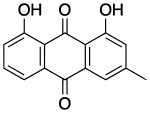
Chrysophanol
Chrysophanol, also known as chrysophanic acid, is a fungal isolate and a natural anthraquinone. It is a C-3 methyl substituted chrysazin of the trihydroxyanthraquinone family.[1]
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,8-Dihydroxy-3-methylanthracene-9,10-dione | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.006.885 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C15H10O4 |
| Molar mass | 254.241 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Chrysophanol (other names; 1,8-dihydroxy-3-methyl-anthraquinone and chrysophanic acid) was found commonly within Chinese medicine and is a naturally occurring anthraquinone.[2] Studies have been conducted on the benefits of chrysophanol and have found that it can aid in preventing cancer, diabetes, asthma, osteoporosis, retinal degeneration, Alzheimer’s disease, osteoarthritis, and atherosclerosis.[2]
Its most common effects are of chemotherapeutic and neuroprotective properties.
History
Chrysophanol was first noted from Rheum rhabarbarum which is a plant belonging to the Polygonaceae family.[3] It has since been discovered to be present in various families such as Liliaceae, Meliaceae, Asphodelaceae and Fabaceae among more.[3] As of 2019, it has been observed in 65 species from 14 genera, not just in plants but animals and microbes as well.[3]
Uses
Chrysophanol has been shown to exhibit a variety of effects. It was shown in 2015 to lower cholesterol and triglyceride levels in zebrafish, as well as increase the frequency of peristalsis. This could therefore be used for lipid metabolic disorders in a clinical setting.[4] Chrysophanol has also been shown to exhibit the same properties lipid lowering in rats in 2013.[4]
It also has the potential to stimulate osteoblast differentiation.[5] as well as alleviate diabetic nephropathy [6] Furthermore, it can protect bronchial cells from cigarette smoke extract induced apoptosis.[7] Chrysophanol can also improve the condition of renal interstitial fibrosis.[8]
Chrysophanol has also been used to inhibit T-Cell activation and protect mice from dextran sulphate sodium induced inflammatory bowel disease.[9] It was shown to have attenuated the pro-inflammatory cytokines that were present in the colon tissue due to sulphate sodium induced inflammatory bowel disease.[9]
Mechanism of action
Chrysophanol can alleviate diabetic nephropathy by inactivating TGF-β/EMT signalling.[6] It also has the potential to protect bronchial cells from cigarette smoke extract by repressing CYP1A expression which is usually produced due to excessive reactive oxygen species.[7] Chrysophanol can increase osteoblast differentiation by inducing AMP-activated protein kinase as well as Smad1/5/9.[5] Chrysophanol acts to improve renal interstitial fibrosis by downregulating TGF-β1 and phospho-Smad3 and by upregulating Smad7.[8]
Chrysophanol can also aid in treatment for inflammatory bowel disease by inhibiting inflammation by targeting pro-inflammatory cytokines that are in tumour necrosis factor α.[9] It has also been shown that it inhibits the mitogen-activated protein kinase pathway.[9]
Chrysophanol blocks the proliferation of colon cancer cells in vitro.[10] It induces the necrosis of cells via a reduction in ATP levels.[11] Chrysophanol attenuates the effects of lead exposure in mice by reducing hippocampal neuronal cytoplasmic edema, enhancing mitochondrial crista fusion, significantly increasing memory and learning abilities, reducing lead content in blood, heart, brain, spleen, kidney and liver, promoting superoxide dismutase and glutathione peroxidase activities and reducing malondialdehyde level in the brain, kidney and liver.[12]
Potential therapeutic uses
Chrysophanol can act as an antineoplastic drug. This has been shown in multiple organisms. It has been reported that chrysophanol causes necrosis-like cell death in renal cancer cells.[13] It also has expressed the capability to be classes as an ATC code A10 drug due to its effect on diabetic nephropathy as well as being able to lower lipid absorption.[4][6]
Production
Chrysophanol is naturally made by a variety of plant species. The most intake is from consumption of rhubarb.[1]
Drug interactions
Chrysophanol has been shown to be able to be co-administered with atorvastatin, to lower cholesterol levels.[4] This is due to the different mechanisms for each, with chrysophanol thought to bind to the stomach to disturb lipid absorption, while atorvastatin decreases cholesterol production in the liver.[4]
Toxicity
Anthraquinones, chrysophanol derivatives among them, have been shown to be hepatotoxic.[14] They can cause apoptosis in normal human liver cells.[14] Chrysophanol derivatives such as chrysophanol-8-o-glucoside, have also been shown to possess anti-coagulant and anti-platelet properties.[15] The derivatives also have potential to cause abnormal oxidative phosphorylation which can result in decreased mitochondrial membrane potential, as well as an increase in abundance of reactive oxygen species, and ultimately will lead to mitochondrial damage and eventual apoptosis.[14]
There is also evidence that chrysophanol could cause damage to DNA.[2] This has been demonstrated in two strains of Salmonella (strains TA 2637 and 1537).[2] It is also important to note, that in treating liver cancer cells, it does so in a way that induced necrosis-like cell death.[16] Necrosis damages the cellular environment, meaning that while it may treat potential issues, it can also damage the surrounding tissue.[16]
References
- PubChem. "Chrysophanol". pubchem.ncbi.nlm.nih.gov. Retrieved 2021-05-31.
