HDMP-28
HDMP-28 or methylnaphthidate is a piperidine based stimulant drug, closely related to methylphenidate, but with the benzene ring replaced by naphthalene. It is a potent dopamine reuptake inhibitor, with several times the potency of methylphenidate and a short duration of action,[1] and is a structural isomer of another potent dopamine reuptake inhibitor, N,O-Dimethyl-4-(2-naphthyl)piperidine-3-carboxylate. It has been sold as a designer drug since around 2015.[2]
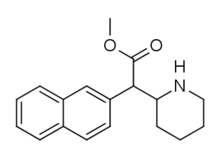 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C18H21NO2 |
| Molar mass | 283.365 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
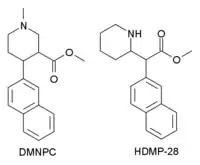
Most of the TMP analogs of HDMP-28 have SERT Ki values in the range >10,000 and so are selective for dopamine and noradrenaline reuptake, with little or no effect on serotonin. HDMP-28 has high affinity to SERT, and so behaves as a triple reuptake inhibitor.[3]
| Inhibition of specific analogs at displacing CFT from binding to DAT & RTI-55 from binding to SERT | ||||||
| Ar | [3H]CFT DAT | [3H]DA Uptake | [3H]RTI-55 SERT | Inhibition by 10μM | D.R. | Potency |
|---|---|---|---|---|---|---|
| Ph | 83.9 | 224 | >>10,000 | 19.6 | 2.7 | 1.00 |
| p-F | 35.0 | 142 | >10,000 | 36.9 | 4.1 | 3.33 |
| m-Cl | 5.1 | 23.0 | >10,000 | 45.5 | 4.5 | 2.42 |
| p-Me | 33.0 | 126 | >10,000 | 45.0 | 3.8 | 0.74 |
| p-NH2 | 34.5 | 114 | >>10,000 | 7.9 | 3.3 | 2.18 |
| m,p-Cl2 | 5.3 (2.67)b | 7.0 | 1,064 (>10,000)b | 93.3 | 1.3 | 7.98 |
| β-Naphthyl | 33.9b 11.0c | 53.0c | 71.6b | nd | 4.8c | — |
| Cocaine | 160 | 404 | 401 | nd | 2.5 | 0.41 |
| aSchweri, et al. (2002);[4] bDavies, et al. (2004);[5] cDeutsch, et al. (2001).[6] | ||||||
D.R. = Discrimination Ratio = [3H]DA ÷ [3H]CFT.
A low D.R. = addictive, whereas a high D.R. = low propensity for self-administration.
Legality
HDMP-28 is illegal in Switzerland as of December 2015.[7]
See also
References
- Lile JA, Wang Z, Woolverton WL, France JE, Gregg TC, Davies HM, Nader MA (October 2003). "The reinforcing efficacy of psychostimulants in rhesus monkeys: the role of pharmacokinetics and pharmacodynamics". The Journal of Pharmacology and Experimental Therapeutics. 307 (1): 356–66. doi:10.1124/jpet.103.049825. PMID 12954808. S2CID 5654856.
- Carlier J, Giorgetti R, Varì MR, Pirani F, Ricci G, Busardò FP. Use of cognitive enhancers: methylphenidate and analogs. Eur Rev Med Pharmacol Sci. 2019 Jan;23(1):3-15. doi:10.26355/eurrev_201901_16741 PMID 30657540
- Davies HM, Hopper DW, Hansen T, Liu Q, Childers SR (April 2004). "Synthesis of methylphenidate analogues and their binding affinities at dopamine and serotonin transport sites". Bioorganic & Medicinal Chemistry Letters. 14 (7): 1799–802. doi:10.1016/j.bmcl.2003.12.097. PMID 15026075.
- Schweri MM, Deutsch HM, Massey AT, Holtzman SG (May 2002). "Biochemical and behavioral characterization of novel methylphenidate analogs". The Journal of Pharmacology and Experimental Therapeutics. 301 (2): 527–35. doi:10.1124/jpet.301.2.527. PMID 11961053.
- Davies HM, Hopper DW, Hansen T, Liu Q, Childers SR (April 2004). "Synthesis of methylphenidate analogues and their binding affinities at dopamine and serotonin transport sites". Bioorganic & Medicinal Chemistry Letters. 14 (7): 1799–802. doi:10.1016/j.bmcl.2003.12.097. PMID 15026075.
- Deutsch HM, Ye X, Shi Q, Liu Z, Schweri MM (April 2001). "Synthesis and pharmacology of site specific cocaine abuse treatment agents: a new synthetic methodology for methylphenidate analogs based on the Blaise reaction". European Journal of Medicinal Chemistry. 36 (4): 303–11. doi:10.1016/s0223-5234(01)01230-2. PMID 11461755.
- "Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien". Der Bundesrat.