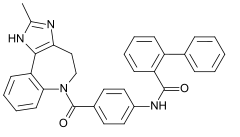
Conivaptan
 | |
 | |
| Names | |
|---|---|
| Trade names | Vaprisol |
| Other names | YM 087 |
IUPAC name
| |
| Clinical data | |
| Drug class | Vasopressin receptor antagonist.[1] |
| Main uses | Low sodium[1] |
| Side effects | Pain at site of injection, fever, low potassium, headache, low blood pressure with standing[1] |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Pregnancy category |
|
| Routes of use | Intravenous |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| License data |
|
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | N/A |
| Chemical and physical data | |
| Formula | C32H26N4O2 |
| Molar mass | 498.586 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Conivaptan, sold under the brand name Vaprisol, is a medication used to treat low sodium in those who either have normal or high total body fluid.[1] While it may improve lab values, it is unclear if it improves outcomes.[1] It is given by injection into a vein.[1]
Common side effects include pain at the site of injection, fever, low potassium, headache, and low blood pressure with standing.[1] Other side effects may include osmotic demyelination syndrome if sodium is corrected too rapidly.[2] Safety in pregnancy is unclear as is safety in heart failure.[2] It is a vasopressin receptor antagonist.[1]
Conivaptan was approved for medical use in the United States in 2005.[1] It has not been approved in the United Kingdom as of 2011.[3] In the United States it costs nearly 1,000 USD per 20 mg dose as of 2022.[4]
Medical uses
Conivaptan is most commonly used in the hospital in cases of euvolemic and hypervolemic hyponatremia, conditions where the sodium level in the blood falls significantly below normal. In the United States hyponatremia affects about four percent of hospitalized patients. Although many patients do not show symptoms, extreme cases may result in brain swelling, respiratory arrest and even death. Hypervolemic hyponatremia specifically is when the body’s serum sodium levels fall below the total body water increase, which results in edema. This is associated with congestive heart failure, liver disease and kidney failure.[5]
Dosage
It is given as an initial dose of 20 mg over 30 minutes followed by 20 mg/day over 24 hours for 2 to 4 days.[1]
Side effects
Most side effects are found at the site of infusion. Other complications include blood and lymphatic system disorders, gastrointestinal disorders, metabolism and nutrition disorders, psychiatric disorders as well as vascular disorders.[5] There is risk of too rapid correction of low sodium causing osmotic demyelination syndrome.[6]
Pharmacology
Conivaptan is an arginine vasopressin (AVP) receptor antagonist with affinity for human V1A and V2 receptors in the nanomolar range in vitro. AVP levels in blood are crucial for water and electrolyte regulation and balance. V2 receptors maintain plasma osmolality and are coupled to aquaporin channels in the collecting ducts of kidneys where they regulate AVP levels. Conivaptan hydrochloride functions by antagonizing V2 receptors in the renal collecting ducts and thus causing aquaresis or water secretion. Typical pharmacodynamic effects of the drug are an increase in net fluid loss, increase in urine output, and decrease in the osmolality of urine. Conivaptan inhibits its own metabolism in the body, displaying non-linear pharmacokinetics. About 99% of conivaptan found is bound to human plasma proteins over the range of 10 ng/mL to 1000 ng/mL. The mean half-life of the drug is 5 hours and mean clearance is 15.2 L/hr.[5]
Chemistry
Conivaptan hydrochloride is an off-white or a pale yellow power with a solubility of 0.25 mg/mL in water at 23 °C. The injectable formulation consists of 20 mg conivaptan hydrochloride, 0.4 g ethanol, 1.2 g propylene glycol and water.[5]
History
Conivaptan hydrochloride was discovered by Yamanouchi Pharmaceuticals and marketed by Astellas. Yamanouchi Pharmaceuticals filed a new drug application to the FDA for conivaptan hydrochloride to treat hyponatremia on 2 February 2004. On 1 December, Yamanouchi received approval for investigational hyponatremia treatment using conivaptan hydrochloride. Yamanouchi pharmaceuticals and Fujisawa Pharmaceuticlas merged and were renamed as Astellas’ in 2005. Astellas gained FDA approval for Conivaptan hydrochloride on 29 December 2005, and marked the drug under the brand name of Vaprisol in 2006.[7]
On 2 March 2007, vaprisol also gained FDA approval for the treatment of hypervolemia hyponatremia. On 22 October 2008, Vaprisol further gained approval as a 5% Dextrose premixed formulation for the treatment of hyponatremia. In 2014 Cumberland pharmaceuticals bought Vaprisol from Astellas and took full responsibility of all development and marketing of the drug.[7][8]
Cumberland Pharmaceuticals is a pharmaceutical company based in Nashville, Tennessee. In addition to Vaprisol, Cumberland produces Acetadote, Caldolor, Kristalose, Omeclamox, and Ethyol. Hepatoren, Boxban, Vasculan, and currently in phase II clinical trials. Protaban, Methotrexate, and Totect are waiting for pre-approval.[9][10]
References
- 1 2 3 4 5 6 7 8 9 10 "DailyMed - VAPRISOL DEXTROSE IN PLASTIC CONTAINER- conivaptan hydrochloride injection, solution". dailymed.nlm.nih.gov. Archived from the original on 21 April 2021. Retrieved 6 January 2022.
- 1 2 "Conivaptan Monograph for Professionals". Drugs.com. Archived from the original on 23 January 2021. Retrieved 7 January 2022.
- ↑ Gardner, Roy S.; Clark, Andrew L.; Dargie, Henry (14 July 2011). Oxford Textbook of Heart Failure. OUP Oxford. p. 154. ISBN 978-0-19-957772-9. Archived from the original on 11 January 2022. Retrieved 7 January 2022.
- ↑ "Vaprisol Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 14 April 2021. Retrieved 7 January 2022.
- 1 2 3 4 Astellas Pharma US, inc. Vaprisol. (2007).
- ↑ Goodman & Gilman's the pharmacological basis of therapeutics. Brunton, Laurence L.,, Knollmann, Björn C.,, Hilal-Dandan, Randa (Thirteenth ed.). [New York]. ISBN 9781259584732. OCLC 994570810.
{{cite book}}: CS1 maint: others (link) - 1 2 Vaprisol Approval History. Drugs.Com (2017). Available at: https://www.drugs.com/history/vaprisol.html Archived 29 November 2020 at the Wayback Machine. (Accessed: 3 November 2017)
- ↑ Inc., C. P. No TitleCumberland Pharmaceuticals Acquires Vaprisol® from Astellas Pharma US, Inc. Cision PR Newswire Available at: https://www.prnewswire.com/news-releases/cumberland-pharmaceuticals-acquires-vaprisol-from-astellas-pharma-us-inc-248179001.html Archived 18 August 2019 at the Wayback Machine.
- ↑ Cumberland Pharmaceuticals Inc. | Annual Report 2016. (2016).
- ↑ Inc., C. pharmaceuticals. Cumberland Business Milestones. (2017). Available at: http://www.cumberlandpharma.com/history Archived 1 March 2021 at the Wayback Machine. (Accessed: 3 November 2017)
External links
| External sites: |
|
|---|---|
| Identifiers: |