Low-molecular-weight heparin
Low-molecular-weight heparin (LMWH) is a class of anticoagulant medications.[1] They are used in the prevention of blood clots and treatment of venous thromboembolism (deep vein thrombosis and pulmonary embolism) and in the treatment of myocardial infarction.
 Nadroparin in prefilled syringe | |
| Pharmacokinetic data | |
|---|---|
| Bioavailability | 100% |
| Chemical and physical data | |
| Molar mass | 4-6 kDa |
Heparin is a naturally occurring polysaccharide that inhibits coagulation, the process that leads to thrombosis. Natural heparin consists of molecular chains of varying lengths, or molecular weights. Chains of varying molecular weights, from 5000 to over 40,000 Daltons, make up polydisperse pharmaceutical-grade heparin.[2] LMWHs, in contrast, consist of only short chains of polysaccharide. LMWHs are defined as heparin salts having an average molecular weight of less than 8000 Da and for which at least 60% of all chains have a molecular weight less than 8000 Da. These are obtained by various methods of fractionation or depolymerisation of polymeric heparin.
Heparin derived from natural sources, mainly porcine intestine or bovine lung, can be administered therapeutically to prevent thrombosis. However, the effects of natural, or unfractionated heparin are more unpredictable than LMWH.[3]
Medical uses
Because it can be given subcutaneously and does not require APTT monitoring, LMWH permits outpatient treatment of conditions such as deep vein thrombosis or pulmonary embolism that previously mandated inpatient hospitalization for unfractionated heparin administration.
Because LMWH has more predictable pharmacokinetics and anticoagulant effect, LMWH is recommended over unfractionated heparin for patients with massive pulmonary embolism,[4] and for initial treatment of deep vein thrombosis.[5] As compared to placebo or no intervention, prophylactic treatment of hospitalized medical patients using LMWH and similar anticoagulants reduces the risk of venous thromboembolism, notably pulmonary embolism.[6][7]
More recently these agents have been evaluated as anticoagulants in acute coronary syndrome (ACS) managed by percutaneous intervention (PCI).[8][9]
The use of LMWH needs to be monitored closely in patients at extremes of weight or in-patients with renal dysfunction. An anti-factor Xa activity may be useful for monitoring anticoagulation. Given its renal clearance, LMWH may not be feasible in patients that have end-stage renal disease. LMWH can also be used to maintain the patency of cannulae and shunts in dialysis patients.
Patients with cancer are at higher risk of venous thromboembolism and LMWHs are used to reduce this risk.[10] The CLOT study, published in 2003, showed that, in patients with malignancy and acute venous thromboembolism, dalteparin was more effective than warfarin in reducing the risk of recurrent embolic events.[11] Use of LMWH in cancer patients for at least the first 3 to 6 months of long-term treatment is recommended in numerous guidelines and is now regarded as a standard of care.[10]
Contraindications
The use of LMWHs should be avoided in patients with known allergies to LMWHs, heparin, sulfites or benzyl alcohol, in patients with active major bleeding, or patients with a history of heparin-induced low blood platelet count (also known as heparin-induced thrombocytopenia or HIT). High treatment doses are contraindicated in acute bleedings such as cerebral or gastrointestinal haemorrhage. LMWHs are more dependent on renal function for their excretion than unfractionated heparin so their biological half-life may be prolonged in patients with kidney failure and therefore their use in the setting of creatinine clearance rate (CrCl) <30 mL/min may need to be avoided.[12] Apart from using unfractionated heparin instead, it may be possible to reduce the dose and/or monitor the anti-Xa activity to guide treatment.[3]
The most common side-effects include bleeding, which could be severe or even fatal, allergic reactions, injection site reactions, and increases in liver enzyme tests, usually without symptoms.[13] The use of heparin and LMWHs can sometimes be complicated by a decrease in platelet count, a complication known as Heparin Induced Thrombocytopenia.13 Two forms have been described: a clinically benign, non-immune and reversible form (Type I) and a rare, more serious immune-mediated form or Type II. HIT Type II is caused by the formation of auto antibodies that recognize complexes between heparin and platelet factor 4 (PF4) and is therefore associated with a substantial risk of thrombotic complications. The incidence is difficult to estimate but may reach up to 5% of patients treated with UFH or about 1% with LMWH.[13]
Antidote
In clinical situations in which the antithrombotic effect of LMWHs needs to be neutralized, protamine is used to neutralize heparin by binding to it.[9] Studies in animals and in vitro studies have demonstrated that protamine neutralizes the antithrombin activity of LMWHs, normalizing the aPTT and thrombin time. However, protamine appears to only partially neutralize the anti-factor Xa activity of LMWH. Because the molecular weight of heparin impacts its interaction with protamine, it is likely that the lack of complete neutralization of anti-factor Xa is due to a reduced protamine binding to the LMWHs moieties in the preparation. Protamine is a medicine that requires a high level of caution when used.
