Roflumilast
Roflumilast, sold under the trade name Daxas among others, is a drug that acts as a selective, long-acting inhibitor of the enzyme phosphodiesterase-4 (PDE-4). It has anti-inflammatory effects and is used as an orally administered drug for the treatment of inflammatory conditions of the lungs such as chronic obstructive pulmonary disease (COPD).[7][8][9][10]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Daxas, Daliresp, Zoryve, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a611034 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, topical |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 79%[2][1][5][6] |
| Protein binding | 99%[2][1][5][6] |
| Metabolism | Hepatic via CYP1A2 & CYP3A4[2][1][5][6] |
| Elimination half-life | 17 hours (30 hours [active metabolite])[2][1][5][6] |
| Excretion | Urine (70%)[2][1][5][6] |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.210.960 |
| Chemical and physical data | |
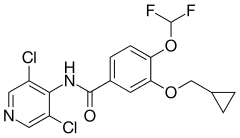
| Formula | C17H14Cl2F2N2O3 |
| Molar mass | 403.21 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
In June 2010, it was approved in the European Union for severe COPD associated with chronic bronchitis.[4][11] In February 2011, it gained FDA approval in the United States for reducing COPD exacerbations.[12][13]
Medical uses
Roflumilast is indicated for the treatment of severe chronic obstructive pulmonary disease (COPD)[2] and for the treatment of plaque psoriasis.[3][14]
It is used in the prevention of exacerbations (lung attacks) in severe chronic obstructive pulmonary disease (COPD).[1][2][4][5][6]
A topical formulation of Rolflumilast was recently approved by the U.S. Food and Drug Administration for plaque psoriasis.
Adverse effects
Common (1–10% incidence) adverse effects include:[2][1][5][6][15]
- Diarrhea
- Weight loss
- Nausea
- Headache
- Insomnia
- Decreased appetite
- Abdominal pain
- Rhinitis
- Sinusitis
- Urinary tract infection
- Depression
References
- "Daxas 250 micrograms tablets - Summary of Product Characteristics (SmPC)". (emc). 11 June 2020. Archived from the original on 19 September 2020. Retrieved 28 September 2020.
- "Daliresp- roflumilast tablet". DailyMed. 12 March 2020. Archived from the original on 25 March 2021. Retrieved 28 September 2020.
- "Archived copy" (PDF). Archived (PDF) from the original on 2022-08-02. Retrieved 2022-08-02.
{{cite web}}: CS1 maint: archived copy as title (link) - "Daxas EPAR". European Medicines Agency. Archived from the original on 12 August 2020. Retrieved 28 September 2020.
- "Daliresp : EPAR - Product Information" (PDF). European Medicines Agency. Takeda GmbH. 26 September 2013. Archived (PDF) from the original on 26 June 2016. Retrieved 18 November 2013.
- "roflumilast (Rx) - Daliresp". Medscape Reference. WebMD. Archived from the original on 12 September 2017. Retrieved 18 November 2013.
- Boswell-Smith V, Spina D (2007). "PDE4 inhibitors as potential therapeutic agents in the treatment of COPD-focus on roflumilast". International Journal of Chronic Obstructive Pulmonary Disease. 2 (2): 121–9. PMC 2695611. PMID 18044684.
- Herbert C, Hettiaratchi A, Webb DC, Thomas PS, Foster PS, Kumar RK (May 2008). "Suppression of cytokine expression by roflumilast and dexamethasone in a model of chronic asthma". Clinical and Experimental Allergy. 38 (5): 847–56. doi:10.1111/j.1365-2222.2008.02950.x. PMID 18307529. S2CID 19050454.
- Hohlfeld JM, Schoenfeld K, Lavae-Mokhtari M, Schaumann F, Mueller M, Bredenbroeker D, et al. (August 2008). "Roflumilast attenuates pulmonary inflammation upon segmental endotoxin challenge in healthy subjects: a randomized placebo-controlled trial" (PDF). Pulmonary Pharmacology & Therapeutics. 21 (4): 616–23. doi:10.1016/j.pupt.2008.02.002. PMID 18374614. Archived (PDF) from the original on 2019-08-28. Retrieved 2019-06-24.
- Field SK (May 2008). "Roflumilast: an oral, once-daily selective PDE-4 inhibitor for the management of COPD and asthma". Expert Opinion on Investigational Drugs. 17 (5): 811–8. doi:10.1517/13543784.17.5.811. PMID 18447606. S2CID 73241684.
- ""Nycomed's Anti-Inflammatory Gains Approval in EU for COPD"". Archived from the original on 2017-08-24. Retrieved 2010-07-10.
- "Drug Approval Package: Daliresp Tablets (roflumilast) NDA #022522". U.S. Food and Drug Administration (FDA). 24 December 1999. Archived from the original on 27 October 2020. Retrieved 28 September 2020. Lay summary (PDF).
{{cite web}}: Cite uses deprecated parameter|lay-url=(help) - "FDA approves new drug to treat chronic obstructive pulmonary disease" (Press release). U.S. Food and Drug Administration (FDA). March 1, 2011. Archived from the original on January 18, 2017. Retrieved December 16, 2019.
- "FDA Approves Arcutis' Zoryve (Roflumilast) Cream 0.3% For the Treatment of Plaque Psoriasis in Individuals Age 12 and Older" (Press release). Arcutis Biotherapeutics. 29 July 2022. Archived from the original on 1 August 2022. Retrieved 1 August 2022 – via GlobeNewswire.
- Spina D (October 2008). "PDE4 inhibitors: current status". British Journal of Pharmacology. 155 (3): 308–15. doi:10.1038/bjp.2008.307. PMC 2567892. PMID 18660825.