Crisaborole
 | |
| Names | |
|---|---|
| Pronunciation | /juːˈkrɪsə/ yoo-KRIS-ə |
| Trade names | Eucrisa, Staquis |
| Other names | AN-2728 |
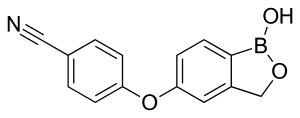
IUPAC name
| |
| Clinical data | |
| Drug class | PDE-4 inhibitor[1] |
| Main uses | Atopic dermatitis (eczema)[1][2] |
| Side effects | Burning or stinging at application site[2] |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Pregnancy category |
|
| Routes of use | Topical (ointment) |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a617019 |
| Legal | |
| License data |
|
| Legal status | |
| Chemical and physical data | |
| Formula | C14H10BNO3 |
| Molar mass | 251.05 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Crisaborole, sold under the brand name Eucrisa among others, is a medication used to treat atopic dermatitis (eczema).[1][2] It is used for mild to moderate disease in those over the age of two months.[1] It is applied to the skin.[1]
Common side effects include burning or stinging at the site of application.[2] Other side effects may include allergic reactions.[1] It is a phosphodiesterase 4 (PDE-4) inhibitor and beleived to work by blocking the release of cytokines.[1][2]
Crisaborole was approved for medical use in the United States in 2016 and Europe in 2020.[1][2] In the United States a tube of 60 grams costs about 680 USD as of 2022.[3] While it is approved in the United Kingdom, it is not commercially available as of 2022.[4]
Medical uses
In the US, crisaborole is indicated for topical treatment of mild to moderate atopic dermatitis in people three months of age and older.[1]
In the EU, crisaborole is indicated for treatment of mild to moderate atopic dermatitis in people two years of age and older with ≤ 40% body surface area (BSA) affected.[2]
Dosage
It is used twice a day at a dose of 2%.[1]
Side effects
At the site of application, crisaborole may cause burning or stinging. Rarely, there may be an allergic reaction.[5]
Pharmacology
Pharmacodynamics
Crisaborole is a phosphodiesterase-4 inhibitor, mainly acting on phosphodiesterase 4B (PDE4B), which causes inflammation.[6] Chemically, crisaborole is a phenoxybenzoxaborole.[6] Inhibition of PDE4B appears to suppress the release of tumor necrosis factor alpha (TNFα), interleukin-12 (IL-12), IL-23 and other cytokines, proteins believed to be involved in the immune response and inflammation.[6]
People with atopic dermatitis produce high levels of proteins called cytokines, which can cause the inflammation of the skin seen in dermatitis.[2] Crisaborole blocks the release of certain cytokines involved in the inflammation process such as tumor necrosis factor alpha, interleukins (IL‑2, IL-4, IL-5), and interferon gamma.[2] By blocking their release, crisaborole is expected to ease the inflammation and therefore relieve symptoms of the disease.[2]
Chemistry
Crisaborole (chemical name: 4-[(1-hydroxy-1,3-dihydro-2,1-benzoxaborol-5-yl)oxy]benzonitrile) is a member of the class of benzoxaboroles characterized by the presence of a boronic acid hemiester with a phenolic ether and a nitrile.[7] Crisaborole crystallizes into two polymorphs that differ in the conformation of the oxaborole ring. A cocrystal with 4,4'-bipyridine has been prepared and studied by X-ray crystallography.[8]
History
Crisaborole was developed by Anacor Pharmaceuticals for the topical treatment of psoriasis.[9][6][10] During preclinical and clinical development, crisaborole was called AN2728 and PF-06930164.[11] The drug was assumed to be potential $2bn-a-year blockbuster, when Pfizer acquired Anacor Pharmaceuticals.[12] However, the drug was commercially not successful, reaching only US$147 million in sales in 2018, and US$138 million in sales in 2019.[13]
Crisaborole was approved for use in the United States in December 2016[14][15] and for use in Canada in June 2018.[16]
The safety and efficacy of crisaborole were established in two placebo-controlled trials with a total of 1,522 participants ranging in age from two years of age to 79 years of age, with mild to moderate atopic dermatitis.[15] In both trials participants received treatment with either crisaborole or placebo twice daily for 28 days.[17] Neither the participants nor the health care providers knew which treatment was being given until after the trials were completed.[17] Overall, participants receiving crisaborole achieved greater response with clear or almost clear skin after 28 days of treatment.[15][17] The trials were conducted in the US.[17]
Crisaborole was approved for use in the European Union in March 2020, for the treatment of mild to moderate atopic dermatitis.[2]
See also
- Tavaborole – a structurally related topical antifungal
References
- 1 2 3 4 5 6 7 8 9 10 "Eucrisa- crisaborole ointment". DailyMed. 21 April 2020. Archived from the original on 23 March 2021. Retrieved 28 April 2020.