Precautions
LMWH trials usually excluded individuals with unpredictable pharmacokinetics, and as a result patients with risks such as the severely obese or in advanced stages of kidney failure show decreased benefits due to fractionated heparin's increased half-life.[14] LMWHs should be used with extreme caution in patients undergoing any procedure involving spinal anaesthesia/puncture, in conditions with increased risk of bleeding or in patients with a history of heparin-induced thrombocytopenia.
Pharmacology
Mechanism of action
Coagulation cascade is a normal physiological process which aims at preventing significant blood loss or hemorrhage following vascular injury. Unfortunately, there are times when a blood clot (thrombus) will form when it is not needed. For instance, some high risk conditions such as prolonged immobilization, surgery, or cancer can increase the risk of developing a blood clot which can potentially lead to significant consequences.
The coagulation cascade consists of a series of steps in which a protease cleaves and subsequently activates the next protease in the sequence.[2] Since each protease can activate several molecules of the next protease in the series, this biological cascade is amplified. The result of these reactions is to convert fibrinogen, a soluble protein, to insoluble threads of fibrin. Together with platelets, the fibrin threads form a stable blood clot.
Antithrombin (AT), a serine protease inhibitor, is the major plasma inhibitor of coagulation proteases.[15] LMWHs inhibit the coagulation process through binding to AT via a pentasaccharide sequence. This binding leads to a conformational change of AT which increases the rate at which it inhibits activated factor X (factor Xa). Once dissociated, the LMWH is free to bind to another antithrombin molecule and subsequently inhibit more activated factor X. Unlike AT activated by heparin, AT activated by LMWH cannot inhibit thrombin (factor IIa), but can only inhibit clotting factor Xa.
The effects of LMWHs cannot be acceptably measured using the partial thromboplastin time (PTT) or activated clotting time (ACT) tests.[16] Rather, LMWH therapy is monitored by the anti-factor Xa assay, measuring anti-factor Xa activity rather than a clotting time. The methodology of an anti-factor Xa assay is that patient plasma is added to a known amount of excess recombinant factor X and excess antithrombin. If heparin or LMWH is present in the patient plasma, it will bind to antithrombin and form a complex with factor X, inhibiting it from becoming factor Xa.[17] The amount of residual factor Xa is inversely proportional to the amount of heparin/LMWH in the plasma. The amount of residual factor Xa is detected by adding a chromogenic substrate that mimics the natural substrate of factor Xa, making residual factor Xa cleave it, releasing a colored compound that can be detected by a spectrophotometer.[17] Antithrombin deficiencies in the patient do not affect the assay, because excess amounts of antithrombin is provided in the reaction.[17] Results are given in units/mL of antifactor Xa, such that high values indicate high levels of anticoagulation and low values indicate low levels of anticoagulation in the plasma sample.[17]
LMWHs have a potency of greater than 70 units/mg of anti-factor Xa activity and a ratio of anti-factor Xa activity to anti-thrombin activity of >1.5.[18] (see table 1)
| LMWH | Average molecular weight | Ratio anti-Xa/anti-IIa activity |
|---|---|---|
| Bemiparin | 3600 | 9.7 |
| Nadroparin | 4300 | 3.3 |
| Reviparin | 4400 | 4.2 |
| Enoxaparin | 4500 | 3.9 |
| Parnaparin | 5000 | 2.3 |
| Certoparin | 5400 | 2.4 |
| Dalteparin | 6000 | 2.5 |
| Tinzaparin | 6500 | 1.6 |
Table 1 Molecular weight (MW) data and anticoagulant activities of currently available LMWH products. Adapted from Gray E et al. 2008.[19]
Manufacturing process
Various methods of heparin depolymerisation are used in the manufacture of low-molecular-weight heparin.[2] These are listed below:
- Oxidative depolymerisation with hydrogen peroxide. Used in the manufacture of ardeparin (Normiflo)
- Deaminative cleavage with isoamyl nitrite. Used in the manufacture of certoparin (Sandoparin)