- Xie L, Tang H, Song J, Long J, Zhang L, Li X (October 2019). "Chrysophanol: a review of its pharmacology, toxicity and pharmacokinetics". The Journal of Pharmacy and Pharmacology. 71 (10): 1475–1487. doi:10.1111/jphp.13143. PMID 31373015. S2CID 199380101.
- Yusuf MA, Singh BN, Sudheer S, Kharwar RN, Siddiqui S, Abdel-Azeem AM, et al. (February 2019). "Chrysophanol: A Natural Anthraquinone with Multifaceted Biotherapeutic Potential". Biomolecules. 9 (2): 68. doi:10.3390/biom9020068. PMC 6406798. PMID 30781696.
- Chen K, Wang CQ, Fan YQ, Xie YS, Yin ZF, Xu ZJ, et al. (2015-07-15). "Application of chrysophanol in zebrafish to reduce dietary introduced lipid and its possible mechanism". International Journal of Clinical and Experimental Medicine. 8 (7): 10558–67. PMC 4565228. PMID 26379845.
- Lim YJ, Kim KM, Jang WG (April 2021). "Chrysophanol increases osteoblast differentiation via AMPK/Smad1/5/9 phosphorylation in vitro and in vivo". Clinical and Experimental Pharmacology & Physiology. 48 (4): 515–523. doi:10.1111/1440-1681.13443. PMID 33300218. S2CID 228087337.
- Guo C, Wang Y, Piao Y, Rao X, Yin D (2020-11-16). "Chrysophanol Inhibits the Progression of Diabetic Nephropathy via Inactivation of TGF-β Pathway". Drug Design, Development and Therapy. 14: 4951–4962. doi:10.2147/DDDT.S274191. PMC 7678702. PMID 33235436.
- Wu G, Yuan T, Zhu H, Zhang H, Su J, Guo L, et al. (2020-12-15). "Chrysophanol protects human bronchial epithelial cells from cigarette smoke extract (CSE)-induced apoptosis". International Journal of Molecular Epidemiology and Genetics. 11 (3): 39–45. PMC 7811954. PMID 33488953.
- Dou F, Ding Y, Wang C, Duan J, Wang W, Xu H, et al. (October 2020). "Chrysophanol ameliorates renal interstitial fibrosis by inhibiting the TGF-β/Smad signaling pathway". Biochemical Pharmacology. 180: 114079. doi:10.1016/j.bcp.2020.114079. PMID 32511988.
- Lee HS, Jeong GS (March 2021). "Chrysophanol Attenuates Manifestations of Immune Bowel Diseases by Regulation of Colorectal Cells and T Cells Activation In Vivo". Molecules. 26 (6): 1682. doi:10.3390/molecules26061682. PMC 8002617. PMID 33802855.
- Lee MS, Cha EY, Sul JY, Song IS, Kim JY (June 2011). "Chrysophanic acid blocks proliferation of colon cancer cells by inhibiting EGFR/mTOR pathway". Phytotherapy Research. 25 (6): 833–7. doi:10.1002/ptr.3323. PMID 21089180. S2CID 2937534.
- Burnstock G, Di Virgilio F (December 2013). "Purinergic signalling and cancer". Purinergic Signalling. 9 (4): 491–540. doi:10.1007/s11302-013-9372-5. PMC 3889385. PMID 23797685.
- Zhang J, Yan C, Wang S, Hou Y, Xue G, Zhang L (May 2014). "Chrysophanol attenuates lead exposure-induced injury to hippocampal neurons in neonatal mice". Neural Regeneration Research. 9 (9): 924–30. doi:10.4103/1673-5374.133141. PMC 4146226. PMID 25206913.
- Choi JS (June 2016). "Chrysophanic Acid Induces Necrosis but not Necroptosis in Human Renal Cell Carcinoma Caki-2 Cells". Journal of Cancer Prevention. 21 (2): 81–7. doi:10.15430/JCP.2016.21.2.81. PMC 4933431. PMID 27390736.
- Lin L, Yuan F, Liu Y, Zhong M, Xie T, Ni J, Li H (December 2019). "Hepatotoxicity and mechanism study of chrysophanol-8-O-glucoside in vitro". Biomedicine & Pharmacotherapy. 120: 109531. doi:10.1016/j.biopha.2019.109531. PMID 31648163.
- Seo EJ, Ngoc TM, Lee SM, Kim YS, Jung YS (2012). "Chrysophanol-8-O-glucoside, an anthraquinone derivative in rhubarb, has antiplatelet and anticoagulant activities". Journal of Pharmacological Sciences. 118 (2): 245–54. doi:10.1254/jphs.11123fp. PMID 22302018.
- Ni CH, Chen PY, Lu HF, Yang JS, Huang HY, Wu SH, et al. (May 2012). "Chrysophanol-induced necrotic-like cell death through an impaired mitochondrial ATP synthesis in Hep3B human liver cancer cells". Archives of Pharmacal Research. 35 (5): 887–95. doi:10.1007/s12272-012-0514-z. PMID 22644856. S2CID 5631532.
Further reading
Trybus W, Król T, Trybus E, Stachurska A, Król G (June 2021). "The potential antitumor effect of chrysophanol in relation to cervical cancer cells". Journal of Cellular Biochemistry. 122 (6): 639–652. doi:10.1002/jcb.29891. PMID 33417255. S2CID 231193505.