED28 | MED28 | MED28 | |||
| MED29 | MED29 | MDT-19 | MED29 | ||
| MED30 | MED30 | MED30 | |||
| MED31 | MED31 | MDT-31 | MED31 | SOH1 | med31 |
| CCNC | CCNC | cic-1 | CycC | SSN8 | pch1 |
| CDK8 | CDK8 | cdk-8 | Cdk8 | SSN3 | srb10 |
Notes
- Mediator is also referred to in scientific literature as the vitamin D receptor interacting protein (DRIP) coactivator complex and the thyroid hormone receptor-associated proteins (TRAP).
- However note that more recently it has been found that the CDK module and MED26 cannot be present concurrently in a complex.[3]
- The sharp bend in the DNA associated with the transcription bubble is shown in the graphical abstract and first figure of this research paper
- Some of those changes are diagrammed in figure 1 of the review article, which can be viewed in slightly larger form by clicking it at that site.
- Note that Med 17 (shown in blue) also has that sort of spline
- These non-coding activating RNAs have not been mentioned yet in the ncRNA article as of 16 February 2017
- This is the +1 nucleosome, which "covers" the transcription start site during the preinitiation phase.
- This is diagrammed in figure 2 of the review article, which can be viewed in slightly larger form by clicking it at that site.
- This is diagrammed in figure 3 of the review article, which can be viewed in slightly larger form by clicking it at that site. That figure also shows Pol II disengaged from mediator, etc, which remains on the DNA
- Also known as ARC105 in Xenopus laevis, the model species in which the work was done.
- Fungal-specific
- Protein-name in Sch. pombe
References
- Kelleher RJ, Flanagan PM, Kornberg RD (June 1990). "A novel mediator between activator proteins and the RNA polymerase II transcription apparatus". Cell. 61 (7): 1209–15. doi:10.1016/0092-8674(90)90685-8. PMID 2163759. S2CID 4971987.
- Flanagan PM, Kelleher RJ, Sayre MH, Tschochner H, Kornberg RD (April 1991). "A mediator required for activation of RNA polymerase II transcription in vitro". Nature. 350 (6317): 436–8. Bibcode:1991Natur.350..436F. doi:10.1038/350436a0. PMID 2011193. S2CID 4323957.
- Allen BL, Taatjes DJ (March 2015). "The Mediator complex: a central integrator of transcription". Nature Reviews Molecular Cell Biology. 16 (3): 155–66. doi:10.1038/nrm3951. PMC 4963239. PMID 25693131.
- Robinson PJ, Trnka MJ, Pellarin R, Greenberg CH, Bushnell DA, Davis R, Burlingame AL, Sali A, Kornberg RD (September 2015). "Molecular architecture of the yeast Mediator complex". eLife. 4: e08719. doi:10.7554/eLife.08719. PMC 4631838. PMID 26402457.
- Bernecky C, Grob P, Ebmeier CC, Nogales E, Taatjes DJ (March 2011). "Molecular architecture of the human Mediator-RNA polymerase II-TFIIF assembly". PLOS Biology. 9 (3): e1000603. doi:10.1371/journal.pbio.1000603. PMC 3066130. PMID 21468301.
- Petrenko, N; Jin, Y; Wong, KH; Struhl, K (3 November 2016). "Mediator Undergoes a Compositional Change during Transcriptional Activation". Molecular Cell. 64 (3): 443–454. doi:10.1016/j.molcel.2016.09.015. PMC 5096951. PMID 27773675.
- Biddick R, Young ET (September 2005). "Yeast mediator and its role in transcriptional regulation". Comptes Rendus Biologies. 328 (9): 773–82. doi:10.1016/j.crvi.2005.03.004. PMID 16168358.
- Björklund S, Gustafsson CM (May 2005). "The yeast Mediator complex and its regulation". Trends in Biochemical Sciences. 30 (5): 240–4. doi:10.1016/j.tibs.2005.03.008. PMID 15896741.
- Robinson, Philip J.; Trnka, Michael J.; Pellarin, Riccardo; Greenberg, Charles H.; Bushnell, David A.; Davis, Ralph; Burlingame, Alma L.; Sali, Andrej; Kornberg, Roger D. (24 September 2015). "Molecular architecture of the yeast Mediator complex". eLife. 4: e08719. doi:10.7554/eLife.08719. ISSN 2050-084X. PMC 4631838. PMID 26402457.
- Soutourina, Julie (6 December 2017). "Transcription regulation by the Mediator complex". Nature Reviews Molecular Cell Biology. 19 (4): 262–274. doi:10.1038/nrm.2017.115. ISSN 1471-0072. PMID 29209056. S2CID 3972303.
- Lai, F; et al. (2013). "Activating RNAs associate with mediator to enhance chromatin architecture and transcription" (PDF). Nature. 494 (7438): 497–501. Bibcode:2013Natur.494..497L. doi:10.1038/nature11884. hdl:11858/00-001M-0000-0019-1122-4. PMC 4109059. PMID 23417068.