- Alkaline beta-eliminative cleavage of the benzyl ester of heparin. Used in the manufacture of enoxaparin (Lovenox and Clexane)
- Oxidative depolymerisation with Cu2+ and hydrogen peroxide. Used in the manufacture of parnaparin (Fluxum)
- Beta-eliminative cleavage by the heparinase enzyme. Used in the manufacture of tinzaparin (Innohep and Logiparin)
- Deaminative cleavage with nitrous acid. Used in the manufacture of dalteparin (Fragmin), reviparin (Clivarin), and nadroparin (Fraxiparin)
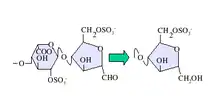
Deaminative cleavage with nitrous acid results in the formation of an unnatural anhydromannose residue at the reducing terminal of the oligosaccharides produced. This can subsequently be converted to anhydromannitol using a suitable reducing agent as shown in figure 1.
-GlcNS(6S).svg.png.webp)
Likewise both chemical and enzymatic beta-elimination result in the formation of an unnatural unsaturated uronate residue(UA) at the non-reducing terminal, as shown in figure 2.
In addition, low molecular weight heparins can also be chemoenzymatically synthesized from simple disaccharides.[20]
Differences between LMWHs
Comparisons between LMWHs prepared by similar processes vary. For example, a comparison of Dalteparin and Nadroparin suggests they are more similar than products produced by different processes. However, comparison of enoxaparin and tinzaparin shows they are very different from each other with respect to chemical, physical, and biological properties.
As might be expected, products prepared by distinctly different processes are dissimilar in physical, chemical, and biological properties.[2][15] Hence a slight change in the depolymerisation process could result in substantial variation of the structure or composition of a given LMWH.
Therefore, for every LMWH, a strictly defined depolymerisation procedure is needed to guarantee the sameness of the final LMWH product and the predictability of clinical outcomes. LMWHs, as biological origin products, rely on stringent manufacturing procedures to guarantee the absence of biological or chemical contamination. It is therefore critical to adopt stringent manufacturing practices, through rigorous quality assurance steps, to ensure the highest quality of the produced LMWHs and to guarantee patient safety. These quality assurance steps, to be effective, need to be implemented from the raw material (crude heparin) collection to the final LMWH product.
Due to these identified and potential differences, several organizations, including the United States Food and Drug Administration, the European Medicines Agency, and the World Health Organization, regard LMWHs as individual products that should not be considered as clinically equivalent, as they differ in many crucial aspects such as molecular, structural, physiochemical, and biological properties.[21][22][23] According to international guidelines, the choice of an individual LMWH should be based on its proven clinical safety and efficacy for each indication.[13] Therefore, switching from LMWH to another LMWH during treatment is not recommended during clinical practice.[24]
Differences from unfractionated heparin
Differences from heparin (i.e. "unfractionated heparin") include:
- Average molecular weight: heparin is about 15 kDa and LMWH is about 4.5 kDa.[25]
- Less frequent subcutaneous dosing than for heparin for postoperative prophylaxis of venous thromboembolism.
- Once or twice daily subcutaneous injection for treatment of venous thromboembolism and in unstable angina instead of intravenous infusion of high dose heparin.
- No need for monitoring of the APTT coagulation parameter as required for high dose heparin.[26]
- Possibly a smaller risk of bleeding.
- Smaller risk of osteoporosis in long-term use.
- Smaller risk of heparin-induced thrombocytopenia, a potential side effect of heparin.
- The anticoagulant effects of heparin are typically reversible with protamine sulfate, while protamine's effect on LMWH is limited.
- LMWH has less of an effect on thrombin compared to heparin, but about the same effect on Factor Xa.
- Due to its renal clearance, LMWH is contraindicated in patients with kidney disease in whom unfractionated heparin can be used safely.
