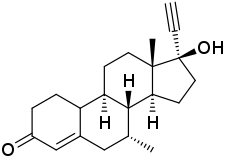
Δ4-Tibolone
 | |
 | |
| Clinical data | |
|---|---|
| Other names | ORG-OM-38; Delta-4-Tibolone; 7α-Methylnorethisterone; 7α-Methyl-17α-ethynyl-19-nortestosterone; 17α-Ethynyl-17β-hydroxy-7α-methyl-4-estren-3-one |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C21H28O2 |
| Molar mass | 312.453 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Δ4-Tibolone (developmental code name ORG-OM-38), also known as 7α-methylnorethisterone or as 7α-methyl-17α-ethynyl-19-nortestosterone, is a synthetic androgen and progestin which was never marketed.[1][2] The compound is a major active metabolite of tibolone, which itself is a prodrug of δ4-tibolone along with 3α-hydroxytibolone and 3β-hydroxytibolone (which, in contrast to δ4-tibolone, are estrogens).[1] Tibolone and δ4-tibolone are thought to be responsible for the androgenic and progestogenic activity of tibolone, while 3α-hydroxytibolone and 3β-hydroxytibolone are thought to be responsible for its estrogenic activity.[1]
See also
References
- 1 2 3 Kuhl H (2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration" (PDF). Climacteric. 8 Suppl 1: 3–63. doi:10.1080/13697130500148875. PMID 16112947. S2CID 24616324.
- ↑ Escande A, Servant N, Rabenoelina F, Auzou G, Kloosterboer H, Cavaillès V, Balaguer P, Maudelonde T (2009). "Regulation of activities of steroid hormone receptors by tibolone and its primary metabolites". J. Steroid Biochem. Mol. Biol. 116 (1–2): 8–14. doi:10.1016/j.jsbmb.2009.03.008. PMID 19464167. S2CID 18346113.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.