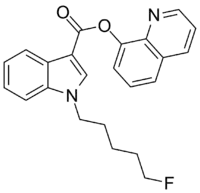
5F-PB-22
5F-PB-22 (5F-QUPIC or quinolin-8-yl 1-pentyfluoro-1H-indole-3-8-carboxylate) is a designer drug which acts as a cannabinoid agonist.[2] The structure of 5F-PB-22 appears to have been designed with an understanding of structure–activity relationships within the indole class of cannabinoids.[3]
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C23H21FN2O2 |
| Molar mass | 376.431 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Pharmacology
5F-PB-22 acts as a full agonist with a binding affinity of 0.468 nM at CB1 and 0.633 nM at CB2 cannabinoid receptors.[4]
Legal status
As of October 2015 5F-PB-22 is a controlled substance in China.[5]
In January 2014, 5F-PB-22 was designated as a Schedule I controlled substance in the United States after several deaths were associated with its use.[6][7]
In the United Kingdom, 5F-PB-22 is now classified and controlled as a Class B drug, following the November 2016 amendment to the Misuse of Drugs Act 1971. Several other synthetic cannabinoids structurally related to JWH-018, like 5F-PB-22, were also classified in this amendment.[8]
References
- Anvisa (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.
- Banister SD, Stuart J, Kevin RC, Edington A, Longworth M, Wilkinson SM, et al. (August 2015). "Effects of bioisosteric fluorine in synthetic cannabinoid designer drugs JWH-018, AM-2201, UR-144, XLR-11, PB-22, 5F-PB-22, APICA, and STS-135". ACS Chemical Neuroscience. 6 (8): 1445–58. doi:10.1021/acschemneuro.5b00107. PMID 25921407.
- "5F-PB-22". Forendex. Archived from the original on 27 September 2015. Retrieved 24 June 2015.
- Hess C, Schoeder CT, Pillaiyar T, Madea B, Müller CE (1 July 2016). "Pharmacological evaluation of synthetic cannabinoids identified as constituents of spice". Forensic Toxicology. 34 (2): 329–343. doi:10.1007/s11419-016-0320-2. PMC 4929166. PMID 27429655.
- "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in Chinese). China Food and Drug Administration. 27 September 2015. Archived from the original on 1 October 2015. Retrieved 1 October 2015.
- Behonick G, Shanks KG, Firchau DJ, Mathur G, Lynch CF, Nashelsky M, et al. (October 2014). "Four postmortem case reports with quantitative detection of the synthetic cannabinoid, 5F-PB-22". Journal of Analytical Toxicology. 38 (8): 559–62. doi:10.1093/jat/bku048. PMC 4334789. PMID 24876364.
- Trecki J, Gerona RR, Schwartz MD (July 2015). "Synthetic Cannabinoid-Related Illnesses and Deaths". The New England Journal of Medicine. 373 (2): 103–7. doi:10.1056/NEJMp1505328. PMID 26154784.
- "The Misuse of Drugs Act 1971 (Amendment) Order 2016". www.legislation.gov.uk. Retrieved 2017-12-20.