Luzindole
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
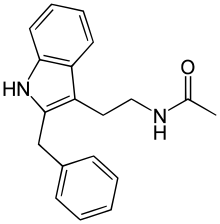
| Formula | C19H20N2O |
| Molar mass | 292.382 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Luzindole (N-0774), (N-acetyl-2-benzyltryptamine), is a drug used in scientific research to study the role of melatonin in the body. Luzindole acts as a selective melatonin receptor antagonist,[1] with approximately 11- to 25-fold greater affinity for the MT2 over the MT1 receptor.[2][3] In animal studies, it has been observed to disrupt the circadian rhythm as well as produce antidepressant effects.[2][4]
References
- ↑ Dubocovich ML (September 1988). "Luzindole (N-0774): a novel melatonin receptor antagonist". The Journal of Pharmacology and Experimental Therapeutics. 246 (3): 902–10. PMID 2843633.
- 1 2 Dubocovich ML, Yun K, Al-Ghoul WM, Benloucif S, Masana MI (September 1998). "Selective MT2 melatonin receptor antagonists block melatonin-mediated phase advances of circadian rhythms". The FASEB Journal. 12 (12): 1211–20. doi:10.1096/fasebj.12.12.1211. PMID 9737724. S2CID 566199.
- ↑ Browning C, Beresford I, Fraser N, Giles H (March 2000). "Pharmacological characterization of human recombinant melatonin mt(1) and MT(2) receptors". British Journal of Pharmacology. 129 (5): 877–86. doi:10.1038/sj.bjp.0703130. PMC 1571913. PMID 10696085.
- ↑ Dubocovich ML, Mogilnicka E, Areso PM (July 1990). "Antidepressant-like activity of the melatonin receptor antagonist, luzindole (N-0774), in the mouse behavioral despair test". European Journal of Pharmacology. 182 (2): 313–25. doi:10.1016/0014-2999(90)90290-M. PMID 2168835.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.