- Plaschka, C.; Larivière, L.; Wenzeck, L.; Seizl, M.; Hemann, M.; Tegunov, D.; Petrotchenko, E. V.; Borchers, C. H.; Baumeister, W. (February 2015). "Architecture of the RNA polymerase II–Mediator core initiation complex". Nature. 518 (7539): 376–380. Bibcode:2015Natur.518..376P. doi:10.1038/nature14229. ISSN 0028-0836. PMID 25652824. S2CID 4450934.
- Nagai S, Davis RE, Mattei PJ, Eagen KP, Kornberg RD (2017). "Chromatin potentiates transcription". Proc Natl Acad Sci U S A. 114 (7): 1536–154. doi:10.1073/pnas.1620312114. PMC 5320956. PMID 28137832.
- Kornberg, RD. "The Molecular Basis of Eukaryotic Transcription". YouTube. Israel Institute for Advanced Studies. Archived from the original on 15 December 2021. Retrieved 17 February 2017.
- Carlsten, JO; Zhu X, López MD, Samuelsson T, Gustafsson CM (February 2016). "Loss of the Mediator subunit Med20 affects transcription of tRNA and other non-coding RNA genes in fission yeast". Biochimica et Biophysica Acta (BBA) - Gene Regulatory Mechanisms. 1859 (2): 339–347. doi:10.1016/j.bbagrm.2015.11.007. PMID 26608234.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Uthe H, Vanselow JT, Schlosser A (2017). "Proteomic Analysis of the Mediator Complex Interactome in Saccharomyces cerevisiae". Sci Rep. 7: 43584. Bibcode:2017NatSR...743584U. doi:10.1038/srep43584. PMC 5327418. PMID 28240253.
- Kato Y, Habas R, Katsuyama Y, Näär AM, He X (2002). "A component of the ARC/Mediator complex required for TGF beta/Nodal signalling". Nature. 418 (6898): 641–6. Bibcode:2002Natur.418..641K. doi:10.1038/nature00969. PMID 12167862. S2CID 4330754.
- Yang F, Vought BW, Satterlee JS, Walker AK, Jim Sun ZY, Watts JL, DeBeaumont R, Saito RM, Hyberts SG, Yang S, Macol C, Iyer L, Tjian R, van den Heuvel S, Hart AC, Wagner G, Näär AM (2006). "An ARC/Mediator subunit required for SREBP control of cholesterol and lipid homeostasis". Nature. 442 (7103): 700–4. Bibcode:2006Natur.442..700Y. doi:10.1038/nature04942. PMID 16799563. S2CID 4396081.
- Canet JV, Dobón A, Tornero P (2012). "Non-recognition-of-BTH4, an Arabidopsis mediator subunit homolog, is necessary for development and response to salicylic acid". Plant Cell. 24 (10): 4220–35. doi:10.1105/tpc.112.103028. PMC 3517246. PMID 23064321.
- Chen, Rong; Jiang, Hongling; Li, Lin; Zhai, Qingzhe; Qi, Linlin; Zhou, Wenkun; Liu, Xiaoqiang; Li, Hongmei; Zheng, Wenguang; Sun, Jiaqiang; Li, Chuanyou (July 2012). "The Arabidopsis Mediator Subunit MED25 Differentially Regulates Jasmonate and Abscisic Acid Signaling through Interacting with the MYC2 and ABI5 Transcription Factors". The Plant Cell. 24 (7): 2898–2916. doi:10.1105/tpc.112.098277. PMC 3426122. PMID 22822206.
- Sun, Wenjing; Han, Hongyu; Deng, Lei; Sun, Chuanlong; Xu, Yiran; Lin, Lihao; Ren, Panrong; Zhao, Jiuhai; Zhai, Qingzhe; Li, Chuanyou (November 2020). "Mediator Subunit MED25 Physically Interacts with PHYTOCHROME INTERACTING FACTOR4 to Regulate Shade-Induced Hypocotyl Elongation in Tomato". Plant Physiology. 184 (3): 1549–1562. doi:10.1104/pp.20.00587. PMC 7608172. PMID 32938743.
- Hartman, Sjon (November 2020). "MED25 Mediates Shade-Induced Hypocotyl Elongation in Tomato". Plant Physiology. 184 (3): 1217–1218. doi:10.1104/pp.20.01324. PMC 7608160. PMID 33139486.
- Clark AD, Oldenbroek M, Boyer TG (2015). "Mediator kinase module and human tumorigenesis". Crit Rev Biochem Mol Biol. 50 (5): 393–426. doi:10.3109/10409238.2015.1064854 (inactive 1 August 2023). PMC 4928375. PMID 26182352.
{{cite journal}}: CS1 maint: DOI inactive as of August 2023 (link) - Croce S, Chibon F (2015). "MED12 and uterine smooth muscle oncogenesis: State of the art and perspectives". Eur J Cancer. 51 (12): 1603–10. doi:10.1016/j.ejca.2015.04.023. PMID 26037152.
- Schiano C, Casamassimi A, Rienzo M, de Nigris F, Sommese L, Napoli C (2014). "Involvement of Mediator complex in malignancy". Biochim Biophys Acta. 1845 (1): 66–83. doi:10.1016/j.bbcan.2013.12.001. PMID 24342527.
