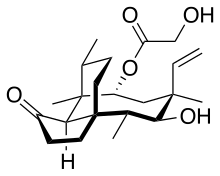
Pleuromutilin
Pleuromutilin and its derivatives are antibacterial drugs that inhibit protein synthesis in bacteria by binding to the peptidyl transferase component of the 50S subunit of ribosomes.[1][2]
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(3aS,4R,5S,6S,8R,9R,9aR,10R)-6-Ethenyl-5-hydroxy-4,6,9,10-tetramethyl-1-oxodecahydro-3a,9-propanocyclopenta[8]annulen-8-yl hydroxyacetate | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.004.316 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C22H34O5 |
| Molar mass | 378.509 g/mol |
| Melting point | 170-171 °C |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
This class of antibiotics includes the licensed drugs lefamulin (for systemic use in humans), retapamulin (approved for topical use in humans), valnemulin and tiamulin (approved for use in animals) and the investigational drug azamulin.
History
Pleuromutilin was discovered as an antibiotic in 1951.[3][4] It is derived from the fungi Omphalina mutila (formerly Pleurotus mutilus) and Clitopilus passeckerianus (formerly Pleurotus passeckerianus),[3] and has also been found in Drosophila subatrata, Clitopilus scyphoides, and some other Clitopilus species.[5]
Biosynthesis
Pleuromutilin belongs to the class of secondary metabolites known as terpenes, which are produced in fungi through the mevalonate pathway (MEP pathway).[10] Its synthetic bottleneck lays on the production of the precursor GGPP and the following formation of the tricyclic structure, which is catalyzed by Pl-cyc, a bifunctional diterpene synthase (DTS). This Cyclase shows a new class II DTS activity, catalyzes a ring contraction and the formation of a 5-6-bicyclic ring structure. Specifically, DTS shows two catalytic distinguishable domains: On the one hand it has at the N-terminal region a class II DTS domain, which catalyzes a cascade cyclization, resulting in a decalin core. Subsequently, variable 1,2-proton and methyl shifts occur to translocate the carbocation towards one of the two interconnecting C-atoms and this intermediate induces a base-catalyzed ring contraction. Therefore, class II DTS promotes in general a ring contraction during the cyclisation of GGPP. On the other hand, at the C-terminal end it has a class I DTS domain, which catalyzes a conjugated dephosphorylation, generating the 8-membered cyclic core, followed by a 1,5-proton shift and a stereospecific hydroxylation to obtain premutilin.[11]
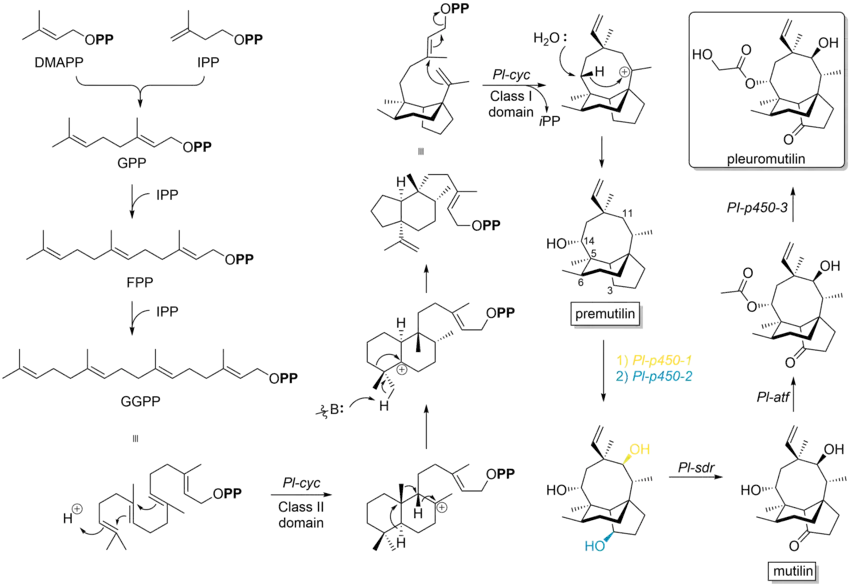
Additionally, three cytochrome P450s (Pl-p450-1, Pl-p450-2 and Pl-p450-3) are involved in the final steps of the pleuromutilin biosynthetic pathway.[14] The P450-1 and P450-2 are essential for hydroxylation of two ring structures regarding the premutilin skeleton, oxidating specifically at position C-11 and C-3, respectively. The short-chain dehydrogenase/reductase enzyme (Pl-sdr) has a regiospecific activity and converts the 3-hydroxy group to a ketone, forming the intermediate mutilin. Acetyltransferase (Pl-atf) catalyzes the transfer of acetyl group to 14-OH of mutilin. Finally, Pl-p450-3 hydroxylates the α-methyl group of the acetyl side chain generating pleuromutilin.[14][15]
References
- Maffioli, Sonia Ilaria (2013). "A chemist's survey of different antibiotic classes". In Gualerzi, Claudio O.; Brandi, Letizia; Fabbretti, Attilio; Pon, Cynthia L. (eds.). Antibiotics: Targets, Mechanisms and Resistance. Wiley-VCH. pp. 1–22. doi:10.1002/9783527659685.ch1. ISBN 978-3-527-65968-5. S2CID 11479628.