Generics and biosimilars
When the commercial patent of a LMWH expires, a generic or biosimilar LMWH can then be marketed. The first 'generic' LMWH was approved by the Food and Drug Administration in July 2010. The FDA has used 5 analytical and pharmacological criteria to establish the authenticity of a generic LMWH, without requiring clinical studies in patients.[27]
From a regulatory viewpoint, the FDA considers LMWHs (as well as insulin, glucagon and somatropin) as "generic" drugs, even though they may be sourced from biological material. The European Medicines Agency considers LMWHs as biologicals so their regulatory approval - as biosimilars - is approached differently compared to the FDA.[28][29]
References
- Weitz JI (September 1997). "Low-molecular-weight heparins". The New England Journal of Medicine. 337 (10): 688–698. doi:10.1056/NEJM199709043371007. PMID 9278467.
- Linhardt RJ, Gunay NS (1999). "Production and chemical processing of low molecular weight heparins". Seminars in Thrombosis and Hemostasis. 25 (Suppl 3): 5–16. PMID 10549711.
- Garcia DA, Baglin TP, Weitz JI, Samama MM (February 2012). "Parenteral anticoagulants: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines". Chest. 141 (2 Suppl): e24S–e43S. doi:10.1378/chest.11-2291. PMC 3278070. PMID 22315264.
- Hull RD (March 2008). "Treatment of pulmonary embolism: The use of low-molecular-weight heparin in the inpatient and outpatient settings". Thrombosis and Haemostasis. 99 (3): 502–510. doi:10.1160/TH07-08-0500. PMID 18327398. S2CID 25247285.
- Snow V, Qaseem A, Barry P, Hornbake ER, Rodnick JE, Tobolic T, et al. (February 2007). "Management of venous thromboembolism: a clinical practice guideline from the American College of Physicians and the American Academy of Family Physicians". Annals of Internal Medicine. 146 (3): 204–210. doi:10.7326/0003-4819-146-3-200702060-00149. PMID 17261857.
- Douketis JD, Moinuddin I (April 2008). "Prophylaxis against venous thromboembolism in hospitalized medical patients: an evidence-based and practical approach". Polskie Archiwum Medycyny Wewnetrznej. 118 (4): 209–215. PMID 18575420.
- Dentali F, Douketis JD, Gianni M, Lim W, Crowther MA (February 2007). "Meta-analysis: anticoagulant prophylaxis to prevent symptomatic venous thromboembolism in hospitalized medical patients". Annals of Internal Medicine. 146 (4): 278–288. CiteSeerX 10.1.1.694.4563. doi:10.7326/0003-4819-146-4-200702200-00007. PMID 17310052. S2CID 27605124.
- Montalescot G, Ellis SG, de Belder MA, Janssens L, Katz O, Pluta W, et al. (February 2010). "Enoxaparin in primary and facilitated percutaneous coronary intervention A formal prospective nonrandomized substudy of the FINESSE trial (Facilitated INtervention with Enhanced Reperfusion Speed to Stop Events)". JACC. Cardiovascular Interventions. 3 (2): 203–212. doi:10.1016/j.jcin.2009.11.012. PMID 20170878.
- Dumaine R, Borentain M, Bertel O, Bode C, Gallo R, White HD, et al. (December 2007). "Intravenous low-molecular-weight heparins compared with unfractionated heparin in percutaneous coronary intervention: quantitative review of randomized trials". Archives of Internal Medicine. 167 (22): 2423–2430. doi:10.1001/archinte.167.22.2423. PMID 18071163.
- Nishioka J, Goodin S (June 2007). "Low-molecular-weight heparin in cancer-associated thrombosis: treatment, secondary prevention, and survival". Journal of Oncology Pharmacy Practice. 13 (2): 85–97. doi:10.1177/1078155207079169. PMID 17873108. S2CID 39517086.
- Lee AY, Levine MN, Baker RI, Bowden C, Kakkar AK, Prins M, et al. (July 2003). "Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer". The New England Journal of Medicine. 349 (2): 146–153. doi:10.1056/NEJMoa025313. PMID 12853587. S2CID 19280602.
