BI 224436
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 7 hrs (simulated)[1] |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
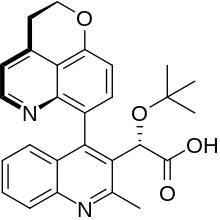
| Formula | C27H26N2O4 |
| Molar mass | 442.515 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
BI 224436 was an investigational new drug under development for the treatment of HIV infection. BI 224436 is the first non-catalytic site integrase inhibitor (NCINI). It inhibits HIV replication via binding to a conserved allosteric pocket of the HIV integrase enzyme. This makes the drug distinct in its mechanism of action compared to raltegravir and elvitegravir, which bind at the catalytic site.[2] In October 2011, Gilead Sciences purchased exclusive rights to develop BI 224436 and several related compounds under investigation in Boehringer Ingelheim’s noncatalytic site integrase inhibitor program.[3][4]
Clinical trials were abandoned in advance of Phase 1.[5]
References
- ↑ Brown A, McSharry J, Kulawy R (17 September 2011). Pharmacodynamics of BI 224436 for HIV-1 in an in vitro hollow fiber infection model system. 51st Interscience conference on antimicrobial agents and chemotherapy. Chicago. pp. 17–20.
- ↑ Fenwick C, Bethell R, Cordingley M, Edwards P, Quinson AM, Robinson P, Simoneau B, Yoakim C (17 September 2011). Levin J (ed.). BI 224436, a non-catalytic site integrase inhibitor, is a potent inhibitor of the replication of treatment-naïve and raltegravir-resistant clinical isolates of HIV-1. 51st Interscience Conference on Antimicrobials and Chemotherapy. Chicago: ICAAC.
- ↑ "Gilead Negotiates Worldwide License to BI's Early Clinical Stage HIV Program". Genetic Engineering and Biotechnology News. 6 October 2011. Archived from the original on 23 January 2013.
- ↑ Highleyman L (7 October 2011). "ICAAC: New Integrase Inhibitor BI 224436 Active against Raltegravir-Resistant HIV". HIVandHepatitis.com.
- ↑ Clinical trial number NCT01276990 for "Safety and Pharmacokinetics of Multiple Rising Oral Doses of BI 224436 in Healthy Male Volunteers." at ClinicalTrials.gov
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.