Technetium (99mTc) medronic acid
Technetium (99mTc) medronic acid is a pharmaceutical product used in nuclear medicine to localize bone metastases[1] as well as other diseases that can alter the natural turn-over in the bone by bone scintigraphy.[2]
 | |
| Clinical data | |
|---|---|
| Trade names | Mdp-Bracco |
| License data | |
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
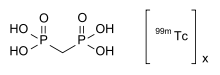
| Formula | CH6O6P2 • x99mTc |
| Molar mass | unknown |
| (verify) | |
Chemistry
The drug is a complex of medronic acid (MDP, methylene diphosphonate), the simplest bisphosphonate, with technetium-99m (99mTc), a radionuclide that emits gamma rays. The exact structure of the complex is not known.[2][3]
Manufacture
99mTc-MDP must be prepared in a radiopharmacy. It is usually supplied as a "cold kit" to which radioactive 99mTc from a generator is added. Kit composition may vary between suppliers, but contents typically includes medronic acid, stannous chloride dihydrate and sometimes ascorbic acid.[4] Pertechnetate, eluted from the generator is added to the kit vial, which is swirled and left to stand. The labelling efficiency, an indication of how much 99mTc remains in pertechnetate form rather than bound to the MDP, can be measured using chromatography.[5][6][7][8]
References
- Uematsu T, Yuen S, Yukisawa S, Aramaki T, Morimoto N, Endo M, et al. (April 2005). "Comparison of FDG PET and SPECT for detection of bone metastases in breast cancer". AJR. American Journal of Roentgenology. 184 (4): 1266–73. doi:10.2214/ajr.184.4.01841266. PMID 15788608.
- FDA Label: MDP Multidose Utilipack
- KEGG Drug: D02029
- "List of labelling kits for the preparation of a technetium-99m labelled bone scanning agent". Human Health Campus. IAEA. Retrieved 3 March 2017.
- "MDP Bracco" (PDF). Bracco Diagnostics. Retrieved 3 March 2017.
- "Kit for the Preparation of Technetium Tc 99m Medronate for Injection" (PDF). Pharmalucence. Retrieved 3 March 2017.
- "TechneScan MDP Kit for the Preparation of Technetium Tc 99m Medronate Package Insert" (PDF). Mallinckrodt Medical. Retrieved 3 March 2017.
- Mather SJ, Ellison D (May 1990). "Reduction-mediated technetium-99m labeling of monoclonal antibodies". Journal of Nuclear Medicine. 31 (5): 692–7. PMID 2341907.
External links
- "Technetium Tc 99m medronate". Drug Information Portal. U.S. National Library of Medicine.