Tromantadine
 | |
| Clinical data | |
|---|---|
| Trade names | Viru-Merz |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Topical (gel) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| NIAID ChemDB | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.053.409 |
| Chemical and physical data | |
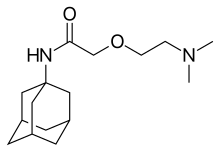
| Formula | C16H28N2O2 |
| Molar mass | 280.412 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
Tromantadine is an antiviral medicine used to treat herpes simplex virus. It is available in a topical gel under trade names Viru-Merz and Viru-Merz Serol. Its performance is similar to aciclovir.[1][2]
Like rimantadine, amantadine, and adapromine, tromantadine is a derivative of adamantane.
Mechanism
Tromantadine inhibits the early and late events in the virus replication cycle.[3] It changes the glycoproteins of the host cells, therefore impeding the absorption of the virus. It inhibits penetration of the virus. It also prevents uncoating of the virions.
References
- ↑ Ostheimer, K. E.; Busch, T.; Görtelmeyer, R.; Hahn, K. D. (September 1989). "Randomized double-blind trial of tromantadine versus aciclovir in recurrent herpes orofacialis". Arzneimittel-Forschung. 39 (9): 1152–1155. ISSN 0004-4172. PMID 2686658.
- ↑ Diezel, W.; Michel, G.; Görtelmeyer, R.; Ostheimer, K. E. (April 1993). "Efficacy of tromantadine and aciclovir in the topical treatment of recurrent herpes orofacialis. Comparison in a clinical trial". Arzneimittel-Forschung. 43 (4): 491–496. ISSN 0004-4172. PMID 8494582.
- ↑ Rosenthal KS, Sokol MS, Ingram RL, Subramanian R, Fort RC (December 1982). "Tromantadine: inhibitor of early and late events in herpes simplex virus replication". Antimicrob. Agents Chemother. 22 (6): 1031–6. doi:10.1128/aac.22.6.1031. PMC 185716. PMID 6297383.
External links
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.