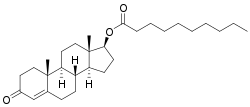
Testosterone decanoate
Testosterone decanoate (BAN) is an androgen and anabolic steroid and a testosterone ester.[1][2][3][4] It is a component of Sustanon, along with testosterone propionate, testosterone phenylpropionate, and testosterone isocaproate.[4][5] The medication has not been marketed as a single-drug preparation.[4] Testosterone decanoate has been investigated as a potential long-acting injectable male contraceptive.[4][6][7][8] It has a longer duration of action than testosterone enanthate, but its duration is not as prolonged as that of testosterone undecanoate.[4]
| Medication | Form | Major brand names | Duration |
|---|---|---|---|
| Testosterone | Aqueous suspension | Andronaq, Sterotate, Virosterone | 2–3 days |
| Testosterone propionate | Oil solution | Androteston, Perandren, Testoviron | 3–4 days |
| Testosterone phenylpropionate | Oil solution | Testolent | 8 days |
| Testosterone isobutyrate | Aqueous suspension | Agovirin Depot, Perandren M | 14 days |
| Mixed testosterone estersa | Oil solution | Triolandren | 10–20 days |
| Mixed testosterone estersb | Oil solution | Testosid Depot | 14–20 days |
| Testosterone enanthate | Oil solution | Delatestryl | 14–28 days |
| Testosterone cypionate | Oil solution | Depovirin | 14–28 days |
| Mixed testosterone estersc | Oil solution | Sustanon 250 | 28 days |
| Testosterone undecanoate | Oil solution | Aveed, Nebido | 100 days |
| Testosterone buciclated | Aqueous suspension | 20 Aet-1, CDB-1781e | 90–120 days |
| Nandrolone phenylpropionate | Oil solution | Durabolin | 10 days |
| Nandrolone decanoate | Oil solution | Deca Durabolin | 21–28 days |
| Methandriol | Aqueous suspension | Notandron, Protandren | 8 days |
| Methandriol bisenanthoyl acetate | Oil solution | Notandron Depot | 16 days |
| Metenolone acetate | Oil solution | Primobolan | 3 days |
| Metenolone enanthate | Oil solution | Primobolan Depot | 14 days |
| Note: All are via i.m. injection. Footnotes: a = TP, TV, and TUe. b = TP and TKL. c = TP, TPP, TiCa, and TD. d = Studied but never marketed. e = Developmental code names. Sources: See template. | |||
 | |
 | |
| Clinical data | |
|---|---|
| Other names | Testosterone decylate; Testosterone 17β-decanoate |
| Routes of administration | Intramuscular injection |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.024.752 |
| Chemical and physical data | |
| Formula | C29H46O3 |
| Molar mass | 442.684 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 49.1 °C (120.4 °F) |
SMILES
| |
InChI
| |
References
- Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 641–642. ISBN 978-1-4757-2085-3.
- Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. ISBN 978-3-88763-075-1.
- Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. ISBN 978-94-011-4439-1.
- Behre HM, Nieschlag E, Nieschlag E, Behre HM, Nieschlag S (26 July 2012). "Testosterone preparations for clinical use in males". In Nieschlag E, Behre HM, Nieschlag S (eds.). Testosterone: Action, Deficiency, Substitution. Cambridge University Press. pp. 309–335. doi:10.1017/CBO9781139003353.016. ISBN 978-1-107-01290-5.
- Fisher BA, Tilstone WJ, Woytowicz C (6 February 2009). Introduction to Criminalistics: The Foundation of Forensic Science. Academic Press. pp. 182–. ISBN 978-0-08-091675-0.
- Hay CJ, Brady BM, Zitzmann M, Osmanagaoglu K, Pollanen P, Apter D, et al. (April 2005). "A multicenter phase IIb study of a novel combination of intramuscular androgen (testosterone decanoate) and oral progestogen (etonogestrel) for male hormonal contraception". The Journal of Clinical Endocrinology and Metabolism. 90 (4): 2042–9. doi:10.1210/jc.2004-0895. PMID 15671109.
- Brady BM, Amory JK, Perheentupa A, Zitzmann M, Hay CJ, Apter D, et al. (January 2006). "A multicentre study investigating subcutaneous etonogestrel implants with injectable testosterone decanoate as a potential long-acting male contraceptive". Human Reproduction. 21 (1): 285–94. doi:10.1093/humrep/dei300. PMID 16172147.
- Chenoweth PJ, Lorton S (30 April 2014). Animal Andrology: Theories and Applications. CABI. pp. 488–. ISBN 978-1-78064-316-8.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.