- Lim W, Dentali F, Eikelboom JW, Crowther MA (May 2006). "Meta-analysis: low-molecular-weight heparin and bleeding in patients with severe renal insufficiency". Annals of Internal Medicine. 144 (9): 673–684. doi:10.7326/0003-4819-144-9-200605020-00011. PMID 16670137. S2CID 29713358.
- Cardiovascular Disease Educational Research Trust; Cyprus Cardiovascular Disease Educational Research Trust; International Surgical Thrombosis Forum; International Union of Angiology; Union Internationale de Phlébologie (June 2006). "Prevention and treatment of venous thromboembolism. International Consensus Statement (guidelines according to scientific evidence)". International Angiology. 25 (2): 101–161. PMID 16763532.
- Hetzel GR, Sucker C (October 2005). "The heparins: all a nephrologist should know". Nephrology, Dialysis, Transplantation. 20 (10): 2036–2042. doi:10.1093/ndt/gfi004. PMID 16030035.
- Jeske W, Walenga J, Fareed J. Differentiating between the Low-Molecular-Weight-Heparin used for VTE treatment and prophylaxis. Thromb Clin. 2008;2(3)
- enotes.com > Encyclopedia of Nursing & Allied Health > Coagulation Tests Retrieved on April 5, 2010
- massgeneral.org > Heparin Antifactor Xa Assay Archived 2009-08-08 at archive.today Page Updated: September 18, 2009
- European Pharmacopedia Commission (October 1991). "Low Molecular Mass FEPAFUNS". Pharmeuropa. 3: 161–165.
- Gray E, Mulloy B, Barrowcliffe TW. Heparin and low-molecular-weight-heparin. Thromb Haemost 2008; 99: 807–818.
- Xu Y, Masuko S, Takieddin M, Xu H, Liu R, Jing J, et al. (October 2011). "Chemoenzymatic synthesis of homogeneous ultralow molecular weight heparins". Science. 334 (6055): 498–501. Bibcode:2011Sci...334..498X. doi:10.1126/science.1207478. PMC 3425363. PMID 22034431.
- WHO Working Group on Biological Standardization of Unfractionated Heparin, WHO Headquarters, Geneva, Switzerland, 7–8 September 1999
- EMA/CHMP/BMWP/118264/2007. Guideline on non-clinical andclinical development of similar biological medicinal products containing Low-molecular-weight-heparins. March 2009.
- Nightingale SL (October 1993). "From the Food and Drug Administration". JAMA. 270 (14): 1672. CiteSeerX 10.1.1.170.9453. doi:10.1001/jama.270.14.1672. PMID 8411485.
- Merli GJ, Groce JB (February 2010). "Pharmacological and clinical differences between low-molecular-weight heparins: implications for prescribing practice and therapeutic interchange". P & T. 35 (2): 95–105. PMC 2827912. PMID 20221326.
- Michael D Randall; Karen E Neil (2004). Disease management. 2nd ed. London: Pharmaceutical Press. 186.
- Hirsh J, Warkentin TE, Shaughnessy SG, Anand SS, Halperin JL, Raschke R, et al. (January 2001). "Heparin and low-molecular-weight heparin: mechanisms of action, pharmacokinetics, dosing, monitoring, efficacy, and safety". Chest. 119 (1 Suppl): 64S–94S. doi:10.1378/chest.119.1_suppl.64s. PMID 11157643.
- Harenberg J. Overview on guidelines and recommendations for generic low-molecular-weight heparins. Thrombosis Research 127 Suppl. 3 (2011) S100–S104
- Jeske W, Walenga JM, Hoppensteadt D, Fareed J (2013). "Update on the safety and bioequivalence of biosimilars - focus on enoxaparin". Drug, Healthcare and Patient Safety. 5: 133–141. doi:10.2147/DHPS.S28813. PMC 3684140. PMID 23788840.
- Blank T, Netzer T, Hildebrandt W, Vogt-Eisele A, Kaszkin-Bettag M (2013). "Safety and toxicity of biosimilars—EU versus US regulation". Generics and Biosimilars Initiative Journal. 2 (3): 144–150. doi:10.5639/gabij.2013.0203.039.
External links
- Low+Molecular+Weight+Heparin at the US National Library of Medicine Medical Subject Headings (MeSH)