- Schiano C, Casamassimi A, Vietri MT, Rienzo M, Napoli C (2014). "The roles of mediator complex in cardiovascular diseases". Biochim Biophys Acta. 1839 (6): 444–51. doi:10.1016/j.bbagrm.2014.04.012. PMID 24751643.
- Utami KH, Winata CL, Hillmer AM, Aksoy I, Long HT, Liany H, Chew EG, Mathavan S, Tay SK, Korzh V, Sarda P, Davila S, Cacheux V (2014). "Impaired development of neural-crest cell-derived organs and intellectual disability caused by MED13L haploinsufficiency". Hum Mutat. 35 (11): 1311–20. doi:10.1002/humu.22636. PMID 25137640. S2CID 42336634.
- Grueter CE (2013). "Mediator complex dependent regulation of cardiac development and disease". Genomics Proteomics Bioinformatics. 11 (3): 151–7. doi:10.1016/j.gpb.2013.05.002. PMC 4357813. PMID 23727265.
- Yang X and Yang F (2013). "Mediating Lipid Biosynthesis: Implications for Cardiovascular Disease". Trends Cardiovasc Med. 23 (7): 269–273. doi:10.1016/j.tcm.2013.03.002. PMC 3744615. PMID 23562092.
- Napoli C, Sessa M, Infante T, Casamassimi A (2012). "Unraveling framework of the ancestral Mediator complex in human diseases". Biochimie. 94 (3): 579–87. doi:10.1016/j.biochi.2011.09.016. PMID 21983542.
- Xu W, Ji JY (2011). "Dysregulation of CDK8 and Cyclin C in tumorigenesis". J Genet Genomics. 38 (10): 439–52. doi:10.1016/j.jgg.2011.09.002. PMC 9792140. PMID 22035865.
- Spaeth JM, Kim NH, Boyer TG (2011). "Mediator and human disease". Semin Cell Dev Biol. 22 (7): 776–87. doi:10.1016/j.semcdb.2011.07.024. PMC 4100472. PMID 21840410.
- Lyons MJ (2008). MED12-Related Disorders (08/11/2016 ed.). University of Washington, Seattle. PMID 20301719.
- Jiang C, Chen H, Shao L, Wang Q (2014). "MicroRNA-1 functions as a potential tumor suppressor in osteosarcoma by targeting Med1 and Med31". Oncol Rep. 32 (3): 1249–56. doi:10.3892/or.2014.3274. PMID 24969180.
- Nilsson EM, Laursen KB, Whitchurch J, McWilliam A, Ødum N, Persson JL, Heery DM, Gudas LJ, Mongan NP (2015). "MiR137 is an androgen regulated repressor of an extended network of transcriptional coregulators". Oncotarget. 6 (34): 35710–25. doi:10.18632/oncotarget.5958. PMC 4742136. PMID 26461474.
- Ito M, Yuan CX, Okano HJ, Darnell RB, Roeder RG (2000). "Involvement of the TRAP220 component of the TRAP/SMCC coactivator complex in embryonic development and thyroid hormone action". Mol Cell. 5 (4): 683–93. doi:10.1016/S1097-2765(00)80247-6. PMID 10882104.
- Landles C, Chalk S, Steel JH, Rosewell I, Spencer-Dene B, Lalani el-N, Parker MG (2003). "The thyroid hormone receptor-associated protein TRAP220 is required at distinct embryonic stages in placental, cardiac, and hepatic development". Mol Endocrinol. 17 (12): 2418–35. doi:10.1210/me.2003-0097. PMID 14500757.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Huszar JM, Jia Y, Reddy JK, Payne CJ (2015). "Med1 regulates meiotic progression during spermatogenesis in mice". Reproduction. 149 (6): 597–604. doi:10.1530/REP-14-0483. PMC 4417004. PMID 25778538.
- Noguchi F, Nakajima T, Inui S, Reddy JK, Itami S (2014). "Alteration of skin wound healing in keratinocyte-specific mediator complex subunit 1 null mice". PLOS ONE. 9 (8): e102271. Bibcode:2014PLoSO...9j2271N. doi:10.1371/journal.pone.0102271. PMC 4133190. PMID 25122137.
- Yoshizaki K, Hu L, Nguyen T, Sakai K, He B, Fong C, Yamada Y, Bikle DD, Oda Y (2014). "Ablation of coactivator Med1 switches the cell fate of dental epithelia to that generating hair". PLOS ONE. 9 (6): e99991. Bibcode:2014PLoSO...999991Y. doi:10.1371/journal.pone.0099991. PMC 4065011. PMID 24949995.
- Bourbon HM, Aguilera A, Ansari AZ, Asturias FJ, Berk AJ, Bjorklund S, et al. (2004). "A unified nomenclature for protein subunits of mediator complexes linking transcriptional regulators to RNA polymerase II". Molecular Cell. 14 (5): 553–7. doi:10.1016/j.molcel.2004.05.011. PMID 15175151.
- Gene names derived from "UniProtKB". Retrieved 12 October 2012.