- 1 2 3 4 5 6 7 8 9 10 11 "Staquis EPAR". European Medicines Agency (EMA). 29 January 2020. Archived from the original on 2 November 2021. Retrieved 28 April 2020. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ↑ "Crisaborole Prices, Coupons & Savings Tips - GoodRx". GoodRx. Retrieved 7 January 2022.
- ↑ "Crisaborole". SPS - Specialist Pharmacy Service. 8 January 2016. Archived from the original on 11 October 2021. Retrieved 7 January 2022.
- ↑ "PRODUCT MONOGRAPH" (PDF). Government of Canada. 7 June 2018. Archived (PDF) from the original on 7 April 2019. Retrieved 7 April 2019.
- 1 2 3 4 Moustafa F, Feldman SR (May 2014). "A review of phosphodiesterase-inhibition and the potential role for phosphodiesterase 4-inhibitors in clinical dermatology" (PDF). Dermatology Online Journal. 20 (5): 22608. PMID 24852768. Archived (PDF) from the original on 5 October 2017. Retrieved 4 October 2020.
- ↑ "WHO Drug Information, Vol. 29, No. 3, 2015. International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended International Nonproprietary Names: List 74" (PDF). World Health Information. p. 391. Archived (PDF) from the original on 18 May 2016. Retrieved 26 April 2016.
- ↑ Campillo-Alvarado G, Didden TD, Oburn SM, Swenson DC, MacGillivray LR (2018). "Exploration of Solid Forms of Crisaborole: Crystal Engineering Identifies Polymorphism in Commercial Sources and Facilitates Cocrystal Formation". Crystal Growth & Design. 18 (8): 4416–4419. doi:10.1021/acs.cgd.8b00375. ISSN 1528-7483.
- ↑ Nazarian R, Weinberg JM (November 2009). "AN-2728, a PDE4 inhibitor for the potential topical treatment of psoriasis and atopic dermatitis". Current Opinion in Investigational Drugs. 10 (11): 1236–42. PMID 19876791.
- ↑ Spreitzer H (16 August 2016). "Neue Wirkstoffe: Crisaborol". Österreichische Apotheker-Zeitung (in German) (17/2016).
{{cite journal}}: CS1 maint: unrecognized language (link) - ↑ "Crisaborole". AdisInsight. Archived from the original on 20 July 2018. Retrieved 24 July 2017.
- ↑ "Pfizer to Acquire Anacor". Pfizer (Press release). 16 May 2016. Archived from the original on 28 April 2020. Retrieved 28 April 2020.
- ↑ "Pfizer Financial Report" (PDF). Pfizer. Archived (PDF) from the original on 2 February 2020. Retrieved 9 April 2020.
- ↑ "Eucrisa (crisaborole) Ointment". U.S. Food and Drug Administration (FDA). 23 January 2017. Archived from the original on 8 August 2020. Retrieved 28 April 2020.
- 1 2 3 "FDA Approves Eucrisa for Eczema" (Press release). U.S. Food and Drug Administration (FDA). 14 December 2016. Archived from the original on 23 April 2019. Retrieved 4 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ↑ "Eucrisa Regulatory Decision Summary". Health Canada. 23 October 2014. Archived from the original on 14 September 2019. Retrieved 14 September 2019.
- 1 2 3 4 "Drug Trials Snapshot: Eucrisa". U.S. Food and Drug Administration (FDA). 14 December 2016. Archived from the original on 28 April 2020. Retrieved 28 April 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
External links
| External sites: |
|
|---|---|
| Identifiers: |