- Eyal Z, Matzov D, Krupkin M, Paukner S, Riedl R, Rozenberg H, Zimmerman E, Bashan A, Yonath A (Dec 2016). "A novel pleuromutilin antibacterial compound, its binding mode and selectivity mechanism". Sci Rep. 6(39004). doi:10.1038/srep39004. PMID 27958389.
- Kavanagh F, Hervey A, Robbins WJ (September 1951). "Antibiotic substances from basidiomycetes: VIII. Pleurotus multilus (Fr.) Sacc. and Pleurotus passeckerianus Pilat". Proceedings of the National Academy of Sciences of the United States of America. 37 (9): 570–574. Bibcode:1951PNAS...37..570K. doi:10.1073/pnas.37.9.570. PMC 1063423. PMID 16589015.
- Novak R, Shlaes DM (February 2010). "The pleuromutilin antibiotics: a new class for human use". Current Opinion in Investigational Drugs. 11 (2): 182–91. PMID 20112168. S2CID 41588014.
- Kilaru S, Collins CM, Hartley AJ, Bailey AM, Foster GD (November 2009). "Establishing molecular tools for genetic manipulation of the pleuromutilin-producing fungus Clitopilus passeckerianus". Applied and Environmental Microbiology. 75 (22): 7196–204. Bibcode:2009ApEnM..75.7196K. doi:10.1128/AEM.01151-09. PMC 2786515. PMID 19767458. S2CID 22127410.
- Gibbons, E. Grant (1982). "Total synthesis of (±)-pleuromutilin". Journal of the American Chemical Society. 104 (6): 1767–1769. doi:10.1021/ja00370a067. S2CID 102155530.
- Boeckman, Robert K.; Springer, Dane M.; Alessi, Thomas R. (1989). "Synthetic studies directed toward naturally occurring cyclooctanoids. 2. A stereocontrolled assembly of (±)-pleuromutilin via a remarkable sterically demanding oxy-Cope rearrangement". Journal of the American Chemical Society. 111 (21): 8284–8286. doi:10.1021/ja00203a043. S2CID 96627402.
- Fazakerley NJ, Helm MD, Procter DJ (May 2013). "Total synthesis of (+)-pleuromutilin". Chemistry. 19 (21): 6718–23. doi:10.1002/chem.201300968. PMID 23589420. S2CID 46105984.
- Murphy SK, Zeng M, Herzon SB (June 2017). "A modular and enantioselective synthesis of the pleuromutilin antibiotics". Science. 356 (6341): 956–959. Bibcode:2017Sci...356..956M. doi:10.1126/science.aan0003. PMC 7001679. PMID 28572392. S2CID 206658420.
- Lange BM, Rujan T, Martin W, Croteau R (November 2000). "Isoprenoid biosynthesis: the evolution of two ancient and distinct pathways across genomes". Proceedings of the National Academy of Sciences of the United States of America. 97 (24): 13172–7. Bibcode:2000PNAS...9713172M. doi:10.1073/pnas.240454797. PMC 27197. PMID 11078528.
- Lin HC, Hewage RT, Lu YC, Chooi YH (January 2019). "Biosynthesis of bioactive natural products from Basidiomycota". Organic & Biomolecular Chemistry. 17 (5): 1027–1036. doi:10.1039/C8OB02774A. PMID 30608100.
- Birch, A.J.; Holzapfel, C.W.; Rickards, R.W. (January 1966). "The structure and some aspects of the biosynthesis of pleuromutilin". Tetrahedron. 22: 359–387. doi:10.1016/S0040-4020(01)90949-4.
- Arigoni D (1968-01-01). "Some studies in the biosynthesis of terpenes and related compounds". Pure and Applied Chemistry. 17 (3): 331–48. doi:10.1351/pac196817030331. PMID 5729285. S2CID 34616923.
- Alberti F, Khairudin K, Venegas ER, Davies JA, Hayes PM, Willis CL, et al. (November 2017). "Heterologous expression reveals the biosynthesis of the antibiotic pleuromutilin and generates bioactive semi-synthetic derivatives". Nature Communications. 8 (1): 1831. Bibcode:2017NatCo...8.1831A. doi:10.1038/s41467-017-01659-1. PMC 5705593. PMID 29184068.
- Bailey AM, Alberti F, Kilaru S, Collins CM, de Mattos-Shipley K, Hartley AJ, et al. (May 2016). "Identification and manipulation of the pleuromutilin gene cluster from Clitopilus passeckerianus for increased rapid antibiotic production". Scientific Reports. 6 (1): 25202. Bibcode:2016NatSR...625202B. doi:10.1038/srep25202. PMC 4855138. PMID 27143514.
Further reading
- Long KS, Hansen LH, Jakobsen L, Vester B (April 2006). "Interaction of pleuromutilin derivatives with the ribosomal peptidyl transferase center". Antimicrobial Agents and Chemotherapy. 50 (4): 1458–62. doi:10.1128/AAC.50.4.1458-1462.2006. PMC 1426994. PMID 16569865. S2CID 8900413.
- Lolk L, Pøhlsgaard J, Jepsen AS, Hansen LH, Nielsen H, Steffansen SI, et al. (August 2008). "A click chemistry approach to pleuromutilin conjugates with nucleosides or acyclic nucleoside derivatives and their binding to the bacterial ribosome". Journal of Medicinal Chemistry. 51 (16): 4957–67. doi:10.1021/jm800261u. PMID 18680270. S2CID 931146.