Fondaparinux
 | |
| Names | |
|---|---|
| Trade names | Arixtra |
| Other names | Fondaparinux sodium |
| Clinical data | |
| Drug class | Factor Xa inhibitor[1] |
| Main uses | Treat and prevent blood clots, unstable angina, heart attacks[1][2] |
| Side effects | Bleeding[1] |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Routes of use | Subcutaneous |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| License data |
|
| Legal status | |
| Pharmacokinetics | |
| Bioavailability | N/A |
| Protein binding | 94% |
| Metabolism | renally excreted unchanged |
| Elimination half-life | 17-21 hours[3] |
| Chemical and physical data | |
| Formula | C31H43N3Na10O49S8 |
| Molar mass | 1728.03 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Fondaparinux, sold under the brand name Arixtra, is an anticoagulant used to treat and prevent blood clots (deep vein, pulmonary, and superficial vein thrombosis) and to treat unstable angina and heart attacks.[1][2] It is given by injection under the skin.[2]
Common side effects include bleeding.[1] Other side effects may include low platelets.[1] Use is not recommended in those with serious kidney problems.[2] There is no evidence of harm to the baby with use in pregnancy.[1] It works by blocking factor Xa.[2]
Fondaparinux was approved for medical use in the United States in 2001 and Europe in 2002.[1][2] It is available as a generic medication.[4] In the United Kingdom 2.5 mg costs the NHS about £6.[4] In the United States this amount costs about 20 USD.[5]
Medical uses
It is used to prevent deep vein thrombosis in people following orthopedic surgery as well as for the treatment of deep vein thrombosis and pulmonary embolism.
Fondaparinux is similar to enoxaparin in reducing the risk of ischemic events at nine days, but it substantially reduces major bleeding and improves long-term mortality and morbidity.[6]
Dosage
It may be used at a dose of 2.5 to 10 mg once per day, depending on the reason.[4]
Comparison to other agents
One potential advantage of fondaparinux over LMWH or unfractionated heparin is that the risk for heparin-induced thrombocytopenia (HIT) is substantially lower. Furthermore, there have been case reports of fondaparinux being used to anti-coagulate patients with established HIT as it has no affinity for PF4. However, its renal excretion precludes its use in patients with renal dysfunction.
Unlike direct factor Xa inhibitors, it mediates its effects indirectly through antithrombin III, but unlike heparin, it is selective for factor Xa.[7]
Pharmacology
Mechanism of action
Fondaparinux is a synthetic pentasaccharide factor Xa inhibitor. Fondaparinux binds antithrombin and accelerates its inhibition of factor Xa.
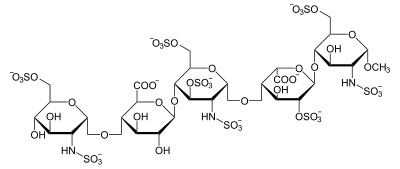
Apart from the O-methyl group at the reducing end of the molecule, the identity and sequence of the five monomeric sugar units contained in fondaparinux is identical to a sequence of five monomeric sugar units that can be isolated after either chemical or enzymatic cleavage of the polymeric glycosaminoglycans heparin and heparan sulfate (HS). Within heparin and heparan sulfate this monomeric sequence is thought to form the high-affinity binding site for the anti-coagulant factor antithrombin (AT). Binding of heparin or HS to AT has been shown to increase the anti-coagulant activity of antithrombin 1000 fold. In contrast to heparin, fondaparinux does not inhibit thrombin.
Chemistry
Abbreviations
- GlcNS6S = 2-deoxy-6-O-sulfo-2-(sulfoamino)-α-D-glucopyranoside
- GlcA = β-D-glucopyranuronoside
- GlcNS3,6S = 2-deoxy-3,6-di-O-sulfo-2-(sulfoamino)-α-D-glucopyranosyl
- IdoA2S = 2-O-sulfo-α-L-idopyranuronoside
- GlcNS6SOMe = methyl-O-2-deoxy-6-O-sulfo-2-(sulfoamino)-α-D-glucopyranoside
Fondaparinux is only accessible by chemical synthesis. Recently, Supriya Dey et al. reported an effective and scalable one-pot synthesis of Fondaparinux.[8]
The sequence of monosaccharides is D-GlcNS6S-α-(1,4)-D-GlcA-β-(1,4)-D-GlcNS3,6S-α-(1,4)-L-IdoA2S-α-(1,4)-D-GlcNS6S-OMe, as shown in the following structure:

Society and culture
It is marketed by GlaxoSmithKline. A generic version developed by Alchemia is marketed within the US by Dr. Reddy's Laboratories.
References
- 1 2 3 4 5 6 7 8 "Fondaparinux Monograph for Professionals". Drugs.com. Archived from the original on 23 January 2021. Retrieved 13 December 2021.
- 1 2 3 4 5 6 "Arixtra". Archived from the original on 21 June 2021. Retrieved 13 December 2021.
- ↑ Walenga JM, Jeske WP, Fareed J (2005). "Biochemical and Pharmacologic Rationale for Synthetic Heparin Polysaccharides". Chemistry and Biology of Heparin and Heparan Sulfate. Elsevier. pp. 143–177. doi:10.1016/b978-008044859-6/50006-x. ISBN 978-0-08-044859-6.
The elimination half-life of AT-bound fondaparinux is 17–21 h (171,172). The subcutaneous bioavailability of fondaparinux is nearly 100% and it is distributed mainly in the blood (165,173).
- 1 2 3 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 139. ISBN 978-0857114105.
- ↑ "Fondaparinux Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 10 April 2021. Retrieved 13 December 2021.
- ↑ Yusuf S, Mehta SR, Chrolavicius S, Afzal R, Pogue J, Granger CB, et al. (Fifth Organization to Assess Strategies in Acute Ischemic Syndromes Investigators) (April 2006). "Comparison of fondaparinux and enoxaparin in acute coronary syndromes". The New England Journal of Medicine. 354 (14): 1464–1476. doi:10.1056/NEJMoa055443. hdl:2437/113091. PMID 16537663.
- ↑ Comp PC (June 2003). "Selective factor Xa inhibition improves efficacy of venous thromboembolism prophylaxis in orthopedic surgery". Pharmacotherapy. 23 (6): 772–87. doi:10.1592/phco.23.6.772.32190. PMID 12820819. S2CID 19516097. Archived from the original on 2019-08-15. Retrieved 2021-10-22.
- ↑ Dey S, Lo HJ, Wong CH (June 2020). "Programmable One-Pot Synthesis of Heparin Pentasaccharide Fondaparinux". Organic Letters. 22 (12): 4638–4642. doi:10.1021/acs.orglett.0c01386. PMC 7347301. PMID 32496799.
External links
| External sites: |
|
|---|---|
| Identifiers: |