N-Methylspiperone
N-Methylspiperone (NMSP) is a derivate of spiperone that is used to study the dopamine and serotonin neurotransmitter systems. Labeled with the radioisotope carbon-11, it can be used for positron emission tomography.[1]
 | |
| Names | |
|---|---|
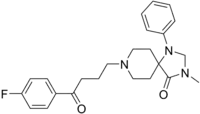
| IUPAC name
8-[4-(4-fluorophenyl)-4-oxobutyl]-3-methyl-1-phenyl-1,3,8-triazaspiro[4.5]decan-4-one | |
| Other names
3-N-Methylspiperone | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C24H28FN3O2 | |
| Molar mass | 409.505 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
References
- Bengt Andree; et al. (August 1998). "Positron Emission Tomographic Analysis of Dose-dependent MDL-100,907 Binding to 5-Hydroxtryptamine-2A Receptors in the Human Brain". Journal of Clinical Psychopharmacology. 18 (4): 313–323.
| 5-HT1 |
| ||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT2 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT3–7 |
| ||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.