Muraglitazar
Muraglitazar (proposed tradename Pargluva) is a dual peroxisome proliferator-activated receptor agonist with affinity to PPARα and PPARγ.[1]
 | |
| Clinical data | |
|---|---|
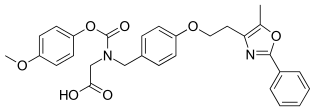
| Other names | 2-[(4-Methoxyphenoxy)carbonyl-[[4-[2-(5-methyl-2-phenyl-1,3-oxazol-4-yl)ethoxy]phenyl]methyl]amino]acetic acid |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C29H28N2O7 |
| Molar mass | 516.550 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
The drug had completed phase III clinical trials,[2] however in May, 2006 Bristol-Myers Squibb announced that it had discontinued further development.[3]
Data on muraglitazar is relatively sparse due to the recent introduction of this agent. One double-blind randomized clinical trial[2] comparing muraglitazar and pioglitazone found that the effects of the former were favourable in terms of HDL-C increase, decrease in total cholesterol, apolipoprotein B, triglycerides and a greater reduction in HbA1c (p <0.0001 for all comparisons). However, the muraglitazar group had a higher all-cause mortality, greater incidence of edema and heart failure and more weight gain compared to the pioglitazone group. A meta-analysis of the phase II and III clinical trials of muraglitazar revealed that it was associated with a greater incidence of myocardial infarction, stroke, transient ischemic attacks and congestive heart failure (CHF) when compared to placebo or pioglitazone.[4]
By calling attention to adverse events made public through the FDA advisory committee process, Dr Nissen came upon a mechanism to steer FDA from the outside. This mechanism came to fruition with rosiglitazone (Avandia)and led to FDA requiring demonstration of cardiac safety for new drugs to treat type 2 diabetes. This process is described by Dr Robert Misbin in INSULIN-History from an FDA Insider, published June 1, 2020 on Amazon.
References
- Waites CR, Dominick MA, Sanderson TP, Schilling BE (November 2007). "Nonclinical safety evaluation of muraglitazar, a novel PPARalpha/gamma agonist". Toxicological Sciences. 100 (1): 248–58. doi:10.1093/toxsci/kfm193. PMID 17675651.
- Kendall DM, Rubin CJ, Mohideen P, Ledeine JM, Belder R, Gross J, et al. (May 2006). "Improvement of glycemic control, triglycerides, and HDL cholesterol levels with muraglitazar, a dual (alpha/gamma) peroxisome proliferator-activated receptor activator, in patients with type 2 diabetes inadequately controlled with metformin monotherapy: A double-blind, randomized, pioglitazone-comparative study" (PDF). Diabetes Care. 29 (5): 1016–23. doi:10.2337/diacare.2951016. PMID 16644631.
- "Bristol-Myers Squibb Announces Discontinuation of Development of Muraglitazar, an Investigational Oral Treatment for Type 2 Diabetes". PR Newswire from Bristol-Myers Squibb. May 18, 2006. Retrieved 9 November 2016.
- Nissen SE, Wolski K, Topol EJ (November 2005). "Effect of muraglitazar on death and major adverse cardiovascular events in patients with type 2 diabetes mellitus". JAMA. 294 (20): 2581–6. doi:10.1001/jama.294.20.joc50147. PMID 16239637.