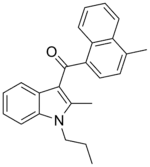
JWH-148
JWH-148 is a synthetic cannabimimetic that was discovered by John W. Huffman. It is the indole 2-methyl analog of JWH-120. It is a moderately selective ligand for the CB2 receptor, with a binding affinity of Ki = 14.0 ± 1.0 nM at this subtype, and more than eight times selectivity over the CB1 subtype.[1]
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C24H23NO |
| Molar mass | 341.454 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
In the United States, all CB1 receptor agonists of the 3-(1-naphthoyl)indole class such as JWH-148 are Schedule I Controlled Substances.[2]
References
- Huffman JW, Zengin G, Wu MJ, Lu J, Hynd G, Bushell K, et al. (January 2005). "Structure-activity relationships for 1-alkyl-3-(1-naphthoyl)indoles at the cannabinoid CB(1) and CB(2) receptors: steric and electronic effects of naphthoyl substituents. New highly selective CB(2) receptor agonists". Bioorganic & Medicinal Chemistry. 13 (1): 89–112. doi:10.1016/j.bmc.2004.09.050. PMID 15582455.
- : Schedules of controlled substances
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.