Thiophosgene
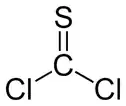
Thiophosgene is a red liquid with the formula CSCl2. It is a molecule with trigonal planar geometry. There are two reactive C–Cl bonds that allow it to be used in diverse organic syntheses.[1]
| |||
 | |||
| Names | |||
|---|---|---|---|
| IUPAC name
Carbonothioyl dichloride | |||
| Other names
Thiophosgene; Thiocarbonyl chloride; Carbonothioic dichloride | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.006.675 | ||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| CSCl2 | |||
| Molar mass | 114.97 g·mol−1 | ||
| Appearance | Red liquid | ||
| Odor | Persistent, choking odor | ||
| Density | 1.50 g/cm3 | ||
| Boiling point | 70 to 75 °C (158 to 167 °F; 343 to 348 K) | ||
| Decomposes | |||
| Solubility in other solvents | Reacts with amines and alcohols, soluble in polar organic solvents | ||
| -50.6·10−6 cm3/mol | |||
Refractive index (nD) |
1.558 | ||
| Structure | |||
| planar, sp2, C2v | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards |
Highly toxic | ||
| Flash point | 62 °C (144 °F; 335 K) | ||
| Related compounds | |||
Related compounds |
| ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |||
Preparation
CSCl2 is prepared in a two-step process from carbon disulfide. In the first step, carbon disulfide is chlorinated to give trichloromethanesulfenyl chloride (perchloromethyl mercaptan), CCl3SCl:
- CS2 + 3 Cl2 → CCl3SCl + S2Cl2
The chlorination must be controlled as excess chlorine converts trichloromethanesulfenyl chloride into carbon tetrachloride. Steam distillation separates the trichloromethanesulfenyl chloride, a rare sulfenyl chloride, and hydrolyzes the disulfur dichloride. Reduction of trichloromethanesulfenyl chloride produces thiophosgene:
- CCl3SCl + M → CSCl2 + MCl2
Tin[2] and dihydroanthracene[3] have been used for the reducing agents.
Reactions
CSCl2 is mainly used to prepare compounds with the connectivity CSX2 where X = OR, NHR.[4] Such reactions proceed via intermediate such as CSClX. Under certain conditions, one can convert primary amines into isothiocyanates. CSCl2 also serves as a dienophile to give, after reduction 5-thiacyclohexene derivatives. Thiophosgene is also known as the appropriate reagent in Corey-Winter synthesis for stereospecific conversion of 1,2-diols into alkenes.[5]
It forms a head-to-tail dimer upon irradiation with UV light:[6]
- 2 CSCl2 → S2(CCl2)2
Unlike thiophosgene monomer, a red liquid, the photodimer, an example of a 1,3-dithietane, is a colourless solid.
Safety considerations
CSCl2 is considered highly toxic.[7]
References
- Manchiu D. S. Lay, Mitchell W. Sauerhoff And Donald R. Saunders "Carbon Disulfide" in Ullmann's Encyclopedia Of Industrial Chemistry, 2000, Wiley-VCH, Weinheim. doi:10.1002/14356007.a05_185
- Dyson, G. M. (1926). "Thiophosgene". Organic Syntheses. 6: 86. doi:10.15227/orgsyn.006.0086.
- K. T. Potts, C. Sapino (1972). "Thiocarbonyl halides". In S. Patai (ed.). Acyl Halides. PATAI'S Chemistry of Functional Groups. pp. 349–380. doi:10.1002/9780470771273.ch11. ISBN 978-0-470-77127-3.
- Pascual, Roxana Martinez "Thiophosgene" Synlett 2015, vol. 26, pp. 1776-1777.doi:10.1055/s-0034-1380659
- Sharma, S. (1978). "Thiophosgene in Organic Synthesis". Synthesis. 1978 (11): 803–820. doi:10.1055/s-1978-24896.
- B. Krebs H. Beyer (1969). "Die Kristall‐ und Molekelstruktur des dimeren Thiophosgens". Z. Anorg. Allg. Chem. 365 (3–4): 199–210. doi:10.1002/zaac.19693650315.
- "Thiophosgene".
Further reading
- Holleman, Arnold Frederik; Wiberg, Egon (2001), Wiberg, Nils (ed.), Inorganic Chemistry, translated by Eagleson, Mary; Brewer, William, San Diego/Berlin: Academic Press/De Gruyter, ISBN 0-12-352651-5