Acetothiolutamide
 | |
| Clinical data | |
|---|---|
| Other names | Thioacetolutamide |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
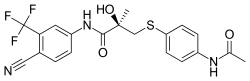
| Formula | C20H18F3N3O3S |
| Molar mass | 437.44 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Acetothiolutamide is a selective androgen receptor modulator (SARM) derived from the nonsteroidal antiandrogen bicalutamide that was described in 2002 and was one of the first SARMs to be discovered and developed.[1][2][3][4] It is a high-affinity, selective ligand of the androgen receptor (AR) (Ki = 2.1–4.9 nM), where it acts as a full agonist in vitro, and has in vitro potency comparable to that of testosterone.[2][4][5] However, in vivo, acetothiolutamide displayed overall negligible androgenic effects, though significant (albeit very low) anabolic effects were observed at high doses.[2] In addition, notable antiandrogen effects were observed in castrated male rats treated with testosterone propionate.[2] The discrepancy between the in vitro and in vivo actions of acetothiolutamide was determined to be related to rapid plasma clearance and extensive hepatic metabolism into a variety of metabolites with differing pharmacological activity, including AR partial agonism and antagonism.[2][4][6] In accordance with its poor metabolic stability, acetothiolutamide is not orally bioavailable, and shows activity only via injected routes such as subcutaneous and intravenous.[2]
See also
References
- ↑ Dalton JT, Mukherjee A, Zhu Z, Kirkovsky L, Miller DD (March 1998). "Discovery of nonsteroidal androgens". Biochemical and Biophysical Research Communications. 244 (1): 1–4. doi:10.1006/bbrc.1998.8209. PMID 9514878.
- 1 2 3 4 5 6 Yin D, Xu H, He Y, Kirkovsky LI, Miller DD, Dalton JT (March 2003). "Pharmacology, pharmacokinetics, and metabolism of acetothiolutamide, a novel nonsteroidal agonist for the androgen receptor". The Journal of Pharmacology and Experimental Therapeutics. 304 (3): 1323–33. doi:10.1124/jpet.102.040832. PMID 12604713. S2CID 6816508.
- ↑ Kearbey JD (2004), Preclinical Pharmacokinetics and Skeletal Pharmacology of a Selective Androgen Receptor Modulator
- 1 2 3 Perera MA, Yin D, Wu D, Chan KK, Miller DD, Dalton J (October 2006). "In vivo metabolism and final disposition of a novel nonsteroidal androgen in rats and dogs". Drug Metabolism and Disposition. 34 (10): 1713–21. doi:10.1124/dmd.106.009985. PMID 16815963. S2CID 14708913.
- ↑ Kim J, Wu D, Hwang DJ, Miller DD, Dalton JT (October 2005). "The para substituent of S-3-(phenoxy)-2-hydroxy-2-methyl-N-(4-nitro-3-trifluoromethyl-phenyl)-propionamides is a major structural determinant of in vivo disposition and activity of selective androgen receptor modulators". The Journal of Pharmacology and Experimental Therapeutics. 315 (1): 230–9. doi:10.1124/jpet.105.088344. PMID 15987833. S2CID 30799845.
- ↑ Yin D, Gao W, Kearbey JD, Xu H, Chung K, He Y, Marhefka CA, Veverka KA, Miller DD, Dalton JT (March 2003). "Pharmacodynamics of selective androgen receptor modulators". The Journal of Pharmacology and Experimental Therapeutics. 304 (3): 1334–40. doi:10.1124/jpet.102.040840. PMC 2040238. PMID 12604714.