Fotemustine
Fotemustine is a nitrosourea alkylating agent used in the treatment of metastatic melanoma. It is available in Europe but has not been approved by the United States FDA.[1] A study[2] has shown that fotemustine produces improved response rates and but does not increase survival (over dacarbazine in the treatment of disseminated cutaneous melanoma. Median survival was 7.3 months with fotemustine versus 5.6 months with DTIC (P=.067). There was also toxicity prevalence in fotemustine arm. The main toxicity was grade 3 to 4 neutropenia (51% with fotemustine v 5% with DTIC) and thrombocytopenia (43% v 6%, respectively).
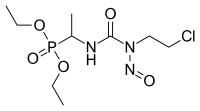 | |
| Clinical data | |
|---|---|
| Trade names | Mustophoran |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| ECHA InfoCard | 100.158.792 |
| Chemical and physical data | |
| Formula | C9H19ClN3O5P |
| Molar mass | 315.69 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
External links
- Menaa, Farid (2013). "Latest Approved Therapies for Metastatic Melanoma: What Comes Next?". Journal of Skin Cancer. 2013: 1–10. doi:10.1155/2013/735282. PMC 3595667. PMID 23533766.
- Avril MF, Aamdal S, Grob JJ, Hauschild A, Mohr P, Bonerandi JJ, Weichenthal M (March 2004). "Fotemustine Compared With Dacarbazine in Patients With Disseminated Malignant Melanoma: A Phase III Study". Journal of Clinical Oncology. 22 (6): 1118–25. doi:10.1200/JCO.2004.04.165. PMID 15020614.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.